[Music] hi it's Mr Anderson and this is AP environmental Sciences video 33 it's on stratospheric ozone or good ozone we call it good because it protects us against ultraviolet radiation this person's skin doesn't look like it has much damage but if we look at it with UV radiation you start to see blemishes freckles pop out right away and if you were to take a picture of my face or somebody who's much older than me there's going to be more damage due to ultraviolet radiation so we should wear sunscreen what sunscreen does you can see it
on the left side of this person's face is absorb that ultraviolet radiation protects us from the ravages of ultraviolet radiation thankfully our planet is wrapped in sunscreen it's wrapped in this ozone layer protects us especially from UVC radiation why is that important well until we had an ozone layer on our planet until we had an appreciable atmosphere life couldn't exist on land all life had to be in the oceans but once we had the ozone layer protected us the problem is with industrialization we started to produce chemicals like CFCs they release chlorine into the atmosphere
and what they do is they destroy ozone so this is a simulation of what would have happened if we didn't stop using uh these cfc's over time this is a NASA prediction so you can see here on the bottom that over time by 2020 by 2040 by the time we get to 2060 essentially all of that ozone is gone all of that protection is gone as well and so ozone is simply oxygen instead of being oxygen diatomic oxygen two atoms of oxygen it's actually three atoms of oxygen connected together it's found in two layers in
our atmosphere in the troposphere we call that the bad ozone because it leads to pollution smog for example is produced through ozone but what I'm talking about is the good ozone the stratospheric ozone that forms the O ozone layer that protects us on this planet protects us from ultraviolet radiation now some of that gets through uh that ultraviolet radiation is ordered from A to B to C in ascending energy and also descending wavelength the UVA radiation moves right through the ozone layer but this is not too damaging if you've ever looked at a a black
