[Music] hello and welcome back to immunology the war is over episode five where we are going to focus on all things antibodies now antibodies b cells and plasma cells are intertwined you cannot tell the story of one without acknowledging the other which is actually very beautiful b cells become plasma cells which generate antibodies and antibodies are potent weapons in our immune system but because there's so much juicy stuff to cover we'll be doing this over two episodes in this episode we're gonna focus on antibodies and the b cell receptor and in the next episode we'll
be discovering exactly what happens when a b cell comes across its matching antigen including how t cells help in the process i am so excited to get into this episode so grab a cuppa and let's jump right in so antibodies also known as immunoglobulins these are by far one of the coolest weapons in our immune system but their powers lie in their ability to work collaboratively they do not work alone so if you took a pathogen and some antibodies directed against that pathogen and you combined these in a test tube nothing would happen the antibody
would bind to the pathogen for sure but on its own it's not going to destroy that pathogen however if you took this same pathogen and placed it into a human being who had antibodies against that pathogen it's a whole new ballgame because the antibodies are now surrounded and supported by the rest of the immune system and this is where the magic happens so the antibody binds to the pathogen and encourages the other parts of the immune system such as complement and immune cells to come along and destroy that pathogen so antibodies are like markers which
point out targets for destruction now we will be coming back to complement and the innate immune cells in upcoming episodes but for now we're going to just touch on what happens when these parts of our immune system come into contact with an antibody stuck to a pathogen let's start with complement complement is an incredibly noxious part of our immune system designed to annihilate pathogens when complement proteins become activated it sparks off a cascade at the end of the complement cascade is the membrane attack complex which punches holes in the microbe causing it to die but
along the way complement also generates inflammatory molecules some of these function as chemoattractants which attract immune cells into the area others are opsinins which bind to the surface of the microbe and encourage phagocytosis of that microbe there are three different pathways which spark complement one of which is known as the classical pathway and this is the one that is initiated by antibodies such as igm and igg so antibodies bound to a pathogen can spark complement in order to destroy that pathogen in an absolute jiffy and at the same time that they spark off complement antibodies
also encourage immune cells to join in the battle and they can do this in two ways igg acts as an opsinin meaning that when it is bound to a microbe it will encourage phagocytosis of that microbe and the other way in which antibodies can encourage immune cells to kill microbes is something known as antibody dependent cellular cytotoxicity or adcc this is when immune cells such as neutrophils eosinophils or natural killer cells spill their toxic granules all over that organism and whilst this property of antibodies was designed by mother nature to fight infection it's something that
we bank on when developing monoclonal antibodies to treat cancer if you think about it just infusing an antibody that binds to cancer cells is not going to shrink that tumor the reason that these drugs work is because the antibodies bound to the tumor cells activate natural killer cells which are capable of destroying those cells and the final thing that antibodies can do is neutralize pathogens so say a pathogen requires a particular surface molecule in order to gain entry into cells if an antibody happens to bind to that surface molecule it can impair its biological function
rendering the organism less invasive overall similarly antibodies can bind to bacterial toxins reducing their harmful effects now you can imagine that in order for an antibody to have these effects it has to be able to bind to the microbe and so naturally extracellular organisms like bacteria come to mind but antibodies also have a role in combating intracellular organisms such as viruses and we see this in our clinical practice when we check for immunity against a virus we'll tend to check the serum antibodies against that virus and this is because viruses eventually do burst out of
cells and into our bloodstream and in our bloodstream antibodies are circulating so when it comes to viruses t cells are going to be the mainstay for killing virus infected cells but viruses and the circulation is very much antibody territory so like i say antibodies are one of the coolest weapons in our immune system they can neutralize toxins and viruses activate complement promote phagocytosis and promote antibody dependent cellular cytotoxicity antibodies are very potent indeed so that's what antibodies do and their ability to do this is built into their design and so over the next few minutes
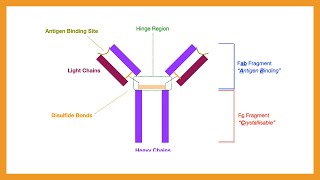
we're gonna see exactly what antibodies are made of antibodies have two heavy chains and two light chains joined together like so each heavy chain and light chain has a constant region and a variable region the variable region of both the heavy chain and light chain come together to form a unique binding site f a b on the other end of the antibody we have the constant region or the fc region which annoyingly doesn't stand for constant it stands for crystallizable fragment anyway this is the part of the antibody which interacts with other parts of the
