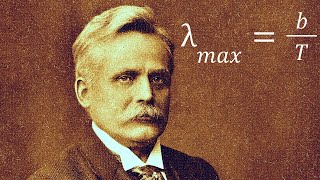after the discovery of the quantization of energy by Max plunk in 1900 scientists began to question their previously established laws of the universe based on classical mechanics and began to look towards the atom in search of answers Neils bore was a revolutionary in this branch of science proposing in Manchester England in 1913 that electrons orbit the atomic nucleus at quantized energy levels absorbing and emitting energy in discret amounts while shifting between levels in the process this new theoretical idea was unconventional at the time and in need of experimental confirmation it wouldn't take long before bour
got his confirmation from two collaborating German physicists James Frank and Gustav Herz who ironically had no idea of B's proposed model until after they published their results both James Frank and Gustav Herz were born in Hamburg Germany in 1882 2 and 1887 respectively to receive their doctorates both men traveled around universities in Germany Frank spent a year at the University of heidleberg and then finished his Doctorate at the University of Berlin in 1906 Herz following in the footsteps of his famous Uncle Hinrich Herz bounced around from the University of gingan to the University of Munich
and finally the University of Berlin where he completed his Doctorate in 1911 that that same year Frank was appointed a physics lecturer at the very same institution Herz followed suit in 1913 being appointed physics assistant at Berlin and the two collaborated that same year having known about plunk proposal the two wanted to experimentally prove quantized energy emission and absorption and so they set up an experiment to measure how current changes with increasing voltage in an electron chamber the chamber they set up was was rather small no more than a few inches across a cathode and
an anode are placed in the chamber with an electric potential difference given between the two so that electrons could travel to the anode and a current can be measured the only thing keeping this tube from being a vacuum chamber is a small droplet of mercury that has been vaporized due to the chamber being heated to 115° C when electrons are fired from the cathode to the anode they collide elastically with the Mercury atoms perfectly bouncing off of them without transferring any energy however when a certain voltage threshold is reached the electrons have enough energy to
collide inelastically with the Mercury atoms transferring some of their energy to the atoms this complicates the experiment because although some electrons have lost energy due to collisions all electrons would still reach the anode and it becomes difficult to measure the energy loss through just the cathode and anode alone so for this experiment a wire mesh was placed in between the two electrodes and was given a positive voltage greater than that of the anode that way the electrons that did collide with the Mercury atoms wouldn't have enough kinetic energy to reach the anode and would be
collected by the wire mesh only electrons that didn't suffer a collision would have sufficient energy to fly past the wire mesh and reach the anode from this setup they plotted the anode current against the voltage applied to the wire mesh and these were their results what they found was that as voltage would increase current would also increase but at distinct increments of 4.9 volts the current on the anode would drastically drop and then after those increments were passed the current would once again continue to increase this led Franken Herz to conclude that the electrons were
experiencing inelastic collisions with Mercury atoms transferring 4.9 electron volts of energy each time using a previously established Quantum relationship between the energy of excitation in the corresponding wavelength of light they were able to equate 4.9 electron volts to a wavelength of 254 nanometers in their second paper regarding this experimental setup Franken Herz released the wavelength of emission from their electron tube which happened to be exactly 254 nanm confirm confirming that mercury atoms were absorbing discrete amounts of energy from the electrons and releasing it in the form of photons with the exact same wavelength each time
although Frank and Herz didn't know about B's proposed Quantum model of the atom their experiment verified the atoms Quantum nature and the three of them received International recognition for their efforts Frank and Herz continued their work on this experiment until the outbreak of the first world war later that year Herz was severely wounded in 1915 but both survived the war and returned to physics after it ended in 1925 the two jointly shared the Nobel Prize in physics for their discovery of the laws governing the impact of an electron upon the atom the two had lucrative
careers after their Nobel Prize Frank ended up collaborating with Max Bourne and pressing the field of quantum physics forward while also mentoring and collaborating with future physics pioneers such as Patrick blackett and J Robert Oppenheimer Herz became an administrator at a Technical University in Berlin in charge of rebuilding its physics department and later in 1932 in his research he developed a method for separating isotopes of neon both physicists made Monumental contributions to the field of physics and their breakthrough experiment in 194 experimentally proved many quantum physics Pioneers correct helping drive a new and unpredictable path
forward through the Golden Age of modern physics if you enjoyed this video please consider liking and subscribing click here if you want to see more scientific progress made during this time period thank you for watching and I will see you in the next video