light that's UVA radiation most of the UVB is filtered out and all of the UVC the most damaging type of ultraviolet radiation is filtered out by the stratospheric ozone the nice thing about that UVC radiation is that when it's hit regular oxygen forms ozone which can then be broken down again so we have this nice feedback cycle as long as the sun is shining as long as we have ultraviolet radiation we're producing ozone to protect us the problem is we started producing chemicals like chlorofluorocarbons which released chlorine into the atmosphere and what it does is
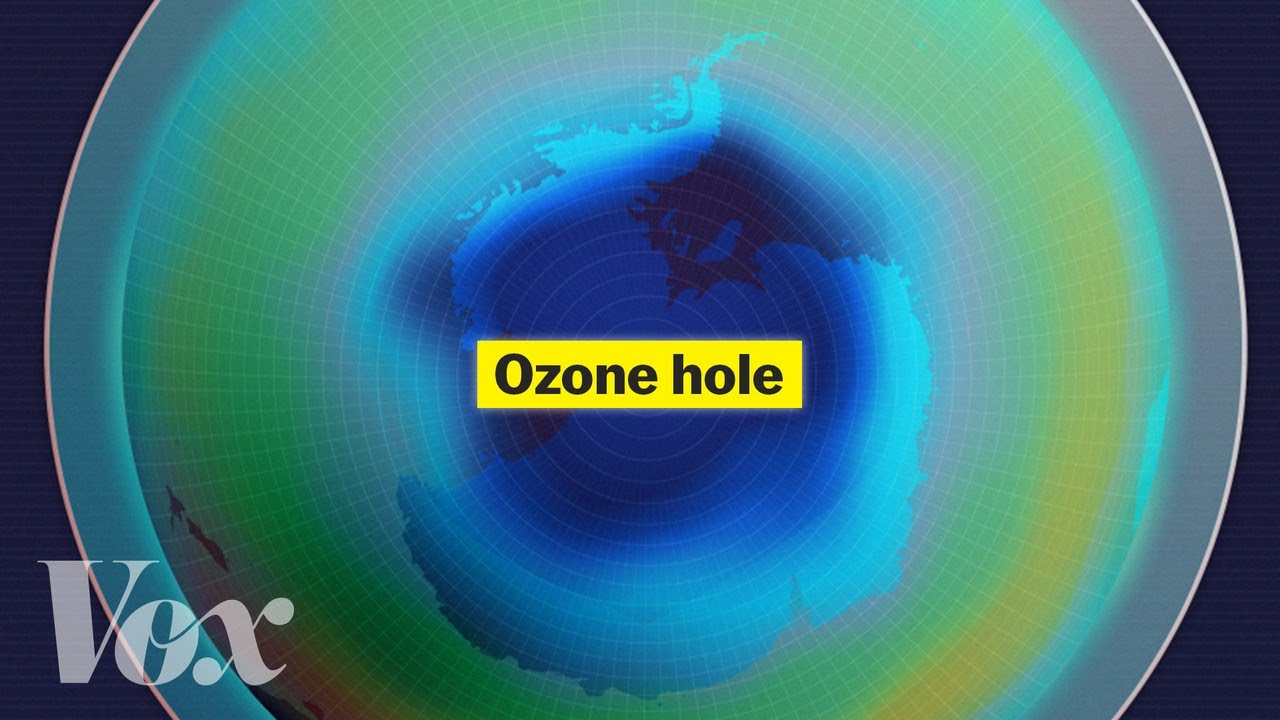
it acts as an enzyme to break down that ozone and so we had the hole in the ozone layer you're probably familiar with that it would have gotten larger if we didn't do something to stop it and so 1989 the Montreal protocol was put forward as a way to limit cfc's in our on our planet so if if we look at the amount of ozone on this graph this is going to be right near the surface of the Earth We're going to have large amounts near the surface and it's going to drop off and then
we're going to have large amounts as we go you know kilometers away from the surface this near the bottom is called tropospheric ozone this is that bad ozone it can create smog also leads to lung irritation can lead to things like lung cancer as we move farther and farther up we get into the stratospheric ozone that's the ozone we're talking about this is the ozone layer large amounts of ozone high in the atmosphere here it's this O3 molecule it doesn't start as an O3 molecule we basically have a bunch of regular oxygen datomic oxygen it's
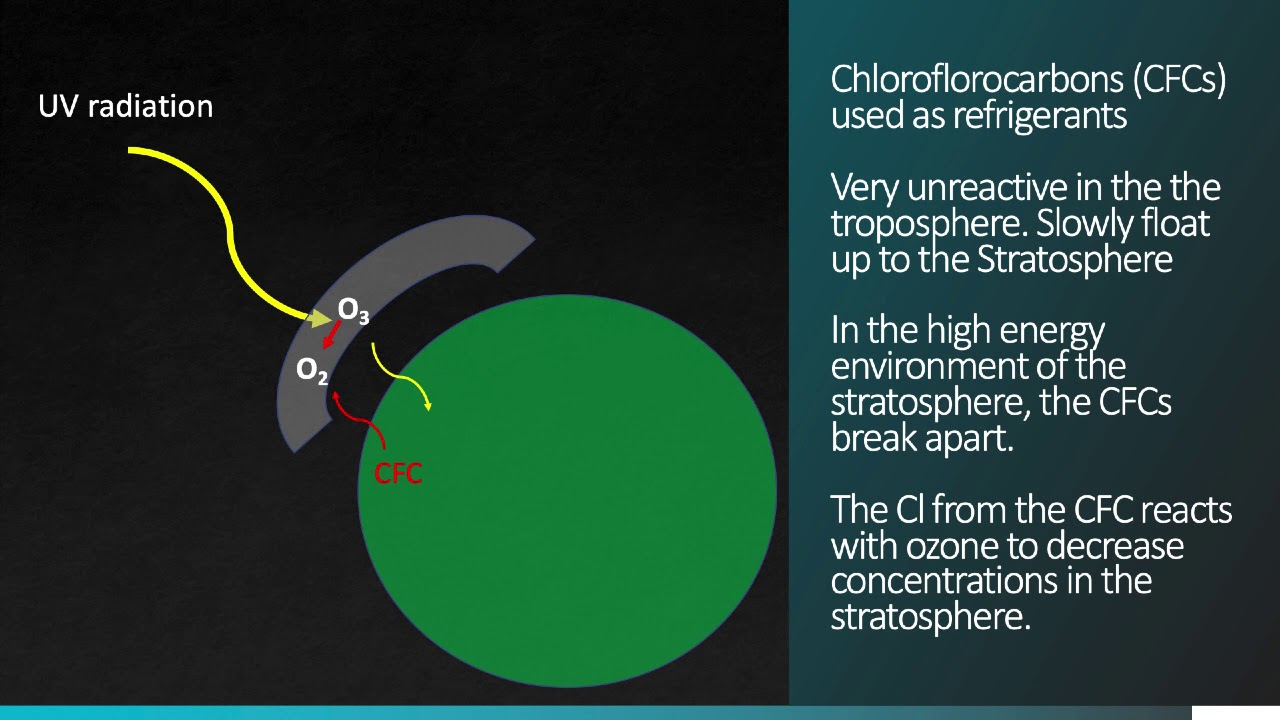
hit by ultraviolet radiation and it breaks it apart into two atoms of oxygen this free atom of oxygen bonds to datomic oxygen and now we've got our ozone that's how it's formed we need the radiation to form the ozone it's also how it breaks down so if it's hit by another bit of UV radiation then it breaks out again and now we're doing the cycle we're doing the cycle of solar form when we take regular oxygen hit it with a photon break it into these free atoms which then bond to another oxygen atom to make
ozone and then we have solar breakdown that's when we have the ozone it's hit with a photon we break it down and then we make that good oldfashioned oxygen again and so it's a nice cycle as long as the sun is shining we're going to have ozone and the importance of ozone is protection so if we look at the amount of UV radiation it moves right through those layers in our atmosphere UV UVB is mostly filtered out and UVC is completely filtered out and you might think why is that well it has to do with
