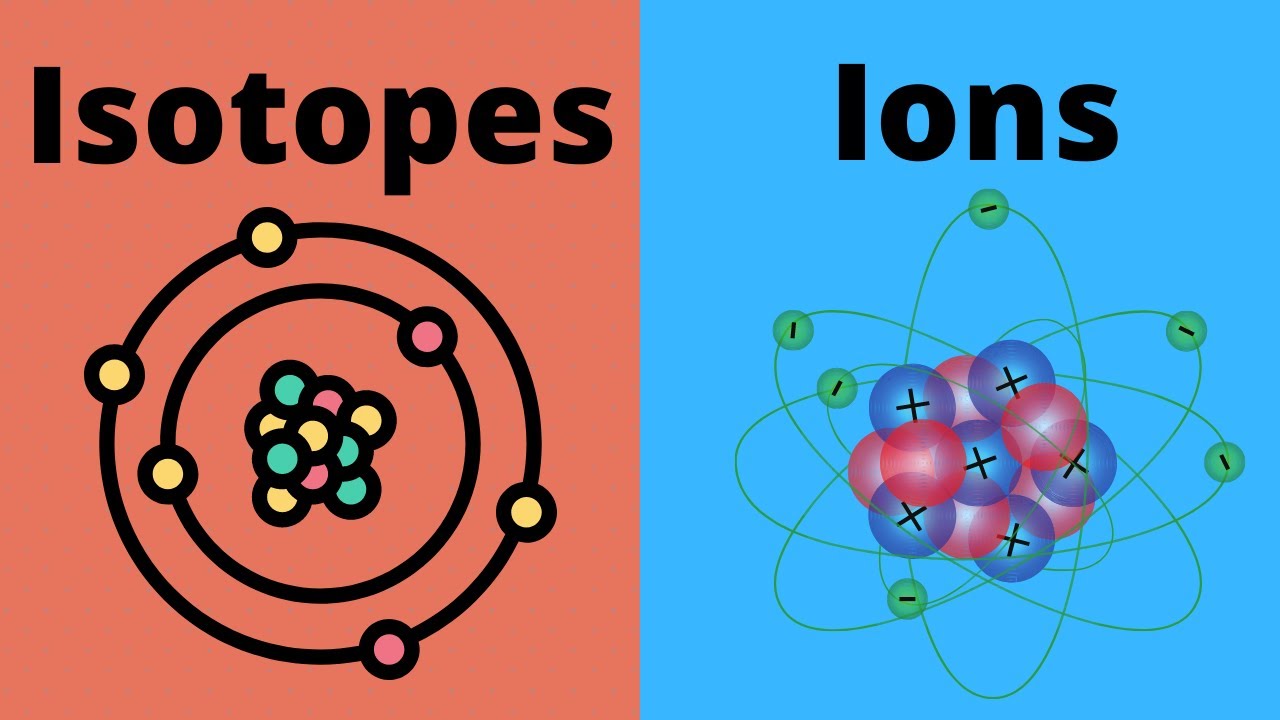
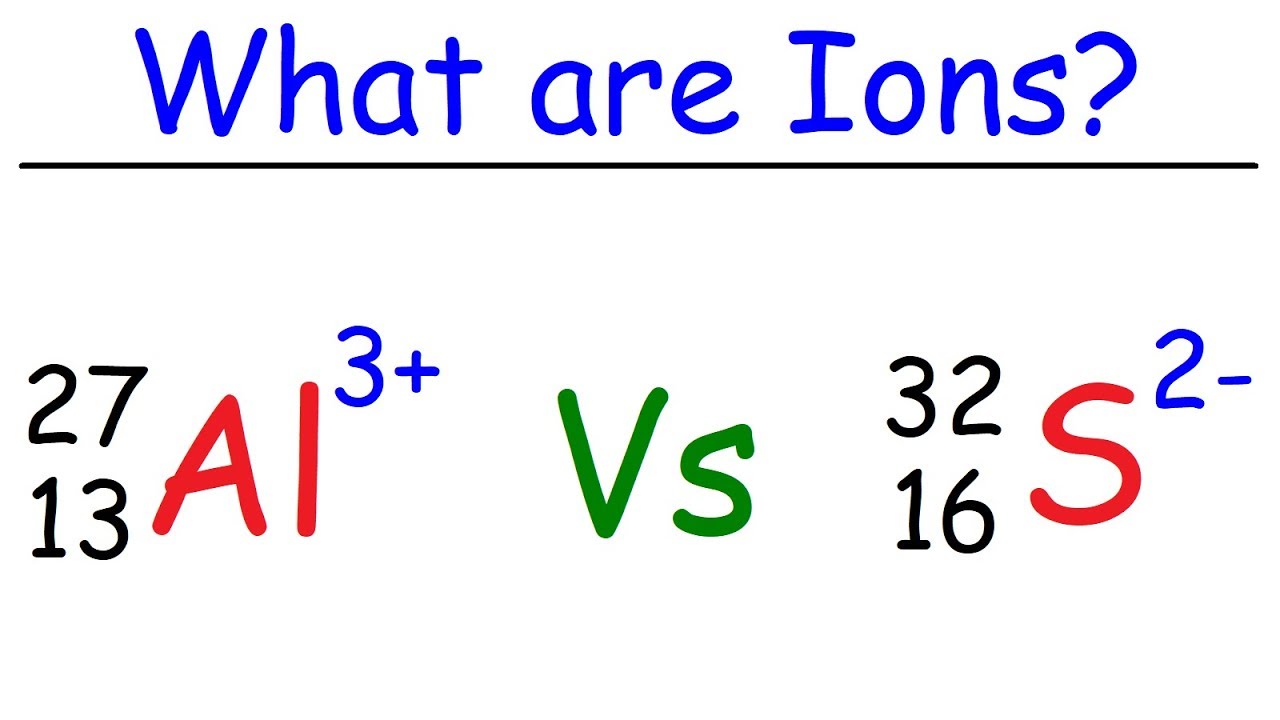What are Ions? Previously, we learned that atoms have equal numbers of protons and electrons which makes them electronically neutral. Now, an atom becomes an ion when it loses or gains electrons.
In an ion, the numbers of protons and electrons are different. Thus, an ion can carry either positive or negative charges. The positive ions or cations are mostly formed by metals.
The negative ones or anions are mostly formed by non-metals. Why ions are formed? Atoms have varied valence electrons, ranging from 1 to 8.
Between them, the valence electron of 8, achieved by noble gases, is the most stable and desirable configuration. To achieve them, atoms need to give their own electrons or receive others’ electrons, depending on their configuration. The protons and neutrons will remain intact as they are located in the nucleus, thus will be harder to move.
Formation of Cations Atoms of metals usually have only a few valence electrons. Therefore, they tend to lose electrons to form cations. In this way, they become stable just like a noble gas.
When an atom loses electrons, it has more protons than electrons, making the overall charge of cations positive. Some metals can form more than one ion. An example of this is that of iron.
Iron can form Fe 2+ and Fe 3+. Wonder how that happens? Psst, it is related to the suborbitals inside the iron atom.
For these metals, the charge on the ion is shown in the name of the compound and can be written as iron (II) sulfate or iron (III) chloride. There are a few non-metallic cations as well. An example of this includes hydrogen ion, H+, and ammonium ion, NH4+.
