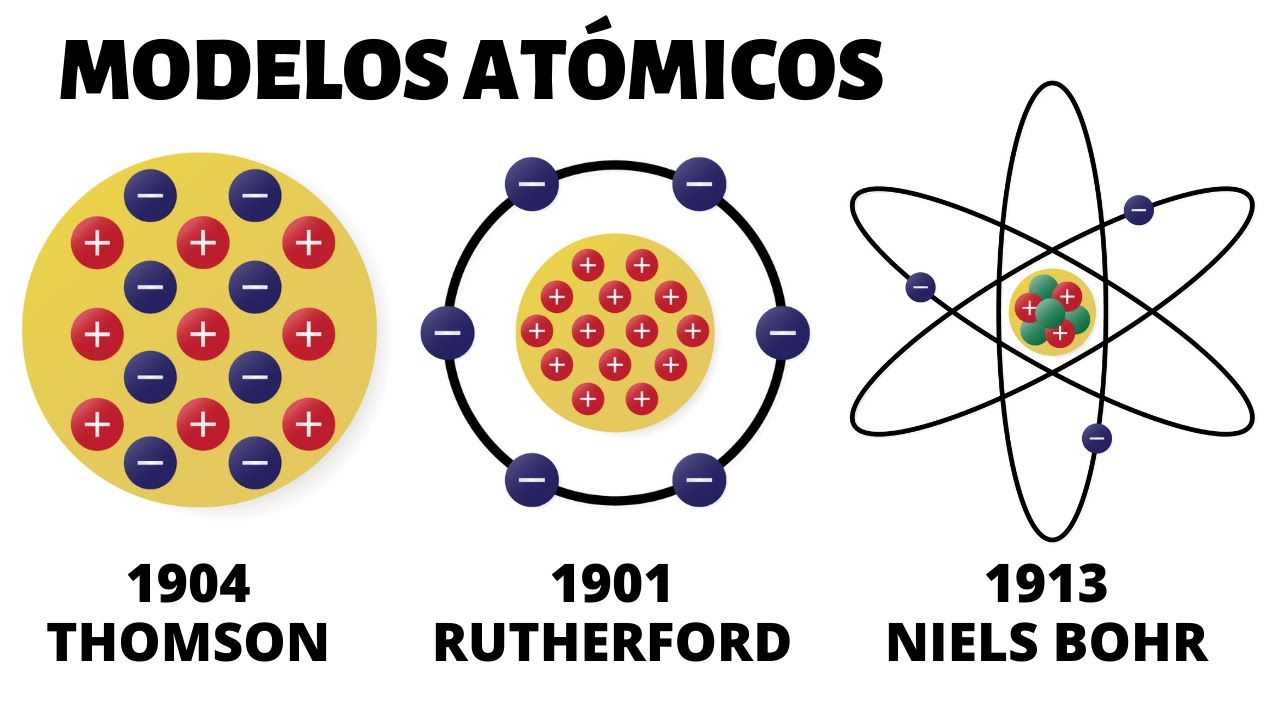
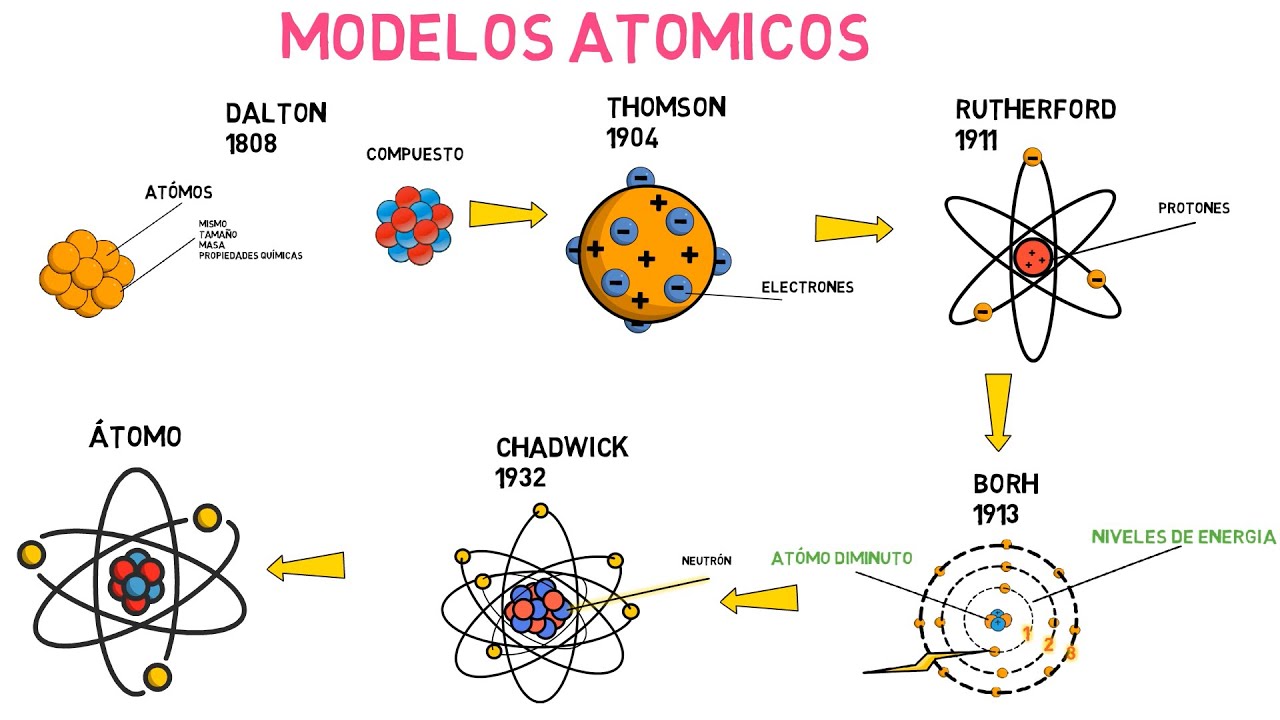
Thompson's atomic model was created by the celebrated English physicist Jay Jay Thomson who discovered the electron for this discovery and his work on electrical conduction in gases and was awarded the Nobel Prize in Physics in 1906 for his work with the cathode rays it became clear that the atom was not an indivisible entity as dalton had postulated in the preceding model but that it contained a well-defined internal structure thomson developed a model of the atom based on the results of his cathode ray experiments in it he stated that the electrically neutral atom was made up of positive and negative charges of equal magnitude the positive charge was distributed throughout the atom and the negative charges were embedded in it like raisins in a pudding from this comparison the term raisin pudding arose as he met the model informally although at present Thompson's idea looks quite primitive for At the time, it represented a novel contribution during the brief validity of the model from 1904 to 1910. It had the support of many scientists, although many others considered it heresy. Finally, in 1910, new evidence arose about the atomic structure and the Thompson model was quickly left behind.
On the other hand, this happened as soon as Rutherford published the results of his scattering experiments that revealed the existence of the atomic nucleus. However, Thompson's model was the first to postulate the existence of subatomic particles and his results were the result of a fine and Rigorous experimentation in this way set the precedent for all subsequent discoveries Characteristics and postulates of Thompson's model Thompson arrived at his atomic model from several observations The first was that X-rays newly discovered by Ruth at Ghent were capable of ionizing air molecules until then the only way to Ionizing was chemically separating ions in a solution, but the English physicist managed to successfully ionize even monoatomic gases like helium using x-rays. This led him to believe that the charge inside the atom could be separated and therefore was not indivisible as well.