the wavelength of that radiation um thankfully the radiation's wavelength fits that oxygen Bond it's able to break it apart and it's able to make that Ozone now the problem is we started producing things like chlorofluorocarbons what is a chlorofluorocarbon if we just break down the word it's three chlorine atoms so that's the chloro part we then have the floro part so it's one Florine atom and then we have a carbon atom and what happens when chlorofluorocarbons are hit by radiation is it breaks off a chlorine atom now watch what happens to that chlorine atom it
grabbed one of the atoms from an ozone so if we look at the atmosphere we've got a lot of ozone at this point but now that we've freed up that chlorine it grabs another one it grabs another one it grabs another one and so what it's doing is it's breaking down all that ozone it's acting as an enzyme it's not consumed in the reaction it simply breaks down the ozone and one chlorine atom can break down 100,000 ozone atoms and so it goes really really quickly and that led to the ozone hole now why is
that ozone hole found on the pole remember there's going to be an area where we're not having a lot of light there's not a lot of radiation so we can't build that ozone back up and so we started to see it here first but it would have spread across the whole planet and so what humans did is they got together and they formed the Montreal protocol it's a treaty that all of these countries were signing and they said we have to stop using these CFCs and so if you look at where we were were headed
with the amount of CFCs we were producing things in freon for example aerosols for example we were leading to a destruction of the ozone layer but thanks to the Montreal protocol we've dropped off the amount of CFCs in the environment and over time that ozone is going to build itself up again and so did you learn the following could you pause the video at this point and fill in the blanks well let me do it for you ozone remember is o03 rather than O2 it mostly filters out UVC radiation but also filtered some of the
UVB this is Ozone found in the stratosphere or stratospheric ozone CFCs release chlorine which form the ozone hole thankfully the the Montreal protocol uh helped to avoid that and the misconception you want to avoid is that we're not talking about tropospheric ozone this polluting ozone near the atmosphere we're talking about that protective good ozone high in the atmosphere and I hope that was helpful [Music] a [Music]