hi everyone welcome to IGCSE study buddy where you can revise biology topics from the Cambridge IGCSE syllabus this video summarizes topic 4 biological molecules the bodies of all living things are made of many different kinds of chemicals the three main categories of molecules in living organisms are carbohydrates fats and proteins since these molecules contain carbon they are known as organic molecules these are the chemical elements present in these molecules so as you can see carbohydrates fats and proteins all three contain carbon hydrogen and oxygen but only protein has nitrogen additionally and proteins even contain small
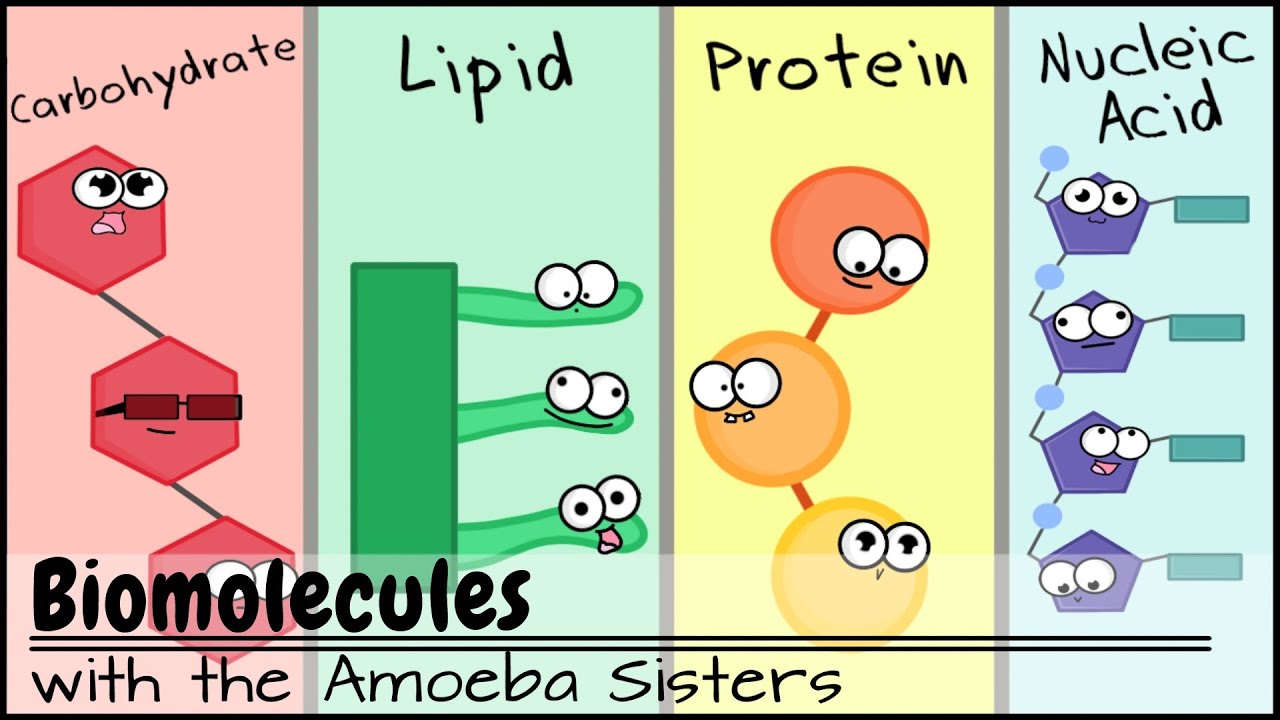
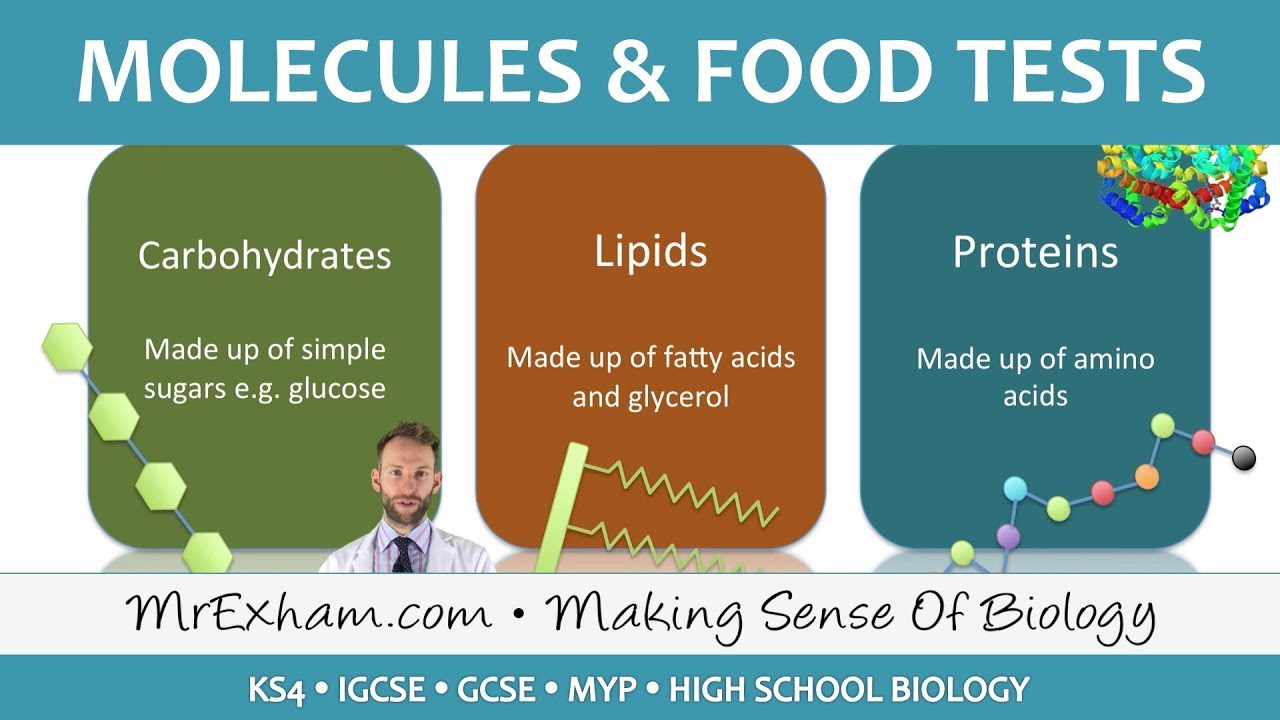
amounts of sulfur let's look into these chemical elements in more detail we need to know that large molecules are made from smaller molecules the first is carbohydrates they are large molecules made up of simple sugar molecules the simplest form of sugar molecules are also known as monosaccharides glucose is an example of a monosaccharide when two glucose molecules join together maltose is formed which is a disaccharact when lots of glucose molecules join together starch glycogen or cellulose can form they are called polysaccharides this picture illustrates the three types of carbohydrates a monosaccharide is a single sugar
molecule a disaccharide is when two sugar molecules are linked a polysaccharide is when many sugar molecules are linked examples of many glucose molecules join together are starch glycogen or cellulose the next category of biological molecules are fats or lipids fats are large molecules made from smaller units of fatty acids and glycerol their basic unit is one glycerol molecule chemically bonded to Three fatty acid shades fats that are liquids at room temperature are called oils so this picture shows that each fat molecule consists of one glycerol molecule bonded to Three fatty acid chains next is proteins
protein molecules are made of long chains of smaller molecules joined end to end these smaller molecules are called amino acids there are about 20 different amino acids the amino acids can be arranged in any order resulting in hundreds of thousands of different proteins so different combinations of these amino acids will give rise to different proteins even a small difference in the order of the amino acids results in a different protein being formed here's an illustration of amino acids that when combined forms a protein next we'll be looking into food tests different tests can be performed
on food to identify if they contain starch reducing sugars proteins fats and oils or vitamin C let's first take a look at the test for starch in order to find out if some food contains starch we should add drops of iodine solution to the food sample if the food sample contains touch the color will change from orange brown to blue black here's a picture to show this test as you can see a few drops of iodine are poured onto a potato since potatoes contain starch the orange brown color of the iodine turns blue black the
