let's take a closer look at atoms atoms are the building blocks that make up everything all stuff everywhere here I've got some drawings this is table salt a solid here's water a liquid and finally oxygen gas and what we're looking at here is what we'd see if we could take these three things and zoom in zillions and zillions and zillions of times and if we could look at these closely enough we'd be able to see the atoms these little building blocks that make everything up now atoms aren't really little colored circles but it's a good
way to think about them and it's the way we'll usually draw them when we're learning about this stuff atoms are super tiny they are so tiny it can be really hard to wrap your head around how tiny they are here is something that might help you think about this take a piece of paper look at how wide it is here all right this is so incredibly thin now atoms are so small that it would take one million atoms lined up next to each other in a row to be as thick as this tiny width of
paper that's how small atoms are now what I want to do now is look even closer at an at let's take one of these atoms here and zoom in even more so we can see what's inside of it here's a magnified view of one atom it's not a perfect representation of exactly what an atom would look like but it's close enough for right now and check this out the atom itself is made up of even smaller things right so that we said adoms are like the building block for all of stuff but then if you
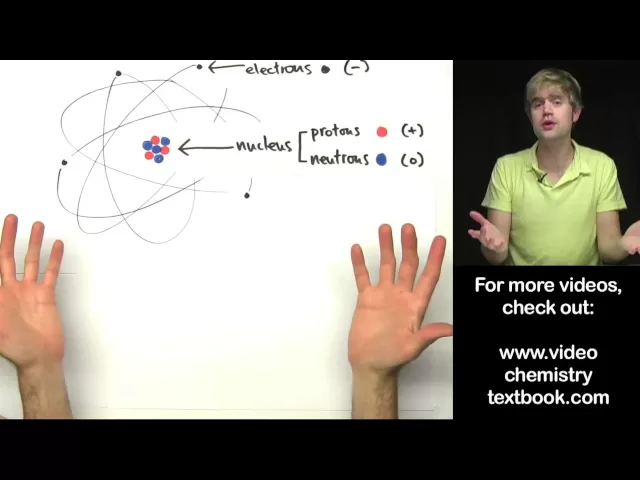
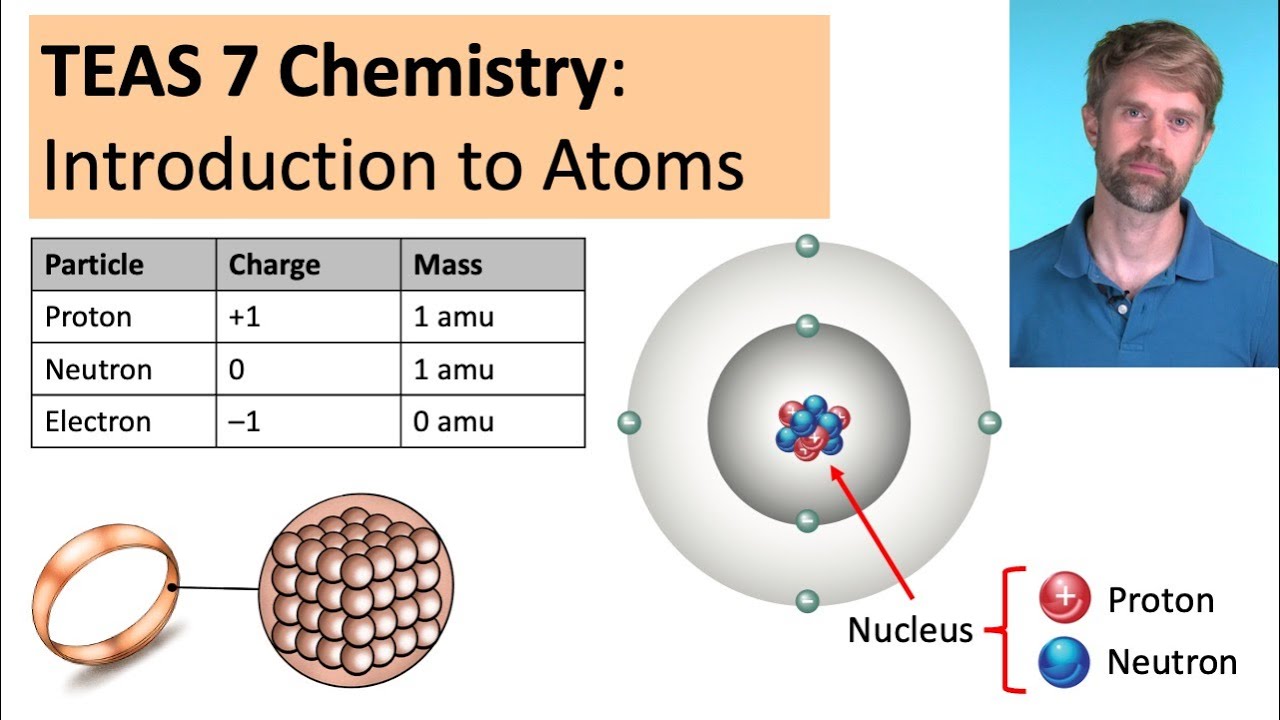
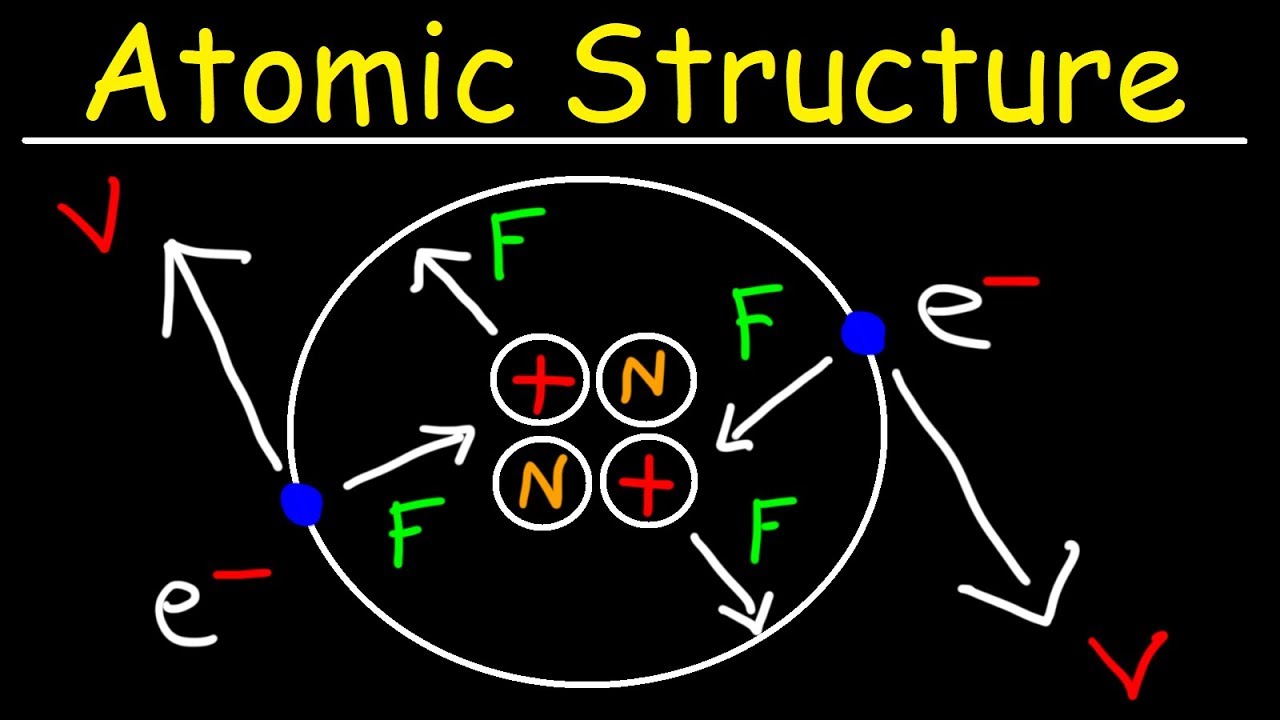
look at an atom it has even smaller building blocks that make it up so in the center of the atom is something called the nucleus and the nucleus is made up of two tiny particles there are protons which I've drawn as red circles here and then they're neutrons which I've drawn as blue circles nucleus is here in the middle of the atom and then on the outside of the atom there are these uh little black circles that I'm using to uh to represent electrons the electrons you'll see are connected to these sort of oval paths
