second law of thermodynamics on our way to becoming a thermodynamic energy specialist the second law is a little bit tricky and there's still people studying this law there's a lot to it the actual law of states establishes the concept of entropy as a physical properties of thermodynamic system so that doesn't really mean a lot to us so we can do is we're going to simplify this and we're going to simplify it to my definition of it just for hvac and i like the word it is this heat moves from a warmer place or substance to
a cooler less heated place or substance so it's moving from a warmer area to a cooler area that's how heat is going to work if they're both the same temperature heat's not going to move the bigger the temperature difference the faster heat's going to move we'll get into the speed of that shortly i was taught that it's called simply heat seeks cold we know that cold doesn't really exist but if you want to think of it heat seeks cold really it's better to think of as a cooler substance that's how heat is going to move
now it goes much deeper than this this is a simplified version to help you appreciate the second law of thermodynamics so if i want heat to move there needs to be a temperature difference to give you an example with this i have two pieces in the same room i have this rag and i have this piece of metal which one do you think is cooler hmm some of you may have said the rag and some of you may have said the metal but you're both wrong the answer is really going to be they're both the
same temperature they've both been in the same height in the same place in the same room at the same temperature they're now equalized the difference is my body temperature and the second law of thermodynamics so my body temperature is around 98.7 these are both in the same room at 75 degrees fahrenheit my body temperature is warmer than both of these components the difference is the heat moves from my body temperature through this material at a much slower rate this conducts heat at a much slower rate the metal conducts heat at a much faster rate so
when i touch the metal heat's leaving my body and going to the metal at a much faster rate so because the heat's leaving my body faster i perceive this one as being colder but reality the same temperature if you were to hold both of these long enough the heat would eventually replace itself i would heat up this metal i would heat up this component as well and it would start to fill the same temperature the smaller the metal the faster your body is going to heat that up so it's the heat movement a lot of
people talk about which is warmer a carpet floor or a wood floor and really the answer is kind of tricky it's really both the same temperature there is some some changes in that depending on what type of heat's underneath that there's air movement blah blah blah blah blah what type of insulation but let's think for the sake of argument they're both identical and identical type of soil they're both a wood floor and there's a carpet floor exactly together and you were to walk barefoot on one which would feel warmer and their carpet would feel warmer
