types of chemical reactions i will teach you six types of chemical reactions with daily life examples the first type is combination reaction or addition reaction in combination reaction two or more substances combined together to form a single product like a plus b combined together to form a b product for example carbon plus oxygen gas would combine together to form carbon dioxide gas secondly ethene plus bromine gas would combine together to form ethene bromide thirdly sodium plus chlorine gas would combine together to form sodium chloride so two different substances combine together to form a single product
these types of reactions are called combination reactions are addition reactions the daily life example of combination reaction is photosynthesis we know that photosynthesis is a chemical reaction through which plants prepare their own food for instance carbon dioxide gas plus water would combine together to form glucose so remember that photosynthesis is a combination reaction secondly decomposition reaction in this type of reaction a chemical compound breakdown into simpler substances like a b would break down to a plus b for example consider water when we pass electric current through the water and electrolysis it is broken down to
hydrogen gas plus oxygen gas secondly hydrogen per oxide decomposes to water plus oxygen gas thirdly consider silver bromide we know that silver bromide is a light sensitive compound when light falls on silver bromide it decomposes to silver plus bromine gas remember that this type of reaction can occur spontaneously or they can be initiated by the heat the daily life example of decomposition reaction is when carbonic acid breaks down to produce water and carbon dioxide gas just remember that the breaking down of carbonic acid is a decomposition reaction thirdly displacement reaction when an element or iron
moves out of one compound and to another this type of reaction is called displacement reaction consider a b plus c here b is replaced by the c for example sodium plus water would react together to form sodium hydroxide plus hydrogen gas here sodium displaces the hydrogen ion secondly zinc plus hcl would react together to form zinc chloride plus hydrogen gas thirdly chlorine gas plus calcium iodide would react together to form calcium chloride plus iodine remember that these type of reactions only occur when the element moving to the compound is more reactive for example zinc is
