An emulsion is a mixture of two or more immiscible liquids – one being oil based and the other water based, or “aqueous. ” They are described as “immiscible” liquids because they cannot easily be mixed together without separating. We can explain how to make an emulsion with a common example most people will be familiar with: Salad dressings.
We’ve all seen bottles like this – with the oil and water separated into two distinct parts, or “phases”. Now, you don’t want to pour that on your salad …so how do you mix it? All that energy you put into the liquids by shaking the bottle reduces the phases into smaller and smaller globules until they are completely dispersed into one another.
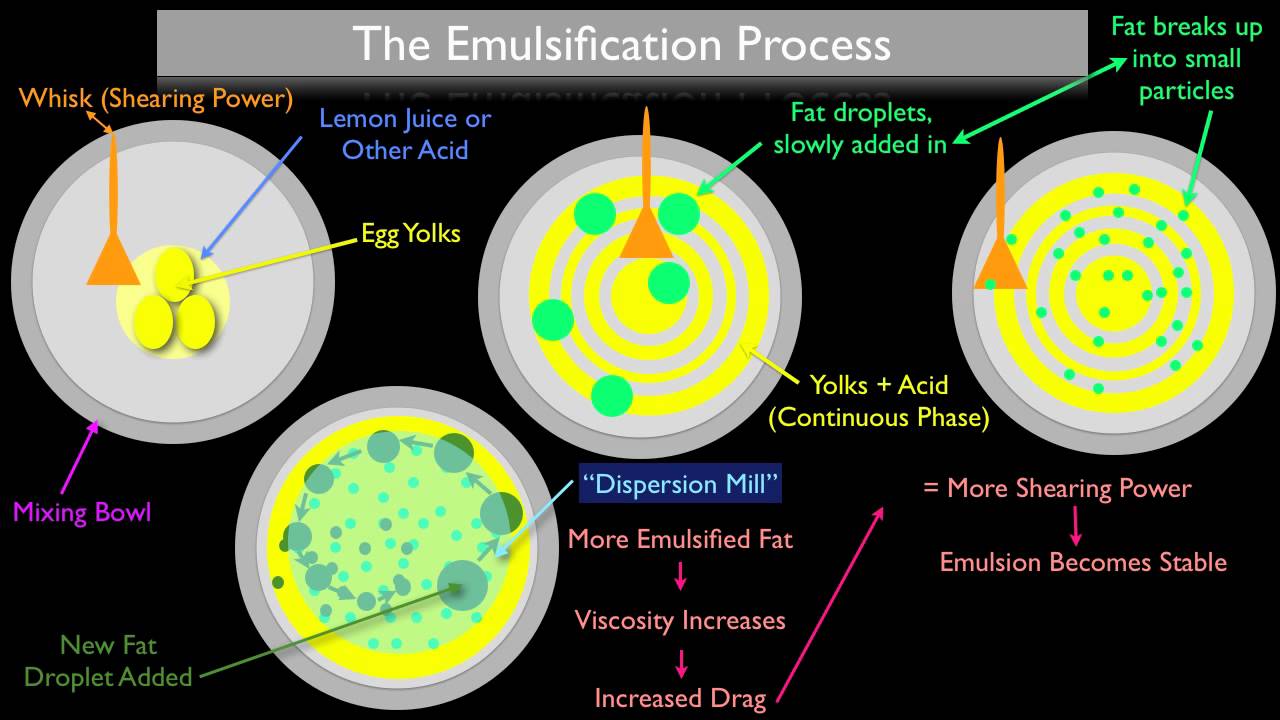
You now have an emulsion, but it’s not stable – and after a while the two phases will start to separate, so next time you reach for the bottle it’s like this again. To prevent that from happening and make a stable emulsion you need to add an emulsifier. Some examples of natural emulsifiers commonly used include egg yolk, mustard and soy lecithin, but there are many synthetic emulsifiers as well.