Formation of Anions Generally, atoms of non-metals have more than four valence electrons. Therefore, they tend to gain electrons to form anions. In this way, they attain the electronic configuration of noble gases.
When an atom gains electrons, it has more electrons than protons. The charge on an anion corresponds to the number of electrons the atom gains. For example, a chlorine atom tends to gain one electron to form Cl- ion.
Hydrogen can also form a negatively charged anion by gaining one electron. It is then called a hydride ion, H-. Polyatomic Ions Polyatomic ions are ions that consist of two or more atoms.
Examples of these are ammonium ion, a combination of an ammonia molecule with a hydrogen ion, NH4+, carbonate ion, a combination of carbon and oxygen bonded together, CO3 2-, and sulfate ion, a combination of sulfur and oxygen. How Do Noble Gases Produce Light? Ions are fascinating particles that can also emit light.
As we studied before, neon is a noble gas that has a complete octet electronic configuration. Its atom does not react with each other and it takes a lot of energy to remove an electron. To force it to emit light, neon gas is filled to a narrow glass tube with electrodes at each end.
A high amount of electric voltage, which can reach as much as 15,000 volts, is applied to the tube. The electricity causes the valence electrons to excite, even remove them from the atom. It produces more collisions that excite more electrons of other atoms.
The excited electron moves to a higher but less stable energy state. To come back to the original state, it needs to release energy in form of the photon that creates the light that is visible to us. The color of the light depends on the composition of gases inside the lamp.
For example, helium produces pinkish light, neon produces orange-red light, argon produces blue, krypton produces green, and xenon produces lavender.