hi it's mr. Andersen and this is chemistry essentials video 69 it's on pH and buffers the proteins in our blood have a problem they have to have a specific pH and if it changes radically out of this range between seven point three five and seven point four five they start to denature and they can't do the job that they're intended to do which is to carry oxygen and carbon dioxide thankfully we can use a buffering system so what happens is the carbonic acid that it's created when we add carbon dioxide to the water is a
weak acid and it has a conjugate base and so that creates what's called a buffer solution what does that mean if we add more protons to it it'll simply push it towards the left and if we add more hydroxide to it it'll push it more towards the right and so it keeps our pH fairly stable and that's how solute buffer solutions work so pH remember is based on the protein availability it's the concentration of that proton in solution and so we want to keep that as stable as we can and so we use a buffer
solution to do it which is essentially a weak acid and its conjugate base and so what's going to affect the pH of that reversible reaction well the first thing is the pKa which is going to be the equilibrium constant and so if we can keep that equal to our pH or around our pH that's going to keep our pH stable and also we could look at the concentration of the acid that weak acid to its conjugate base if we can keep those values equal as well that's going to increase the buffer capacity and we'll look
at that algebraically in just a second but big picture what are we doing here well we've got a weak acid and a reversible reaction that forms this hydronium ion and then it's conjugate base and so in a good buffer solution we want the weak acid and the conjugate base to be equal in values and so what happens let's say we add hydronium to that let's say we add a strong acid to that well the shot leaves principle tells us if we add more of it on this side it's simply going to push it in the
other direction so it's going to push it more towards that weak ass side but since those values are equal it's not going to change it that much and our pH value is not going to change very much as well if we look at adding a base now if we add a hydroxide to it what is it going to do simply going to push it more towards the right and so what happens is we can add strong acids and we can add strong bases and it's can to keep that pH around a stable set point and
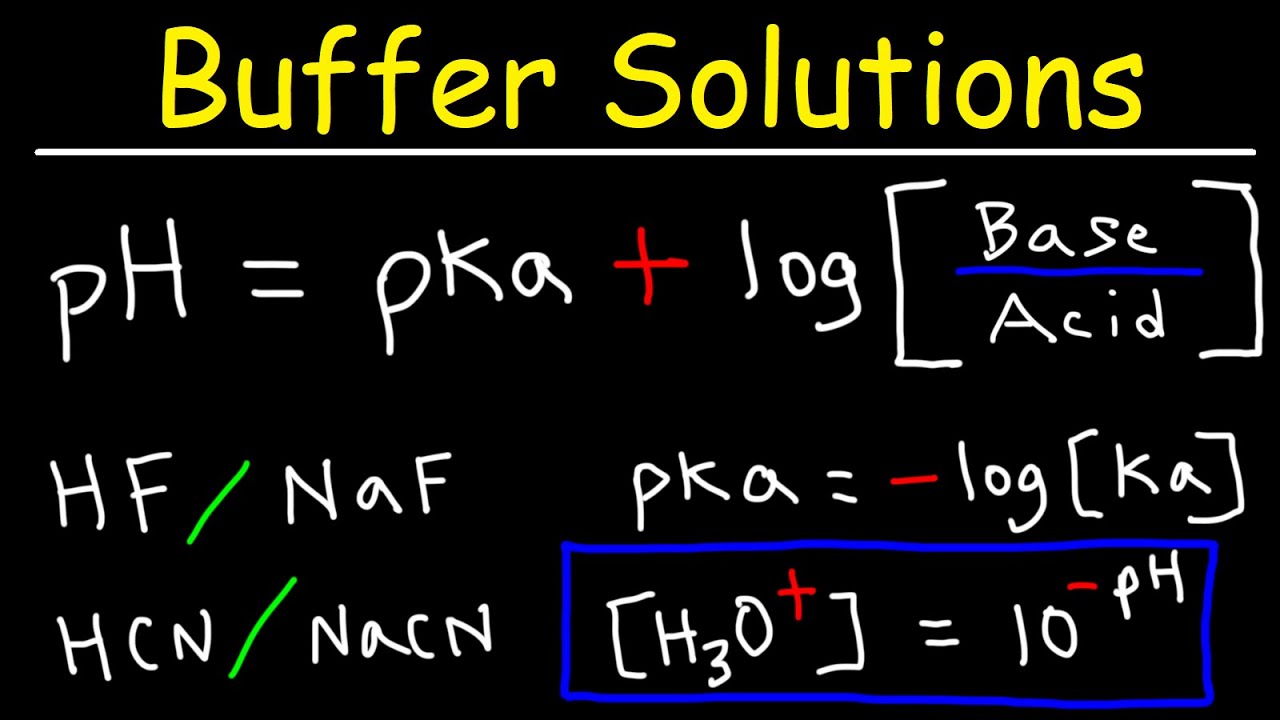
so let's look at this as an equilibrium equation and so if we look at our equilibrium constant if we were to write it out how do we do that again it's simply going to be the concentration of our two products over the concentration of our reactant and so if we do a little bit of manipulation algebraically what we can do is isolate the concentration of those hydronium ions on the left side what is that remember if we take the negative log of that that's going to be our pH and we want to keep that as