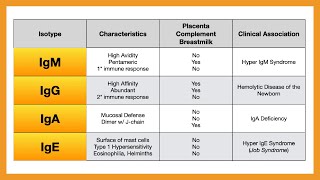
immune system now when it comes to heavy chains and light chains there are a few to choose from but one thing that they each have in common is their entanglement with the greek alphabet there are five different types of heavy chain gamma alpha epsilon me and delta which corresponds to igg iga ige igm and igd the five different classes of antibodies and there are also two different types of light chain kappa and lambda now you might be thinking this is a lot of detail why on earth would i want to know how these antibodies are
structured but there is always method to my educational madness and i promise you that if you understand the structure of these antibodies right now it makes understanding heaps of other things like antibody class switching and absolute doddle but it also has huge clinical relevance in conditions such as myeloma so what i would like you to lock in right now is this the class of an antibody is determined by the type of heavy chain that it has not the light chains not the shape of the antibody not the size of the antibody none of that the
class of the antibody is determined by the type of heavy chain that it has so lock that in for me and as we move through this episode you'll see why that was important okay so in any one antibody we have two heavy chains and two light chains and there will only be one type of heavy chain and one type of light chain meaning you'll either have kappa or lambda never the two together now as you can see there are five classes of antibody which are all designed to work slightly differently as we journey through this
episode we're going to come back to this table again and again as we learn about the various classes of antibodies and what they do but i thought we could start with perhaps some less familiar territory we saw earlier that antibodies have the ability to neutralize toxins and microbes and engage other parts of our immune system to destroy invading pathogens but what if i told you that antibodies have yet another function as b cell receptors get this igm and igd antibodies function as b cell receptors so the first place that you will ever see an antibody
in your existence as a human being is on the surface of your b cells let's unpack that now b cells originate in the bone marrow and here they will undergo the early phases of development maturation and testing before being released and as part of this maturation process the b cell receptor is installed and this b cell receptor takes the form of an antibody now initially b cells can only generate the me heavy chain so the first ever antibody on the surface of a b cell is an igm antibody now this is the basic subunit of
igm there is another version of igm which is composed of five of these subunits and this big pentamer igm with the 10 binding sites is the one that we release into our circulation in response to infection so both of these are still igm antibodies because they both have the me heavy chain but their roles are slightly different so this little igm sticks out of the b cell and acts as the b cell receptor and this b cell receptor is unique it has a unique binding site which will recognize a specific antigen and if you watched
episode one of this series this is gonna sound very familiar we saw that every t cell has a unique t-cell receptor which binds to a specific antigen and this means that we can recognize hundreds of thousands of different antigens throughout our lifetime so just like the t cells the b cell receptor is unique which maximizes the amount of antigens that we can respond to over time and just like we saw with the t-cells these unique binding sites are created by gene shuffling so we take a set of genes shuffle them around line them up in
a row and then transcribe them to make hundreds of thousands of unique binding sites and because this process is entirely random the b-cell receptor just like the t-cell receptor needs to be tested we need to make sure that this randomly generated b-cell receptor is not going to react to self-antigens and cause autoimmune disease so before the b cell leaves the bone marrow the b cell receptor needs to be deemed functional sometimes if a b cell receptor is not quite right then the b cell gets an opportunity to go back and edit that receptor to make
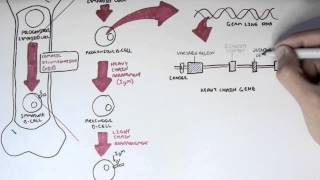
it more functional but ultimately any b-cell which doesn't make the cut will not be permitted to leave the bone marrow and this process i mentioned of shuffling genes to create thousands of unique b-cell receptors and indeed t-cell receptors is known as vdj recombination this is fundamental to creating antibody diversity and so we're going to unpack that now okay so antibody diversity is created by shuffling genes and these genes are the vdjc genes which is why we call it vdj recombination so to illustrate this i'd like you to imagine there are four boxes of genes each
box is labeled v d j and c variable diversity joining and constant now there are a number of genes in each of these boxes the constant genes as you might expect come as standard they're all the same but the vdj boxes contain a variety of genes it's more of a colourful lucky dip situation so in order to construct our antibody to make our b cell receptor we're going to take genes from each of these boxes line them up and then make proteins from them and this is what's happening inside b cells immature b cells start
