foreign executive editor of any JM evidence and this is stat it's a big day you're a clinical trialist and today is the day you begin enrolling participants in your double-blind randomized controlled trial of a new sleep medicine 50 participants will be randomized to the treatment arm and another 50 to the placebo arm now imagine that a few days into the trial one participant assigned to the treatment arm starts having Vivid nightmares worried she's experiencing side effects from the study drug she decides to skip the medication meanwhile a participant in the placebo arm keeps forgetting to
take his pills and still another has unexpected travel and leaves the pills at home at the end of the study period when everyone returns for a pill count these participants and some others still have most of their pills left so what should you do with these participants data and the data from all the others like them who didn't stick to the assigned treatment protocol for one reason or another this question comes up in clinical trials all the time while investigators hope for perfect adherence reality is messy non-adherence happens for lots of reasons some related to
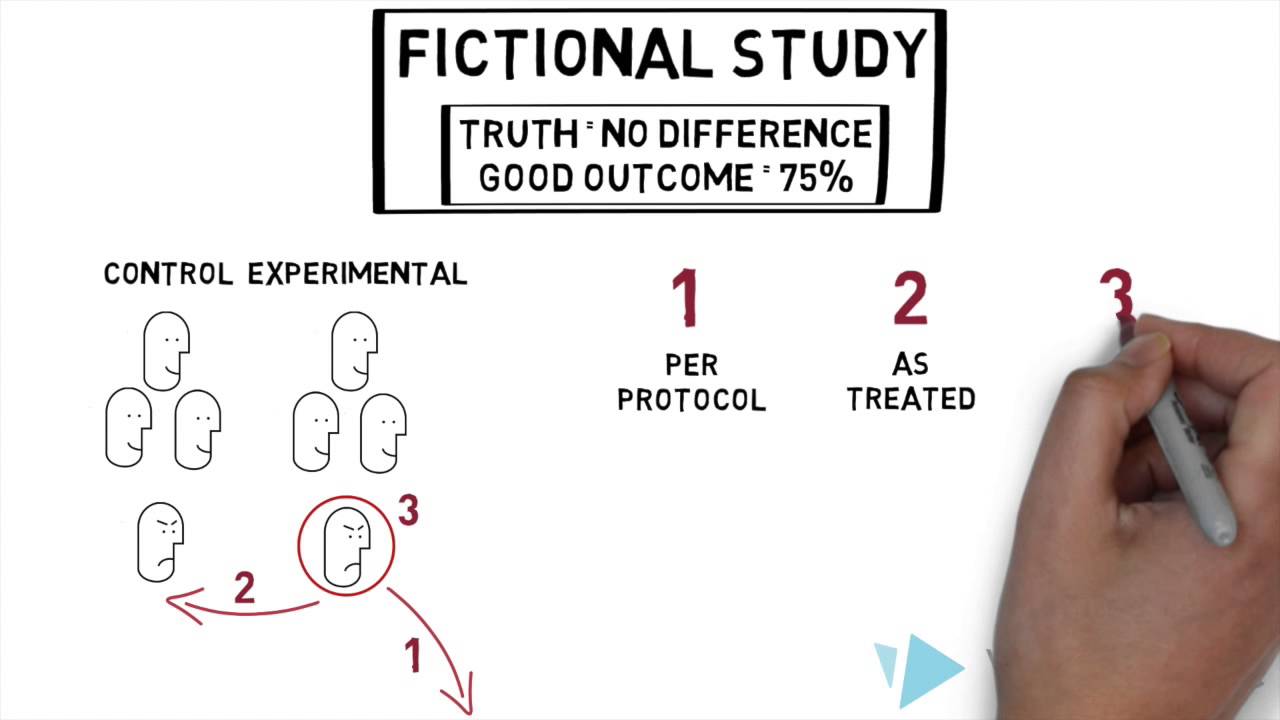
the study drug think side effects and some not think just forgetting to take the pills in all cases investigators must decide how to analyze the data when things don't go according to plan but don't lose sleep over it three common analytic approaches that you may have heard about are per protocol as treated and intention to treat let's take a look at each a per protocol analysis focuses only on participants who stuck to the protocol excluding those who didn't take the pills switch treatment arms or took the pills differently than prescribed it might sound dreamy but
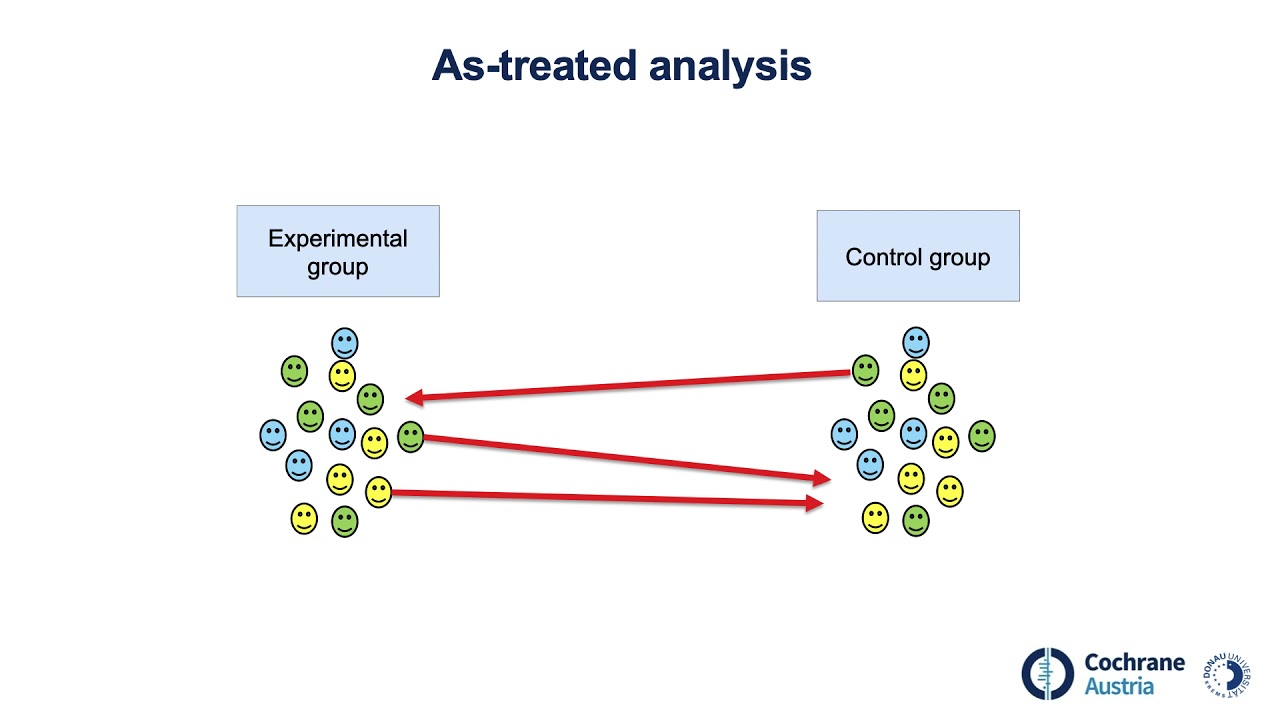