more reactive than hydrogen so zinc is replacing hydrogen that's why we call it displacement reaction now coming to the daily life example the table salt we use in our daily life can be prepared by the displacement reaction for example calcium chloride plus sodium would react together to form sodium chloride plus calcium here sodium is more reactive calcium is less reactive so sodium is replacing the calcium fourthly combustion reaction when oxygen reacts with other compounds to produce light and energy this type of reaction is called combustion reaction remember that all combustion reactions are exothermic reactions there
are two types of combustion reactions complete combustion and incomplete combustion in complete combustion plentiful supply of oxygen gas is provided for example methane plus oxygen gas react together to form water plus carbon dioxide gas while in incomplete combustion limited supply of oxygen gas is provided for example in incomplete combustion methane plus oxygen gas would react together to form carbon monoxide gas plus water plus carbon in our daily life the car engine is run by the combustion for example the burning of petrol is an example of combustion reaction just remember that burning of petrol in the
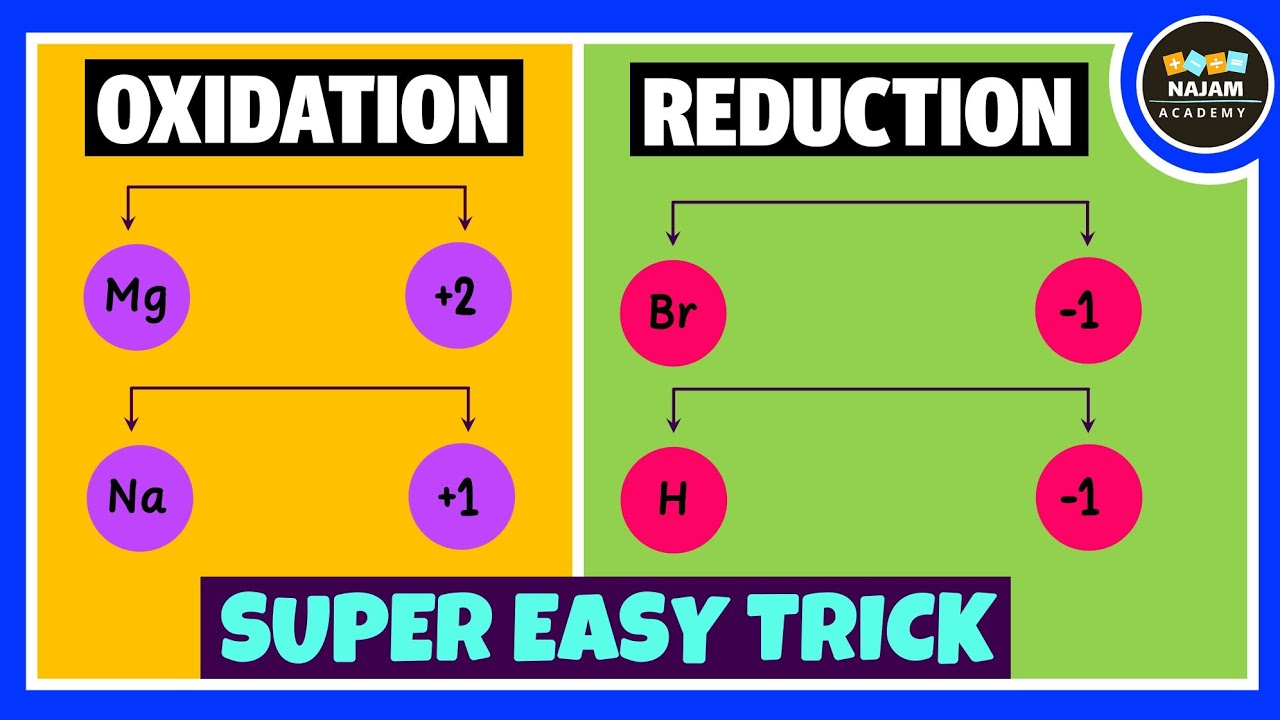
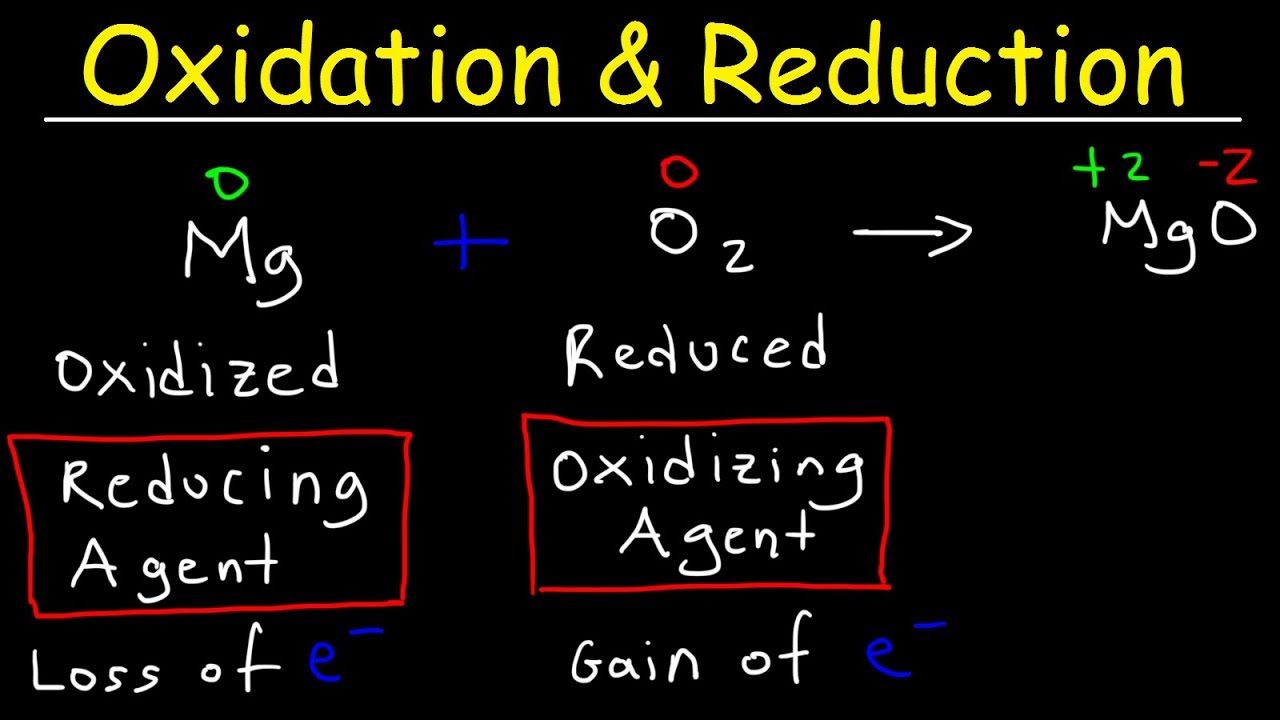
car engine is combustion reaction fifthly redox reaction redox reaction involves a transfer of electrons between two species consider this example here a lose electron to form positive ion b gains electron to form negative ion so we say that a is oxidized and b is reduced for example when magnesium plus oxygen gas react together it form magnesium oxide here magnesium is losing two electron so it is oxidized on the other hand oxygen is gaining two electron so it is reduced so oxidation and reduction takes place in this reaction that's why we call it redox chemical reaction
in our daily life the resting of iron is good example of redox chemical reaction finally neutralization reaction remember that neutralization reaction is also known as double displacement reaction or precipitation reaction a neutralization reaction when acid and base react together they form salt plus water for example hcl plus sodium hydroxide react together to form sodium chloride plus water here hcl is in acid sodium hydroxide is a base and sodium chloride is assault secondly hno3 plus potassium hydroxide react together to form potassium nitrate plus water now coming to the daily life examples the toothpaste we use in
our daily life is best in nature there are bacteria in our mouth which produce different assets when we brush our teeth with the toothpaste the best and the toothpaste neutralizes the acids produced by the bacteria in our mouth secondly due to overeating our stomach produces more acid which causes several problems to neutralize the acid in our stomach we use magnesium hydroxide which is best in nature that reduces acid and our stomach i hope that you have learned all about the different types of chemical reactions