off with our basic dna sequence that is shared by every single cell in our body but unlike other cells b cells are going to change the sequence of their dna in order to create the b cell receptor we said before that every antibody is composed of heavy chains and light chains and the genes which code for these are located on different chromosomes heavy chains are coded for on chromosome 14 kappa light chains on chromosome 2 and lambda light chains on chromosome 22 and remember that the light chains on any one antibody will be either kappa
or lambda never the two together so genes are taken from either chromosome 2 or 22 not from both heavy chains use vdjc genes so genes from every single box whilst light chains only use genes from the vj and c boxes for some reason they completely miss out the d so in our vdj boxes there are a variety of genes to choose from in each category we want to take a gene from each box line up those genes and then seamlessly transcribe them into a unique protein but when we first take the genes out of the
box they come with some extra genetic material attached and this will need to be trimmed away and b cells have a specialized set of enzymes to do this rag 1 and rag2 rag stands for recombination activating gene and you know how much i love love love love when someone names an enzyme after what the enzyme actually does it's so beautiful so these rag enzymes break and join dna they take the v dj and c genes line them up and chop out any extra genetic material in between them and the end result of this is the
creation of unique dna segments which code for both heavy chains and light chains so this process happens separately for heavy chains and light chains and then the two unique structures are combined to form an antibody which will be placed on the cell surface as the b cell receptor now of course sometimes amongst the chaos of making a random heavy chain and a random light chain you might end up in the situation where these two structures don't come together very well if that happens the b cell can go back to the drawing board and try to
fit a different light chain onto this heavy chain until it finds one that sticks so say a lambda light chain from chromosome 22 didn't work the b cell might try again with a kappa light chain from chromosome 2. but once a functional antibody is generated the dna is finalized there will be no more gene shuffling no more sussing out the options the b cell is now committed to making that one antibody with that particular binding site forever and this process is known as allelic exclusion okay so the first antibody that the b cell will ever
make is an igm antibody but once this igm is deemed functional the b cells will start to express igd antibodies as well this is a sign of b cell maturity b cells which express both igm and igd on their surface are permitted to leave the bone marrow and go in search of their matching antigens both igm and igd on the surface of a b cell have the same unique antigen binding site and they both function as b cell receptors the only difference between them is they're heavy chains igm has the me heavy chain and igd
has the delta heavy chain but how can this be we just saw that those vdjc genes we shuffled in order to create the unique antibody are now locked in place we can't do any more shuffling so how enough do you start making the igd when the dna remains the same and the answer lies in the primary mrna transcript although the dna sequence within mature b cells is very much locked in the rna transcript can be changed or spliced in order to create a different heavy chain so from the same dna code this b cell will
make both igd and igm and place both of these on the cell surface now it's not entirely clear why we need both of these on the surface of b cells presumably they have different signaling pathways when they come across their matching antigen but the truth is we don't really know okay so igm and igd are the first antibodies that we ever make and these function as b cell receptors designed to seek out their matching antigen so the b cell can become activated proliferate and turn into plasma cells which release high affinity antibodies into our circulation
and we'll be covering all of this in episode six but before we get there we still have to fill in this table and get to know all of the other antibody classes let's do that now first up igd so igg functions as a b cell receptor it can also be found in our circulation at low levels and likely plays some role in the response to infection but overall its main job by far is as the b-cell receptor igm as we saw can also function as a b-cell receptor but there is also a bigger pentimer version
of igm as well this pentameter igm is the very first antibody we make in response to infection and that's because b cells already know how to make the igm they're basically born with the ability to make igm and later with the help of t cells they'll be able to make the other antibody classes but initially igm is their absolute go-to and this big igm has 10 binding sites which sounds impressive but it's actually necessary these antibodies are binding to 3d structures on the surface of microbes so an antibody may bind to a pathogen but it's
not going to be glued to that pathogen and so antibodies are prone to falling off to get around this igm has 10 binding sites so that it has more of a chance of latching onto the target later b cells will be able to generate higher affinity antibodies which bind more tightly to the specific antigen and require less binding sites for example igg so for any one antigen we can think of the igm as being sort of a rough draft but later the b cells will switch to making igg which has a far more refined deliberate