this approach comes with its own problems first you might exclude participants who stopped the treatment because of its side effects that's important information second excluding participants slashes the sample size and reduces statistical power third a per protocol analysis introduces bias participants who adhere to the protocol may be very different from those who don't by excluding some participants you lose the benefits of randomization effectively turning your experiment into a non-experimental study second option is the as treated analysis in which participants are analyzed based on the treatment they actually received rather than what they were assigned imagine
the participant with nightmares lets you know right away she was opting not to take the treatment and as treated approach might lead you to analyze your data as part of the placebo group this approach may give you a real world snapshot of how people decide on treatment options but it too eliminates randomization again essentially giving you a non-experimental study on the other hand in the intention to treat analysis every participant randomized at the beginning of the trial is analyzed according to their original group assignment regardless of adherence this maintains all the benefits of randomization and
keeps the sample size and similarity between the treatment and Placebo groups intact what are the downsides well because both trial arms are likely to have participants who didn't follow the protocol the outcomes in the two groups may appear more similar than they might have been headed here and it's been better keep in mind an intention to treat analysis estimates the effect of being assigned to treatment for cisibo only with perfect adherence does it estimate the effect of actually taking the medication of these options the intention to treat approach is the only one that preserves the
key benefits of randomization that's why it's the go-to approach in randomized trials and why you decide to stick with analyzing all the participants in both arms according to their assigned group this also makes clear just how important it is to track the treatments participants actually receive instead of just assuming that everything worked like a dream there are other Advanced statistical methods we could mention but for now we'll let those sleeping dogs lie