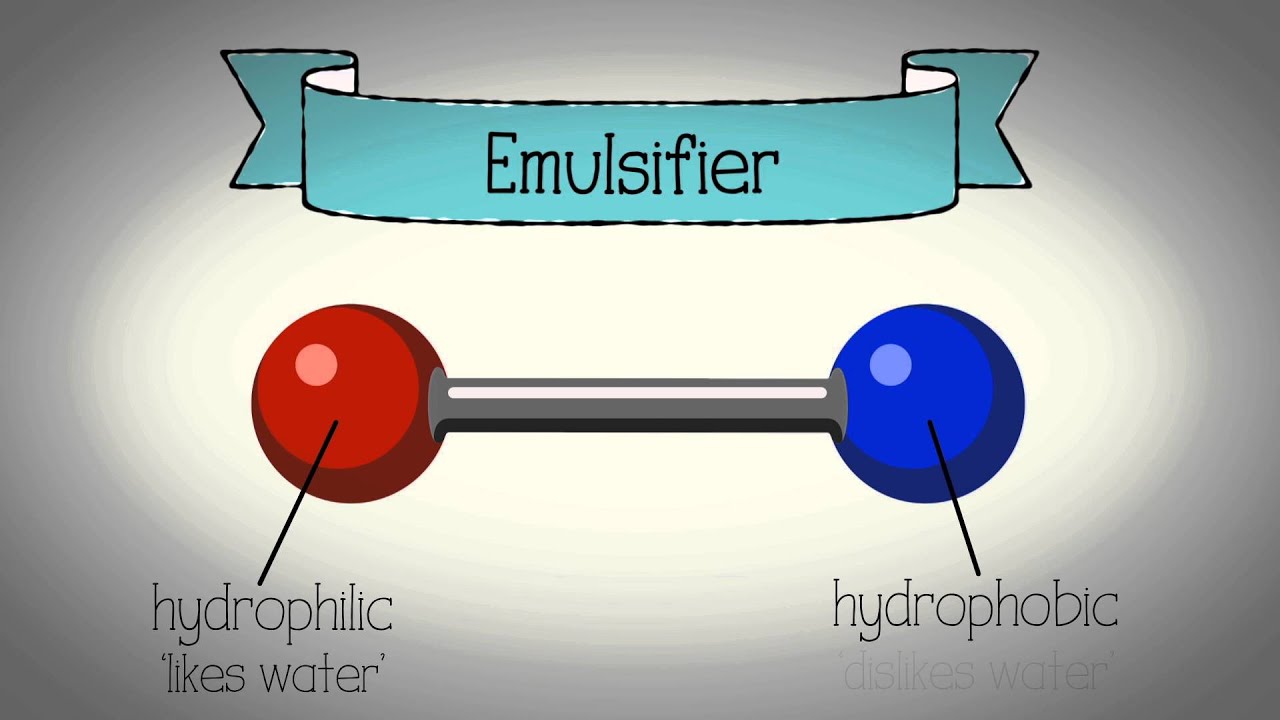
An emulsifier has a hydrophilic (water liking) end and a lipophilic (oil liking) end. The hydrophilic end is attracted to the aqueous phase and lipophilic end to the oil phase, and this binds them together. Emulsifiers also work by forming a film around the globules of one phase, this prevents them from joining back together or “coalescing” and thus keeps them suspended in the continuous phase.
Another way to stabilise your emulsion would be to use hydrocolloids like Xanthan gum or Guar gum. Although they are not technically emulsifiers they help achieve stability by increasing the viscosity of the mixture which keeps globules in suspension. The emulsifying agent is critical – some emulsifiers are more attractive to one phase than another, and this helps you achieve the type of emulsion you require.
Simple emulsions are either oil dispersed in an aqueous phase or water dispersed in oil. For example many sun creams and lotions - are water-in-oil emulsions containing an aqueous dispersion of sunscreen ingredients finely dispersed in oils and waxes. In this case we call the oil the continuous phase, and the water is the dispersed phase.
In contrast, mayonnaise is an example of an oil-in-water emulsion. The vegetable oil forms tiny droplets within the aqueous phase. In both cases, emulsifiers are needed to prevent the dispersed droplets from coalescing and breaking the emulsion.
So how do you manufacture a stable emulsion and stop your salad dressing or other emulsion from separating? Not like that. You need something that can impart a large amount of energy.
Like a rotor/stator mixer for example. It’s a high speed, high shear mixer. Unlike an conventional agitator which just moves the two liquid phases around, the high shear mixer sucks the liquids into the workhead and forces them out through the holes in the stator – in this case an emulsor screen – which reduces them to a fine globule size.
After a few minutes of intense high-energy mixing the two phases are finely and uniformly dispersed – and a stable emulsion is formed. But the way you introduce one liquid to another is also very important. You need to add the dispersed phase to the continuous phase at a controlled rate; and of course, if you’re making an oil in water emulsion you need to add the oil into the water …and the reverse for water in oil.
Successful manufacturing requires the right balance of ingredients, processing techniques and equipment to get it right. With a Silverson mixer you can take one of these factors out of the equation. It will rapidly produce a fine, uniform emulsion in no time, and batch-to-batch consistency is guaranteed, time after time.
Want to find out more? Visit our website Silverson.











![6 Ways to Separate an Oil and Water Emulsion [Oil & Gas Industry Basics]](https://img.youtube.com/vi/-xWQMWJD8hg/maxresdefault.jpg)