stable as we can so if it's a good buffer solution how do we keep it as stable as we can well if we keep our ka value equal to our concentration of our hydronium ion or if we keep our pH equal to our PKA value that's going to create a good buffer if one of those is much larger than the other one changes in one will change the other also we want to look at equal concentrations of that weak acid and its conjugate base and so if we can keep those equal to one we can
have large changes in that tenfold changes in that will only change the pH value a total of 1 and so we want to keep those values of the weak acid and conjugate base equal to each other and also we can use our PKA values which is remember looking at the concentration of reactants and products and we can figure out what's going on in the reaction so if our pH value is less than our PKA that means we have more of this weak acid and if it's greater than our PKA that means that we have more
of the base and so if you think of it like this if pH goes down we've got more of the acid and if pH goes up then we've got more of the base over on this side and so what are some good applications of that well an acid-base indicator is a great example of that so if we're looking at Bron thymol blue so what color is that going to be if we're in a neutral solution it's going to be right at 7 so our pH and our pKa values are essentially equal to each other and
so what happens if our ph value goes down well that's going to shift it more towards the left and so we're going to have more of this form of growth imal blue which is going to give us that yellow color what happens if we go to the right that's going to give us more of this blue color in relation to that neutral and so we can see changes in the color of that indicator and what that's telling us is changes in the pH this is also important biologically remember proteins are made up of an amino
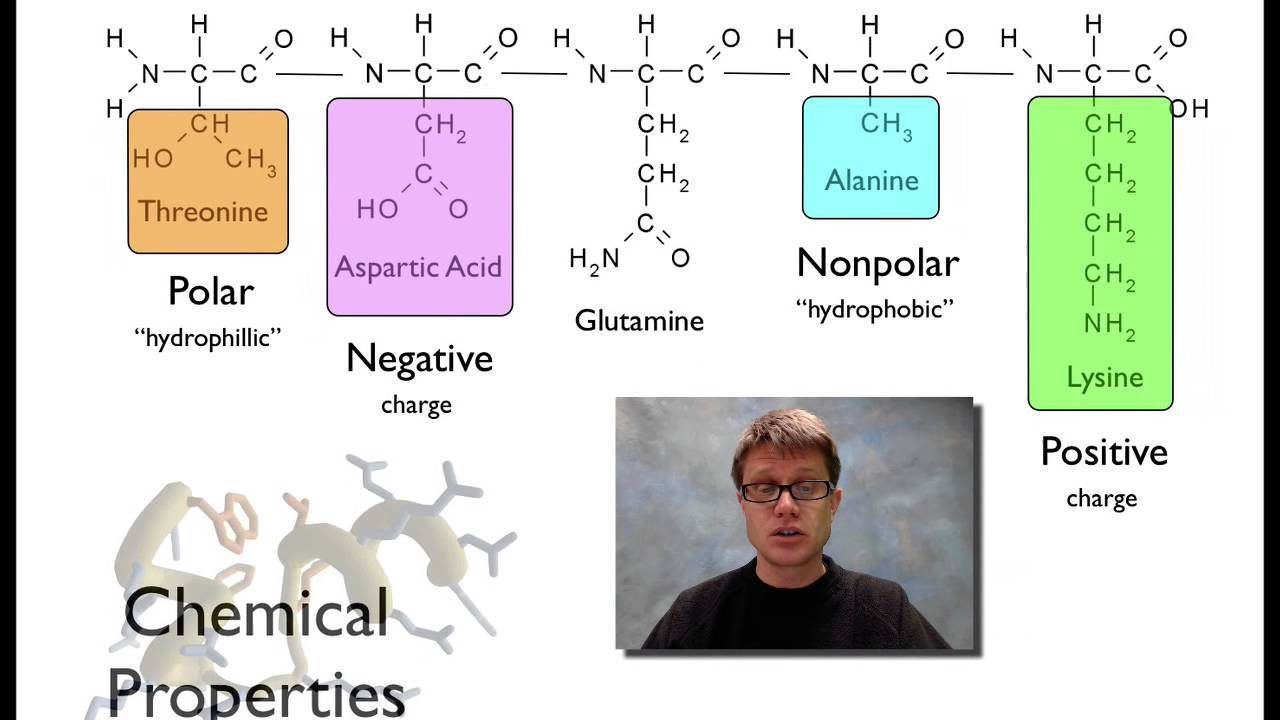
acid and each of those amino acids are going to have a different side chain which is going to be if we look at every one of these amino acids the top part is identical but each of the side chains that drop off the bottom is going to be different and so each of these have a different PKA value and so if we change the pH of the overall protein so this is the myoglobin for example it's going to change the behavior of each of those side chains in the amino acid inside it and so did
you learn that when we're creating a good buffer solution we want to keep our pH and PKA values equal to each other and did you learn that changes in the pH related to the pKa tells us if we're moving more towards the left more of the acid or more of the base and then could you design a good buffer solution remember what we've simply got is a weak acid on the left side and it's in its conjugate base on the right side I hope so and I hope that was helpful