and I've drawn these in to show that the electrons are constantly moving they're flying around the outside of the atom at an incredibly high speed whereas the nucleus stays solid here right in the middle of the atom now electric charge is very important when we're talking about atoms for number number of reasons protons have a positive charge a proton has a charge of 1 plus electrons on the other hand have a negative charge one electron has a charge of one minus now neutrons they don't have an electric charge at all they have a charge of
zero and neutral is a word that we use to refer to something that doesn't have a charge so neutrons sounds a lot like neutral now positive and negative charges attract remember that opposite charge is attract and that has important consequences for how this atom works we said that these electrons are flying around the outside of the atom they're moving really fast so why don't they just fly out into space well the reason why is because the negatively charged electrons are attracted to the positively charged protons so the protons kind of pull the electrons in they
keep them from just flying away but the electrons are moving around so fast that they're not able to actually pull in here and actually touch the protons so that's why they keep staying on the outside and don't just crash in here and all touch the protons now sometimes people ask well neutrons neutrons don't have a charge why are they important at all well it seems that neutrons help keep all of the particles in the nucleus strongly connected to each other now electrical charge is important something else that's important animportant with atoms is what their mass
is how much they weigh so in order to figure that out we have to look at the mass of the various things that make up the atom now a proton and a neutron are very very similar in size and in mass and they both weigh about this many grams look at what a tiny number this is right there's a decimal place all the way over here this is a number that it's best to represent in scientific notation instead but however you write this it's a really tiny unwieldy number so scientists came up with another way