antigen binding site which will bind much more tightly to that antigen but until igg is generated igm will be very much on the case and it does this by activating complement igm is less good at directly activating immune cells you can imagine that because of the way the fc portions are arranged that immune cells would find it hard to place themselves in a way that they could interact with these but that doesn't matter the ring of fc regions easily sparks off complement and then complement brings in the immune cells anyway so igm doesn't have to
igm is the antibody that is first on the scene but once t cells get involved b cells will make other antibody classes the most common of which is igg as we mentioned igg has two binding sites and binds to antigens with high affinity on the other end the fc portion of the antibody can interact with immune cells and complement and a really cool thing about igg is that this is the only antibody class that crosses the placenta which is handy because we don't actually make any of our own igg until later when we come into
contact with pathogens so having some of mom's igg good to go offer some immediate protection until we can make our own and there are four subclasses of igg igg 1 2 3 and 4 numbered in order of their abundance and these subclasses have subtle differences in their hinge regions igg 1 and 3 have long hinge regions which make them more flexible and able to bind to complement and immune cells more easily on the other hand igg2 binds complement less well and igg4 doesn't fix complement at all and these varied properties of igg subclasses are used
in clinical practice by drug developers when it comes to making monoclonal antibodies we tend to use igg these have the longest half-life and so they maximize the length of time in which the drug can take effect but it's not just the half-life that's important drug developers also select the right igg subclass for the task at hand if the goal of a monoclonal antibody is to just block a target molecule and impair its function you don't need to activate complement or immune cells in order to do that and so you would choose igg4 to make your
monoclonal but on the other hand if we're making a drug for use in oncology and you want the drug to bind to a cancer cell and then you want the immune system to see that antibody and destroy the cancer cell you're going to want to use something like igg-1 which would do a beautiful job of invoking adcc by natural killer cells interestingly although it's the most potent we don't tend to use igg3 to make monoclonal antibodies apparently it's quite difficult to make these but also this antibody is prone to degradation and has the shortest half-life
of all of the igg antibodies okay so that's igg the most abundant antibody in our circulation the next antibody to touch on is iga this is the second most common antibody in our circulation but the most common antibody found in human secretions we're talking saliva tears mucus breast milk you name it you'll find some iga antibodies in there and so iga is the first point of defense in areas exposed to the outside world such as our respiratory and gastrointestinal tracts iga has two subclasses iga1 and iga2 iga1 has a flexible hinge region and is vulnerable
to cleavage by pathogens you'll see this iga mainly in our bloodstream as a monomer iga 2 on the other hand is a more durable rigid structure this is the one we see in our secretions and it tends to exist as a dimer with two antibodies joined together and central to this joining of the two antibodies is the secretory component this sounds fancy but it's basically just a molecule made of sugar and protein which joins the two iga molecules together but also provides added strength to the molecule preventing its breakdown and helps the iga to anchor
to epithelial linings and iga is pretty handy it's able to neutralize toxins and trap organisms so iga might even stop an infection before it gets started when iga binds to a microbe it may do this using its dedicated binding sites just like the other antibodies do but iga also has another trek up its sleeve these antibodies are generously coated with glycans which can adhere to and trap pathogens iga antibodies bound to a pathogen can activate antibody dependent cytotoxicity by neutrophils but they don't tend to activate complement so that's iga and lastly we arrive at ige
antibodies these are the least common of the antibody classes but they are very potent and definitely worth a mention ige tends to join forces with mast cells so when ige is released into the circulation it will travel around and find a mast cell to hang out with it will embed itself into the mast cell and the mast cell loves this because it now has an antenna that it can use to look for antigens so if an antigen comes along and binds to one of these iges on the mast cell the master will register the signal
and degranulate its toxic contents all over that antigen now this system was designed by mother nature to help us fight parasites but of course the most common place we encounter ige in mast cells is within allergic conditions such as asthma so that's the five antibody classes in a nutshell and whilst i've discussed each of these antibodies in a singular fashion the truth is that antibodies are much more potent when they come together in immune complexes if you think about it a single antibody bound to a pathogen could easily be missed by complement or immune cells
flowing by but a big complex of antibodies bound to an antigen is going to attract much more attention so that's a wrap for this episode thank you so much for joining me i hope you enjoyed learning about antibodies and the bc receptor i hope you'll join me for the next episode when we'll be exploring what happens when a b-cell comes across its matching antigen i'll see you there bye [Music]