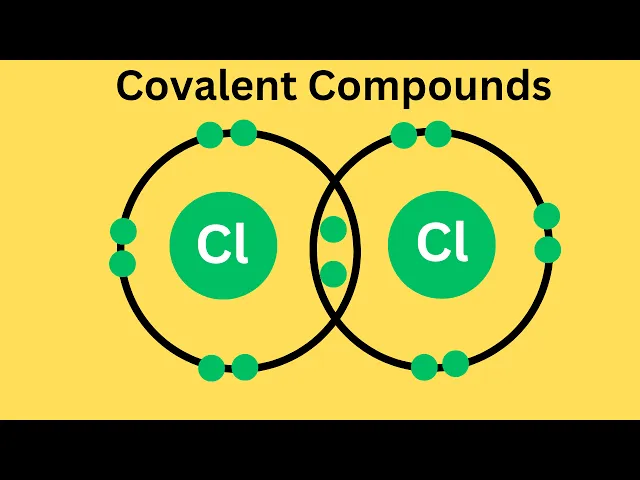
welcome to moomoomath in science and covalent compounds a molecule or compound is made when two or more atoms form a chemical bond that links them together covalent compounds generally result from two non-metals reacting with each other the elements form a compound by sharing electrons resulting in an electrically neutral molecule for the most part if you have two or more non-metals chemically combined it will be a covalent compound for example in water each hydrogen and oxygen share a pair of electrons to make a molecule of two hydrogen atoms single bonded to a single oxygen atom other
examples include ammonia chlorine and carbon dioxide in general covalent bonds occur between elements that are close together on the periodic table solid covalent compounds tend to be soft covalent compounds have relatively low melting and boiling points some covalent compounds dissolve in water like sugar and others do not like lipids most covalent compounds are poor conductors of electricity and poor thermal conductors thanks for watching and remember kindness multiplies kindness be kind to someone today