to uh to measure the mass of a proton or Neutron and that's using a unit called the AMU the atomic mass unit and they said okay one proton or one Neutron weighs about 1 AMU that's a lot easier to use than this measurement here so Proton or Neutron weighs about 1 Amu an electron on the other hand is much much much smaller than a Proton or Neutron it weighs only 0549 AMU so a tiny fraction of how much a Proton or Neutron weighs for this reason when we're talking about the mass of atoms which we'll
talk about later on we usually add up the protons and neutrons to find out how many amus um the atom weighs and then we usually don't even worry about the El r at all because they're just they're so tiny they don't really have much of an influence on how much an atom weighs it's like when you get on a scale to weigh yourself you don't worry about the ring you're wearing or whether you're wearing a necklace or something because those things are just so tiny they weigh so little compared to how much you weigh that
it doesn't really influence your weight so protons and neutrons 1 AMU and electrons a tiny fraction of that now I said the atom the way it's drawn here isn't a perfect representation of what atoms would actually look like we'll talk more about that later but there are two things that I want to bring up for right now the first is the electrons and how they move this drawing might make you think that the electrons are spinning around in nice circles on the outside of the atom that's actually not what happens they're buzzing around much more
randomly kind of like hyperactive flies all over the place we'll talk more about that later the second thing has to do with the size of the nucleus and how far the electrons are from it If This Were a real atom blown up many many times and the nucleus were the size of a grape the electrons out here would be almost a mile away have this grap siiz nucleus and a mile away would be the electron spinning on the outside so that means that most of an atom is actually empty space anyway now that we've talked
about what an atom looks like and the particles that make it up we can go and discuss some of the characteristics that we can use to describe atoms these are things like like atomic number mass number and net charge