meet Bob he's thinking of participating in a clinical trial first off what is the clinical trial clinical trials are studies or investigations that use voluntary human participants to determine the clinical or pharmacological effects of a drug before testing on humans preclinical studies are done using tissue samples or animals if the results from the preclinical studies are promising the drug will advance to clinical trials where it's dosage effectiveness and safety in humans will be determined these clinical trials are held within the early phases of the drug development process before the drugs are approved and available to
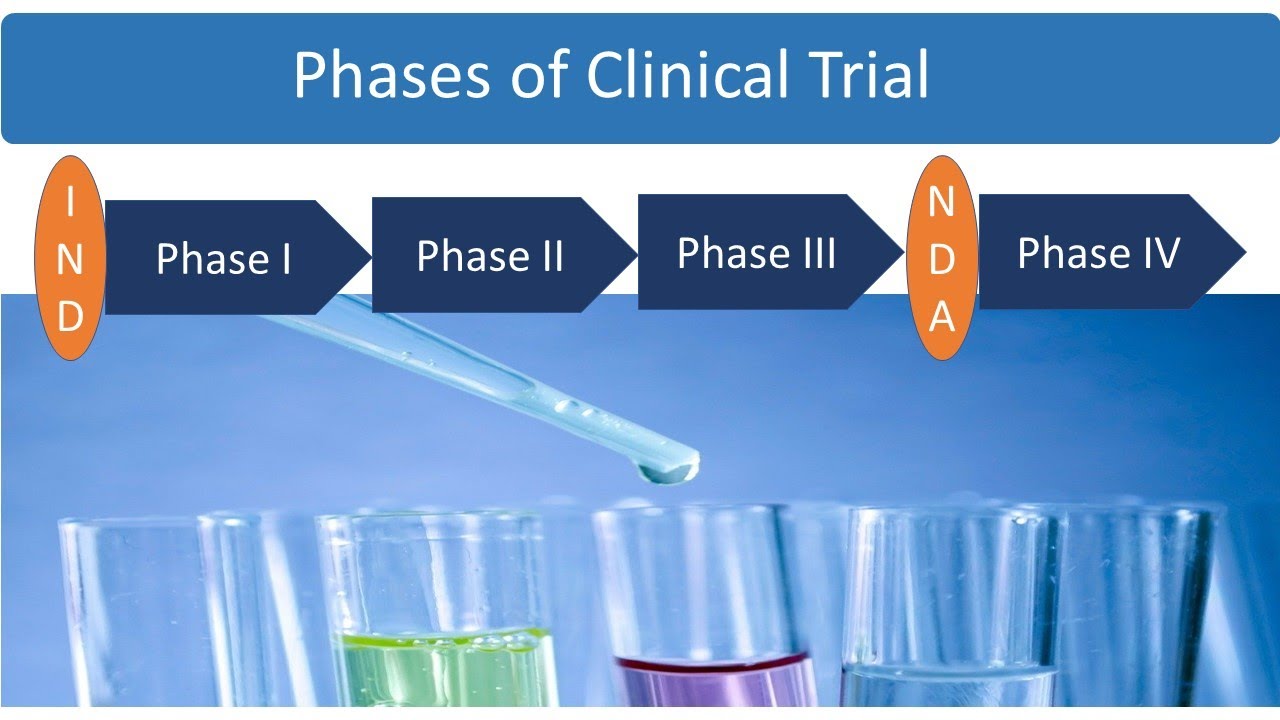
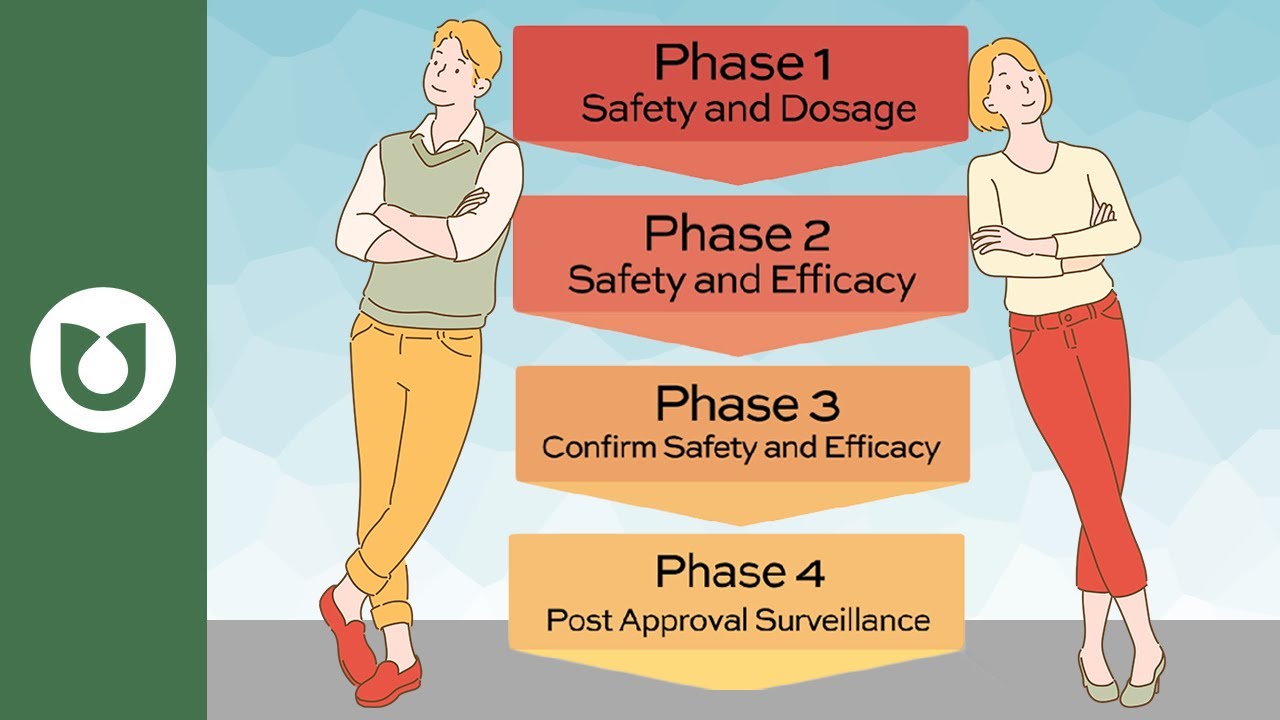
the general public so Bob you might be wondering what does a clinical trial entail in total there are four phases of a clinical trial each phase in a clinical trial is designed to answer specific research questions which are essential to observing the interactions of the drug in the human body phase one of a clinical trial tests the drug on a small group of people usually around 20 to 100 individuals to assess the safety of the drug an appropriate dosage phase two involves several hundred individuals with the condition for which the drug is being developed this
phase tests for additional safety and efficacy and can last several months - around two years during Phase three the trial opens up to a larger audience of hundreds to thousands of individuals the goal of this phase is to confirm the effectiveness of the drug and to continue to monitor any side effects the duration of Phase three typically ranges between one to four years finally phase four can only occur after Health Canada approves the marketing of the new drug at this time the drug is opened up to the entire population researchers will continue to study the
benefits and long-term effects associated with the use of the drug so Bob now that you know the process of clinical trials there are several factors you should consider by participating you would be gaining access to new drugs that are not yet available to the general public if these drugs are effective you'd be one of the first to benefit you would also be contributing to medical research by improving treatment for future patients however keep in mind that there may be drawbacks to participating new treatments may not always be better than existing ones and we therefore present
unexpected and potentially serious side effects the new therapy may also not be effective for some people and you may be spending more time at the hospital or study sites so when discussing with your family and friends make sure you weigh these factors before making a decision but don't worry Bob when you participate in a clinical trial in Canada you can rest assured that the Canadian government is actively working on protecting your safety not only is each clinical trial application reviewed by the government but their processes are continually monitored for example the government checks that drug
companies complete all necessary safety tests to reduce the risk of side effects they visit study sites to make sure patients are monitored properly by their doctors and trials are conducted appropriately they keep track of negative side effects as they occur and take action when needed if there are several side effects the government may interfere and stop the trial more importantly the Canadian government provides information on all phase one two and three drug patient clinical trials in Canada this is updated each night so you can make an informed decision about your participation we hope that this
helped you better understand the process of clinical trials and what it means to be a participant Bob