in this video we'll be talking about cellular aging it's a slightly long video if you spend your 3 minutes you would be able to understand the overall concept and if you spend the entire time of this video you would have a detailed idea about cellular aging we always wonder why do we age and what controls aging can aging be reversed or slowed down these are the long-standing question in humankind scientists are debating among our the themselves to understand which part or organ in our body age at first when we talk about aging do we know
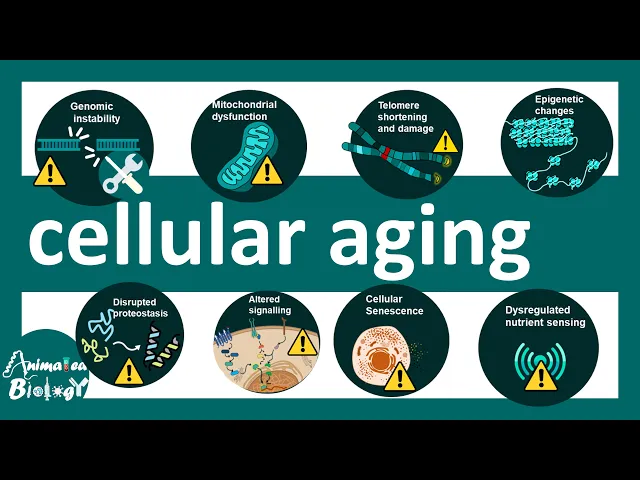
that whether our heart Age first or our liver Age first or is it the all cells of the body age due to specific stress factors or environmental factors are there genetics involved in it all these questions would be answered in this video so there are several physiological traits of cellular aging and in this video we would try to understand how genomic instability mitochondrial dysfunction telome shortening and damage epigenetic changes disrupted proteostasis altered cell signaling cellular cense and lastly disregulated nutrient sensing can affect the process of cellular aging so all in all the these are the
clinical uh signatures or Cardinal signatures of cellular aging all these factors are included or involved in the process of cellular aging now let's talk about which genes are actually involved in the process of Aging the category of genes that are involved encodes for DNA repair enzymes they encode proteins in the insulin signaling pathway they encode components of the mtor signaling pathway which regulates uh translation anabolism and many other metabolic fact processes in our body and also there are genes which are associated with chromatin remodeling enzymes so all of these G category of genes are associated
with aging if we take the example of Progeria which is a disease where aging is accelerated we would see that there is a genetic mutation in LMN a gene or lamin a gene this particular Gene give rise to the intermediate filament lamin a now lamin a works like a supportive sculpture to the nuclear envelope so obviously when there is a mutation in lamin a gene the product is not formed properly and there is a breakdown of the nuclear lamina and as a result there are several cells of our body especially the stem cells they die
and that leads to an accelerated aging in these individuals similarly we can take the example of Verner syndrome in this case the wrn gene which encodes for a protein involved in DNA repair and maintenance so obviously when this particular protein is not there DNA replication stalls and overall repair process is hampered in the circumstances cellular growth and cellular division capability is compromised that is how we understand that genomic instability is a key driver for cellular aging process now the idea about aging is actually in the genes and it turns out Recent research found out mutation
rate actually dictates the lifespan of individual initially the Greek philosophers thought bigger the animal longer it leaves for example a boohead wh basically lives for 200 years whereas a may fly live only one day so the size correlates with the age but it's not true think about a clamp that lives 500 years but it's a very small indiv small particular organism so age doesn't correlate with the lifespan or aging process so what really correlates and this recent article actually found out an answer it turns out that there are specific mutation rate that dictates the lifespan
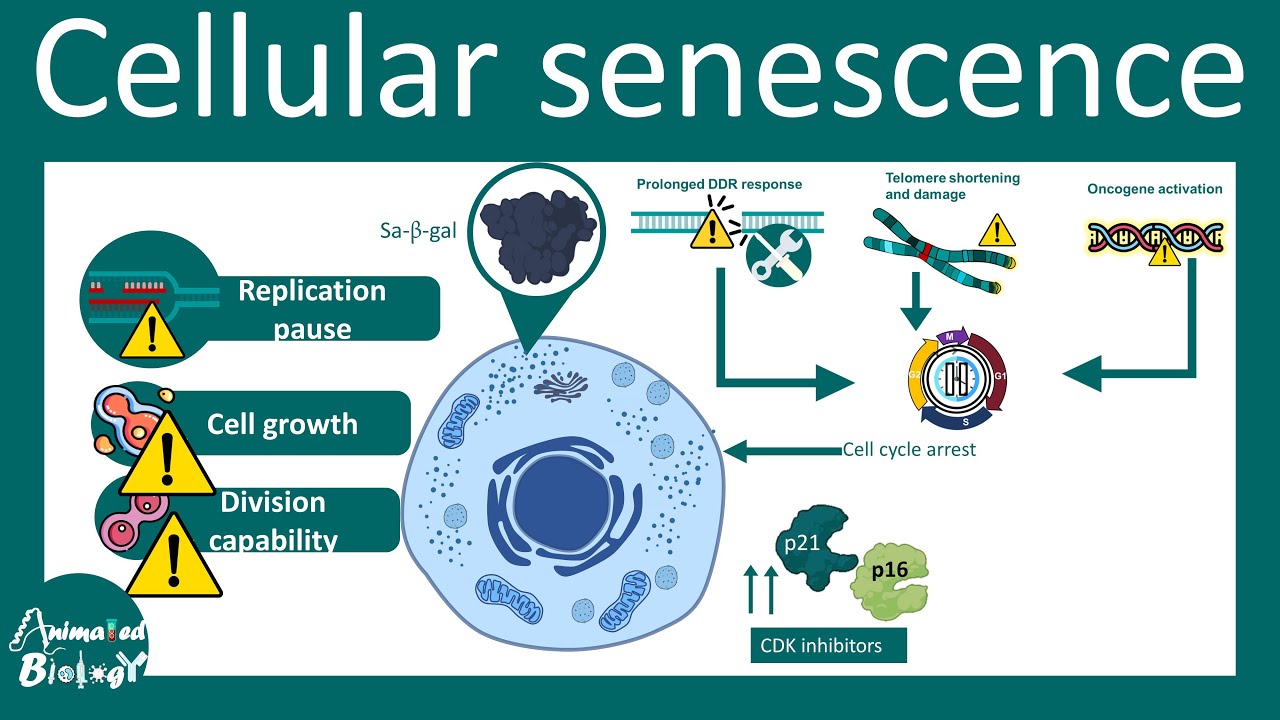