but they're both the same temperature heat is going to leave my foot and go to that flat wood surface at a much faster rate than it would go into the carpet carpet is a type of insulator it slows heat transfer so when you talk about the difference in the two is how you feel not really the actual science behind it a lot of people say they don't like tile floors because they're cold but reality heats leaving their foot going to the floor at a faster rate so the temperature of their foot drops faster see the
difference this time we have to think about heat warmer than cooler than heat transfer so there's going to be three things that affect the speed of the second law of thermodynamics these all tie in together the movement of heat so this rag is made of material that resists heat transfer most insulation types are built with a type of material that slows or resists heat transfer such as fiberglass foam as well as dead airspace dead air pockets they resist heat transfer that's why you use them in the walls of houses they help slow down the movement
of heat other materials most metals also speed up or increase heat transfer if i took both of these metals and put them outside in the sunshine during the hottest part of the day they would both also end up being the same temperature as you touched the rag it wouldn't feel as bad because the heats leave leaving the rag into your body at a much slower rate the heat leaving the metal would be at a much faster rate and it would be burning and i would have to set it down so the material is a key
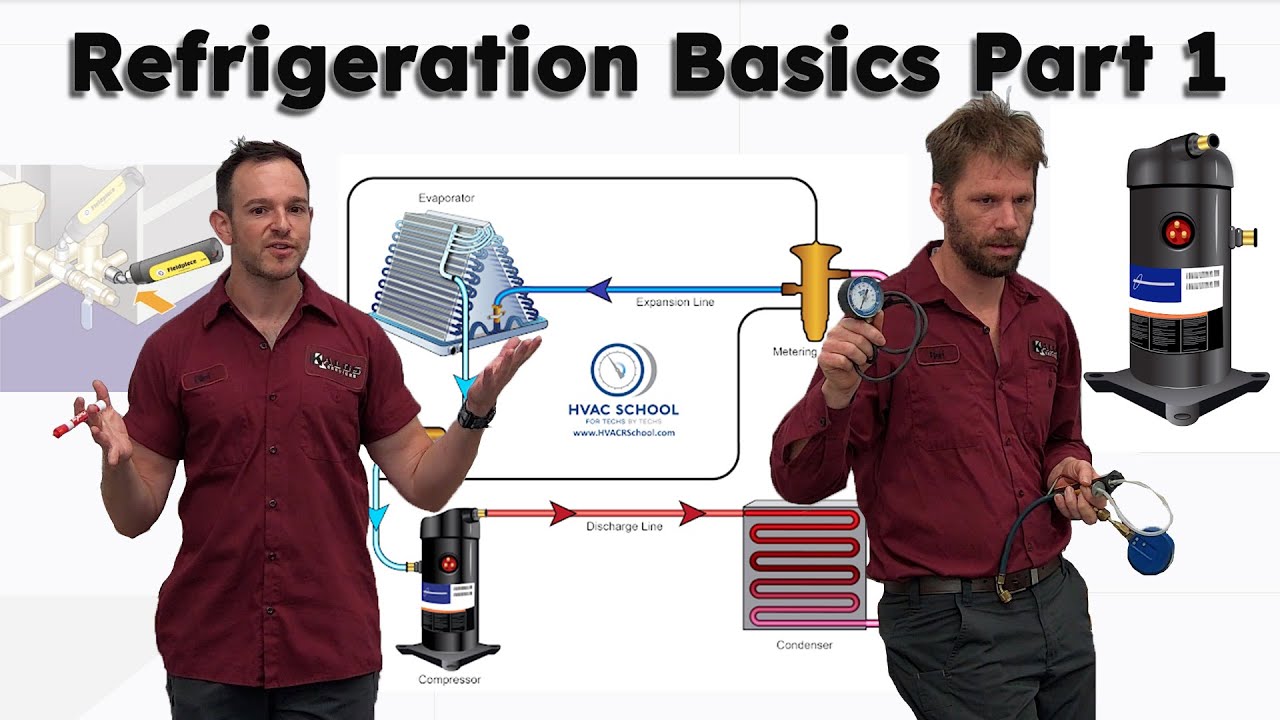
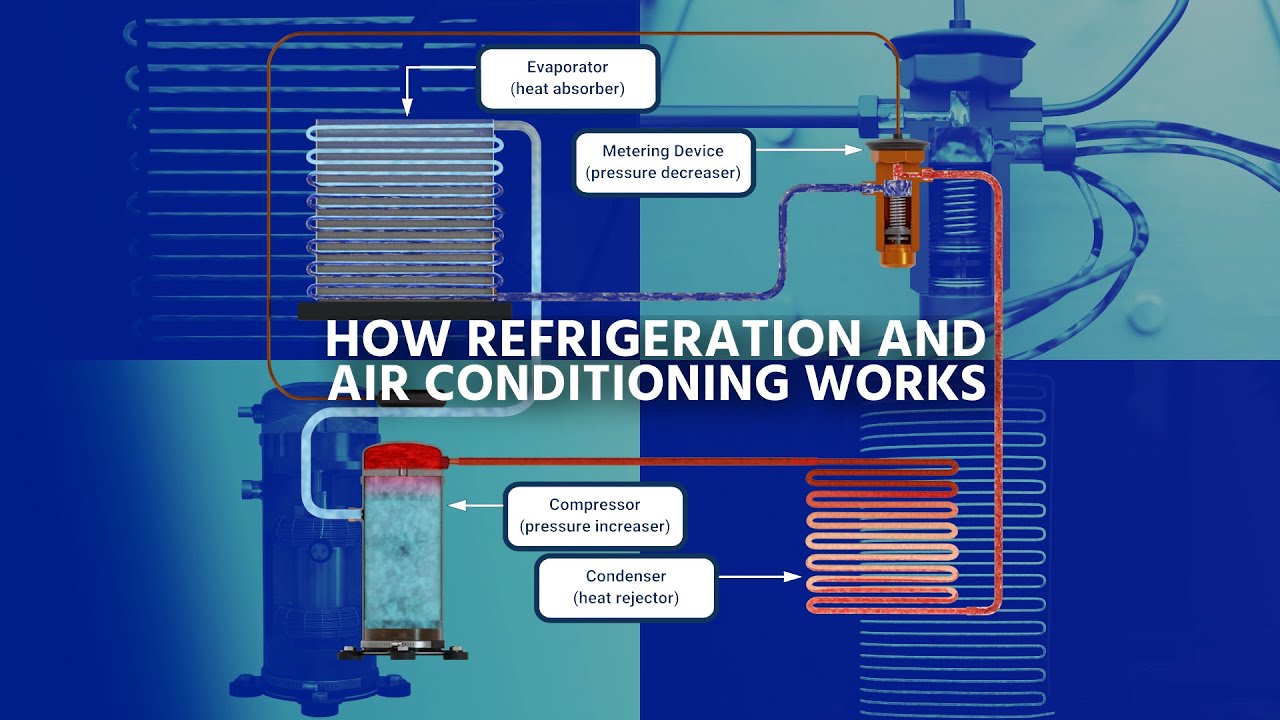
if i had the house one house built with fiberglass insulation and another house built straight with metal the metal house would be much warmer because it's transferring heat from the outside to the inside at a much faster rate so we use insulation to control heat transfer but other times i want to increase heat transfer and that's what we're going to talk about with different types of metals copper and aluminum work great for heat transfer fast so we use this evaporator coil inside of our house and also a condensing coil outside our house to transfer heat
so i'm going to run refrigerant through here and i need the temperature of the refrigerant to be lower than the temperature of the air heat travels from a warmer substance to a lower substance so if this is say 40 degrees and the air temperature is say 78 degrees the air temperature is warmer than the refrigerant so heat is going to leave the warmer air travel to the metal to the aluminum from the aluminum to the copper from the copper to the cooler refrigerant but i want that to be fast that's where different metals come in
if i built my evaporator coil out of a type of insulation it would not be picking up that heat as well so the material is very important sometimes i want a material with fast heat transfer sometimes i want a material that has much slower heat transfer very very important so the type of material plays a part in it another important part is the temperature difference the bigger the temperature difference the faster the heat is going to move so if you're up in the if you're in the northern part of the hemisphere during the winter time
