How is an atom like a flight of stairs? When you picture an atom, you may have something like this in mind. There's the nucleus in the center and the electrons orbiting around it, like planets orbiting the sun in different levels.
There are a few ways that this image is, let's say, not entirely accurate, but it does illustrate something essential to quantum mechanics. The fact that it's, well, quantum. Let's talk about what quantum really means.
Before quantum mechanics, physicists thought electrons really did orbit the nucleus of an atom like planets. If you gave an electron a little more energy, it would change its orbit. And if you gave it too much, it would leave the atom entirely.
But while it's true that some electrons are closer into the nucleus and some are farther out, each one has a specific energy, and there's only a limited set of distinct energy levels that are allowed. It's kind of like going up or down a set of stairs. If I give myself just the right amount of energy, I can go up one step.
With a different amount, I can go up two. A big enough leap will send me off the staircase altogether. But something in between doesn't work.
I can't go up half a step or a third of a step. The energy I need to put in is quantized, meaning it comes in discrete packets or steps. The word quantum just means a fixed amount of something.
A snowflake is a quantum of snow. A person is a quantum of humanity. A stair is a quantum of stairs.
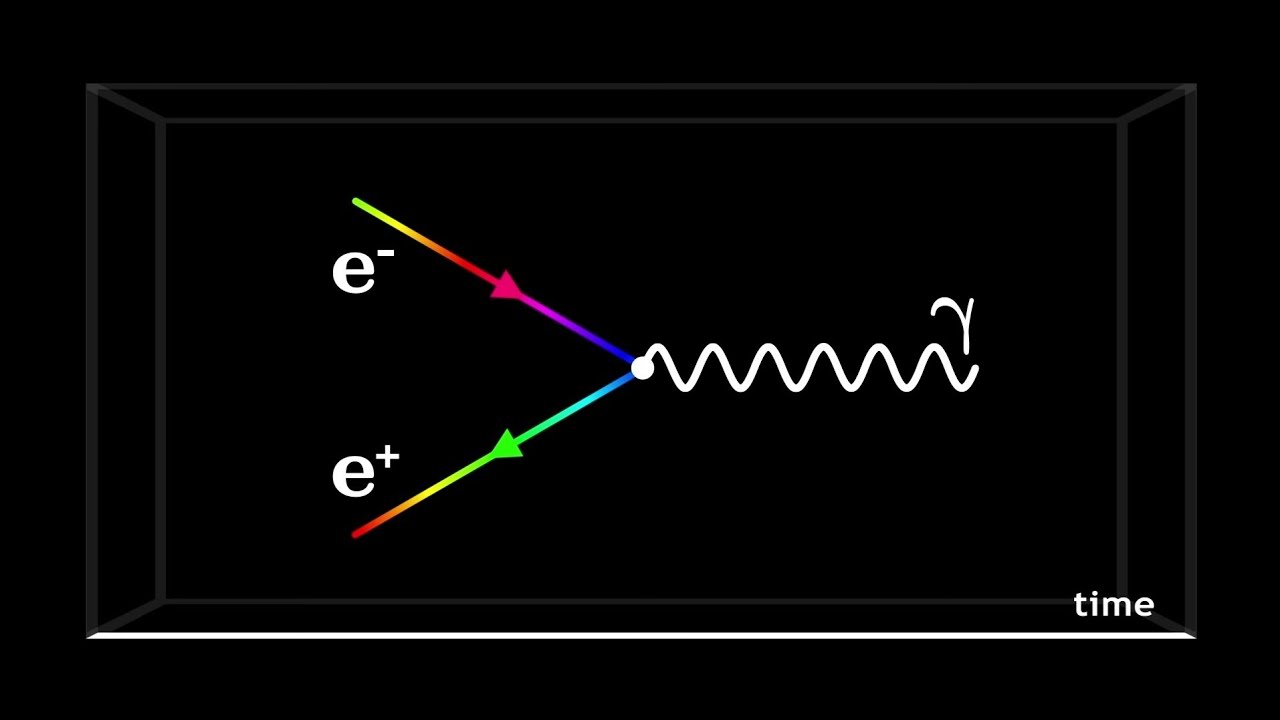
Some of the most important breakthroughs in physics came about due to the discovery that energy, along with momentum, spin, and as you might expect, particles, is quantized. Because energy levels of atoms are quantized, moving an electron up or down can only happen when the packet of energy is just the right size. A photon, which is a quantum of light, or, more generally, electromagnetic energy - can move an electron up to a higher energy level only if it has just the right energy.
Each kind of atom or molecule has a different set of energy levels, like a staircase with steps of different sizes. We can see this most clearly when we look at the spectrum of a beam of light passing through a gas. Light that passes through hydrogen gas will be absorbed by the hydrogen atoms, but only at very specific wavelengths or colours.
Those are the wavelengths at which the energy of the light exactly matches a transition between two energy levels of hydrogen. Because the light at those specific wavelengths is absorbed, when we spread the light out in a prism, we see that there are gaps - dark lines at just those colors. This is how astronomers first figured out that the sun is made of mostly hydrogen, and it's how we can detect which elements are in the atmospheres of other stars and planets.
Spectra like fingerprints. You can use them to ID chemicals, and that's super useful. There are actually two different types of spectra, absorption spectra and emission spectra, but both of them involve set lines at specific colors arranged in a pattern.
One of the coolest things to me about spectra is, it’s kind of tempting to think that Quantum is so small. Regular people could never see it. But with Spectra experiments with your own two eyes, you can see the quantum nature of electrons.
We know from experiments that quantization happens at subatomic scales. But for many years, physicists have been grappling with the possibility that everything, ultimately, is quantum: including gravity and spacetime itself. Maybe the force that holds me on the staircase right now comes in tiny quantum packets called gravitons.
And maybe the space we exist in is quantized too, in tiny spacetime pixels. If physicists can find a way to quantize gravity, they would have the ultimate prize in theoretical physics. A theory of everything.
From where I'm standing, that would definitely be a step up.