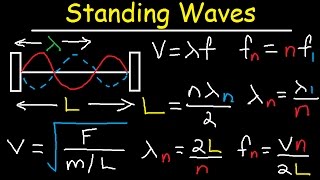
in this video we're going to talk about how to calculate the speed of sound in solids liquids and gases so let's start with this problem estimate the speed of sound in a solid such as steel which has an elastic modulus of 200 times 10 to the nine pascals so what formula do we need the speed of sound is the square root of young's modulus which is the same as the elastic modulus divided by the density of the solid so the elastic modulus of steel is 200 times 10 to the nine pascals or 200 gigapascals the density of steel is 8 000 kilograms per cubic meter so let's divide 200 times 10 to the 9 by 8 000 and then take the square root of that result so the density i mean the speed of sound in steel is about 5 000 meters per second and so it's pretty high now keep in mind this is just an estimate so the actual speed of sound is somewhere around 5000 meters per second it might be more or less but this is just an estimate number two estimate the speed of sound in the liquid such as water which has a bulk modulus of 2. 18 times 10 to the nine pascals or 2. 18 gigapascals so here's the formula that you need so the speed of sound in the liquid is going to be the bulk modulus divided by the density and so we have the bulk modulus and the density of water is one thousand kilograms per cubic meter and so this is going to be about 1476 meters per second so that's an estimate so that's the speed of sound in water number three calculate the speed of sound in air at 20 degrees celsius the ratio of heat capacity is 1.
4 air consists of 78 nitrogen 21 oxygen 1 and we're given the molar mass of these gases what is the speed of sound at 25 as well and there's supposed to be a question mark here but let's focus on the first temperature at 20. so the formula that we need is this equation it's equal to gamma times r which is the gas constant times the kelvin temperature divided by the average molal mass of air so let's calculate this value first we have gamma the ratio of heat capacities that's 1. 4 and r is another constant that's 8.
3145 joules per mole per kelvin so the temperature has to be in kelvin so to convert celsius to kelvin add 273 to it so 20 plus 273 is 293 kelvin the last thing we need is the average molar mass so the molar mass of nitrogen gas if you look at the periodic table it's going to be 14. 01 times 2 or 28. 02 and air is 79 i mean 78 nitrogen so we need to multiply that by 0.
78 to calculate the average molar mass of air oxygen has a molar mass of 32 and air is about 21 oxygen an argon is or has a molar mass of 40 and air consists approximately about one percent of argon so let's put these numbers together in the calculator let's see what we get so the average molar mass is about 29 grams per mole now we need this answer in kilograms per mole so let's convert grams to kilograms one kilogram is equal to a thousand grams so we need to divide this number by a thousand so this is going to be 0. 029 kilograms per mole so now we have everything that we need to plug into this formula so it's going to be 1. 4 times 8.
3145 multiplied by a kelvin temperature of 293 divided by the molar mass which is in kilograms per mole so this works out to be 343 meters per second so that's the speed of sound in air at 20 degrees celsius now let's find out what it is at 25 degrees celsius so first we got to find the kelvin temperature so it's 25 plus 273 so it's a little bit higher it's going to be 298 kelvin so everything is going to be the same except the new temperature so let's go ahead and plug this in so this is going to be 346 meters per second so as we could see as the temperature goes up the speed of sound increases in air now there's another simple equation that relates temperature and the speed of sound and here it is the speed of sound in air is approximately 331 meters per second plus 0. 6 times t where t is in celsius so at 20 degrees celsius it's approximately 331 plus 0. 6 times 20.
and so this will give you 343 meters per second and let's say if we wish to calculate the speed of sound at 25 degrees celsius it's going to be 331 plus 0.