the outdoor temperature is going to be much much lower than the house temperature so the heat's going to be leaving the house going outside as the outdoor air temperature drops heat will leave inside the house to the outside of the house at a much faster rate if the outdoor temperature is just a little bit lower than the indoor temperature heat's still going to travel but a whole lot slower so the material and also going to be the temperature difference let's think about the summertime it gets very very hot it's like think of the midwest for
example you're going to have an issue where you have it very very hot outside and it's going to be much cooler inside so let's say the inside temperature is 78 degrees the outside temperature is 100 degrees heat from outside is going to be coming in at a pretty fast rate big temperature difference heat's going to be moving through the insulation heating up the air inside this is where refrigeration comes in we've got to move it back to the outside so if i want to get heat out of the air i need my inside evaporative coil
to be a lower temperature to get heat to leave the air and go to the cooler refrigerant and outside i'll have another set of coils and i want to raise the temperature of that refrigerant so that heat leaves the refrigerant and goes to the cooler air outside so if it's 100 degrees outside i'll heat this up to say 120 115 degrees now heat's leaving the refrigerant going to the cooler air we're going to get more into that shortly so we know two things affect the speed of heat transfer the material type also the temperature difference
and the next one is surface area so if you think about surface area think about if you had a pie and you had that pie and you had two pies let's have two pies who didn't like two pies you have two of your favorite pies you take one of them and you cut it up into a whole bunch of little pieces and you separate all those pieces the other pie you get out all together which one's going to cool off faster and the answer is the one that's separated a little bitty pieces because there's more
surface area there's more of the air touching more of that pie on this side the pie would be all in one solid piece so there wouldn't be as much heat transfer this also is very important with refrigeration to get heat to move we need material surface area and temperature difference so the outside condensing coils nowadays are built a lot of times a lot larger than they used to be i have more surface say in other words there's more of this metal touching the refrigerant and there's also more of this metal touching the air larger surface
area there's also a temperature difference and the type of metal so outside of rejecting heat inside your absorbing heat a lot of our evaporator coils now they make them in different shapes but they want more surface area more of that surface area helps speed up or assist in heat transfer and a lot of times you can increase your surface area and reduce your temperature difference and have the same amount of heat moving but we're going to be moving heat from a place that's unwanted to a place that makes little no difference or a place that
we'd prefer it to be refrigeration moving heat from a place that's unwanted to somewhere else and that's all part of the second law of thermodynamics how we get heat to move moving heat from warmer place to a cooler place temperature difference surface area material type that affects the speed of the second law of thermodynamics