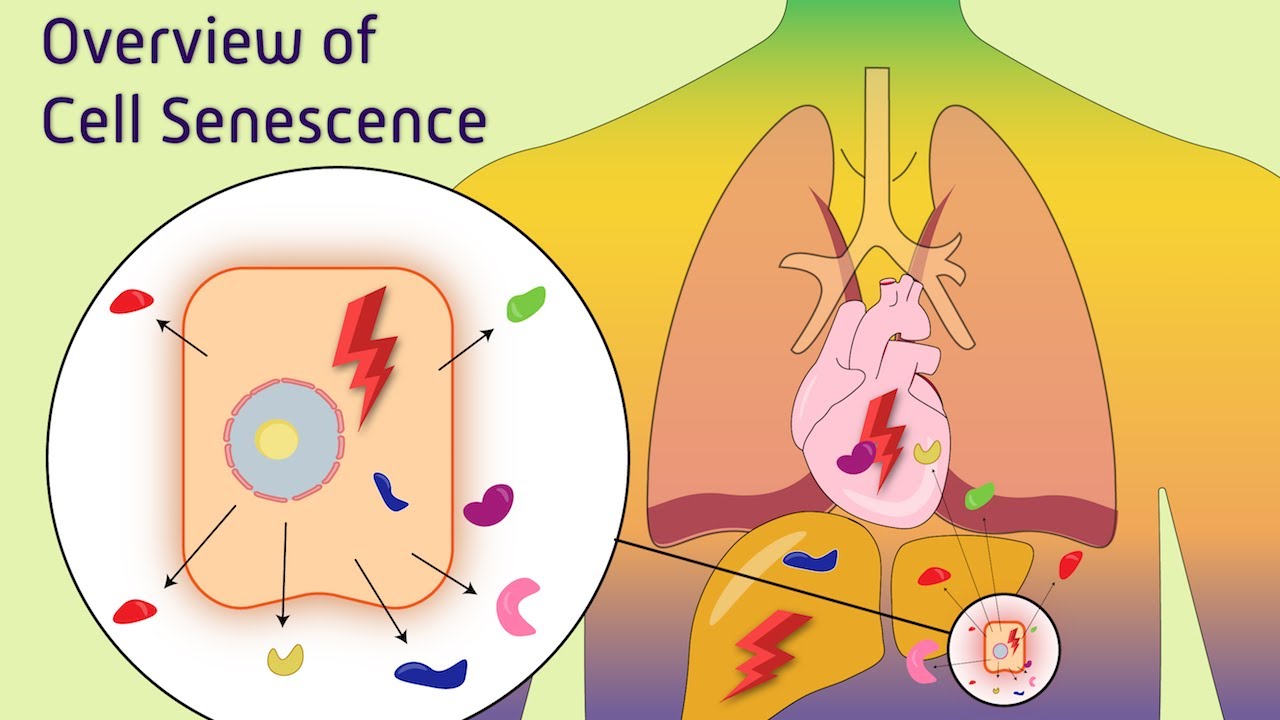
for example these giraffe and this uh mole they have basically different in size but their mutation rates are basically same that is why these naked black mole and the giraffe has comparable lifespan and this is one of the dramatic Discovery in recent times so now let's talk about another important cellular driver of um aging that is cellular cence so cence is a state of the cell where the cell under goes permanent growth arrest and it lose its all capability to divide further so cell growth and division capability is severely compromised in this cellular State now
let's talk about Hallmarks of cense so when we compare a normal cell with a senent cell there are different changes for example accumulation of losal enzyme termed as cense Associated beta galactosidase is very prominent feature of senin cells so question is what really discriminates between these senin cells and normal cells there are many other criterias for example there are morphological changes senin cells has abnormally larged cytoplasm so they have an increased cytoplasm to nucleus ratio they're also flattened in appearance another obvious marker of cense is the lack of DNA replication also increased level of p21
or p16 protein which prevents cycline cdk complexes are also a Hallmark feature of cellular cence people think that cellular cence is a key driver for aging process now this is how a normal nucle look like but in this case in a senin Cell the nucle somewhat looks like this this is known as nuclear cense Associated heterochromatin folky so all the chromatin are condensed in a different way in these cases now recent days transcriptomic approach and proteomic approach has found out several drivers of cence so let's talk about the inducers it turns out when there is
a prolonged double strand DNA break response when there is a tome shortening and damage when there is enogen activation all these key induction factors can lead to an arrest of the cell division and this arrest of cell division is a key feature of the cence so the cense features include obviously just to summarize the sense feature include nuclear associated heterochromatin foli cense associated beta galact todes or sa beta gal then there are several particular genes which are anti-apoptotic protein such as BCL XL bcl2 bclw all of their expression and all of their amount is increased
in the cells abnormally also cdk Inhibitors are upregulated in a senent Cell Ross levels are key driver to uh to perform the cense process and cellular aging so now let's talk about disregulated nutrient sensing for any cell to grow and divide it needs nutrient when nutrients are not a aable cell would not divide at all it is now found as so people have found out that now if the if the rats or many model organisms are fade in a restricted diet known as alternate day feeding then their lifespan has actually increased and there are many
research articles which suggest calorific restriction actually improves the health and overall lifespan in Reas monkeys flies or even Mouse so all these pre-clinical studies suggest that nutrient sensing and nutrient input where we eat and how how do we eat the diet composition all can actually dictate our aging process so there are lot of evidences from the world for example the the S the Okinawa island people the hunza tribe in Pakistan and also the the another tribe in Andes all of them has a average lifespan of greater than 100 years but what is common between them