next food test is for reducing sugars we must first add Benedict solution into the food sample solution in the test tube Heat at 60 to 70 degrees Celsius in a water bath for 5 minutes take the test tube out of the water bath and observe the color if reducing sugars are present in the food sample the solution will turn from Blue to orange or brick red this diagram shows the food test for reducing sugars Benedict's solution is added to the food sample which is heated in a water bath and if the color changes from Blue
to Orange it confirms a positive test of reducing sugars the next food test is for proteins add drops of buy red solution to the food sample a positive test will show a color change from Blue to Violet or purple this illustration shows what happens when a food sample is tested for protein since the color changed from Blue to purple it means that the food sample contains protein now the test for fats and oils the food sample is mixed with two cubic centimeters of ethanol and shaken the ethanol is added to an equal volume of cold
distilled water a positive test will show a cloudy or milky Emulsion forming so if we want to check if a food sample contains fats or Oils we must mix the food sample with ethanol then we must add that to some cold water if the food sample contains fat cell oils a white cloudy Emulsion will form the final food test we'll be learning about is the test for vitamin C add 1 cubic centimeters of dcpip solution to a test tube dcpip is blue in color add a small amount of the food sample as a solution a
positive test will show the blue color of the dye disappearing this picture shows that when the blue dcpip solution is mixed with the food sample the blue color turns colorless and disappears this confirms that the food sample contains vitamin C finally we'll be learning about the structure of a DNA molecule DNA or deoxyribonucleic acid is the molecule that contains the instructions for growth and development of all organisms it consists of two strands of DNA wound around each other in what is called a double helix each strand contains chemicals called bases hydrogen bonds between Pairs of
bases hold the strands together the bases always pair up in the same way a with t or adenine with thymine and C with g that is cytosine with guanine full names are not required to be known for the exams just the letters but you must know which base bonds with which here's a picture to better understand the structure of a DNA molecule you may notice the double-stranded helix held together by base cats the base pairs are held together by hydrogen bonds the base pairs always pair up in a particular way a pairs with t and
c pairs with g so that sums up the main things to be learned in chapter 4 biological molecules hope you found it useful thank you for watching and please don't forget to subscribe to IGCSE study buddy for more biology revision videos bye