all of them spiritually or ritually followed the intermitting intermittent fasting protocol that means they have a restricted feeding pattern or restricted calorie intake pattern there are many human studies which found out intermittent fasting is triggering weight loss and also improving cognitive uh uh benefits giving cognitive benefits in patients so question is why starving is so important because starving creates a stress situation on the body so obviously when you are not eating then there are lot of so the body has energy demand and how does body would basically overcome that energy demand because no nutrient is
coming from outside body would try to recycle what it has inside already and this process is known as autoag so in this process non-functional proteins or organal would be uh phagocytosed and degraded and that bits and pieces would be used to synthesize new proteins so let's say under the uh condition of starvation you need a functional enzyme so normal cases you would make it with the amino acids but when there is no dietary input in that circumstances you can't have external source of amino acids then how does amino acid I mean how does because one
can make functional enzyme that is required at that stressed environment autoag is the key autoag would break down non-functional proteins or protein aggregates to get these building blocks and this would further help to build new enzymes or proteins that are required for cellular function that is why autoag kind of recycles all of the existing products and that is working like an anti-aging mechanism and it turns out that in Alzheimer's disease amloid PLU can also be uh cleared out if autophagy is induced so these are still data from clinical pre-clinical trials so uh many order of
clinical trials are still left so when we need a functional enzyme um the pro the enzyme first has to be created from the amino acids so this is known as protein folding then eventually the protein has to be degraded as well now proteas mediated degradation or autophagic degradation is pretty common and if there is a problem in either degradation or synthesis that might lead to an aggregated protein so these aggregated protein are detrimental because they don't have they are not functional and they also prevent function of other proteins which are important for any biological processes
so obviously clearing out these aggregated proteins are important or refolding them into native conformation is important so this is known as proteo stasis any problem in any of these steps of this pathway can lead to Cellular damage and cellular aging so that is why disrupted proteostasis is a Hallmark of cellular aging another Hallmark that or another driver that lead to cellular aging is reactive oxygen species reactive oxygen species can can act on proteins and lipids they can they can oxidize lipid and lead to lipid peroxidation which damages the membrane ually they can also lead to
damage in the protein they can alter the protein structure thereby altering the proteins functionality moreover Ross can also create double strandard DNA damage and that lead to a overall problem in genomic instability all this thing uh leads to a vicious cycle that triggers the cellular aging process so overall in this video we looked at the key drivers or physiological traits that regulate cellular aging process so I hope this video was informative and good enough if you like this video give it a quick thumbs up don't forget to like share and subscribe you can support our
Channel using super thanks see you in next [Music] video