observed that cathode rays could be deflected by electric and magnetic fields then thompson devised a model that correctly accounted for the fact that the atom is electrically neutral and that cathode rays are composed of negatively charged particles using experimental evidence thomson characterized the atom in the following way the atom is an electrically neutral solid with a radius of approximately 10 m the positive charge is distributed more or less evenly throughout the sphere the atom contains negatively charged corpuscles that ensure its neutrality these corpuscles are the same for all matter when the atom is in equilibrium there are n corpuscles os regularly arranged in rings within the sphere of positive charge the mass of the atom is evenly distributed cathode rays thompson carried out his experiments using cathode rays discovered in 1859 cathode rays are beams of negatively charged particles to produce them tubes are used vacuum glass in which two electrodes called cathode anode are placed, then an electric current is passed through that heats the cathode, which in this way emits invisible radiation that is directed directly to the opposite electrode to detect the radiation that is nothing other than the cathode rays the wall of the tube behind the anode is covered with a fluorescent material when the radiation reaches there the wall of the tube gives off an intense light if a solid object is placed in the path of the cathode rays it casts a shadow on the wall of the tube tube this indicates that the rays travel in a straight line and also that they can be easily blocked the nature of cathode rays was widely discussed because their nature was unknown some thought that they were electromagnetic waves while others maintained that they were particles subatomic particles of the model thompson's atomic model is as we said the first in postulate the existence of subatomic particles thompson's corpuscles are nothing more than electrons the fundamental negatively charged particles of the atom we currently know that the other two fundamental particles are the positively charged proton and the uncharged neutron but these were not discovered at the time in which thompson elaborated his model the positive charge in the atom was distributed in it he did not consider any particle carrying said charge and for the moment there was no evidence of its existence for that reason his model had a fleeting existence since in the course of few years the experiments of dispe Rutherford's reaction paved the way for the discovery of the proton and as for the neutron, Rutherford himself proposed its existence some years before the Crookes tube was finally discovered Sir William Crookes 1832 1919 designed the tube that bears his name around 1870 with the Intending to carefully study the nature of cathode rays, he added electric fields and magnetic fields and observed that the rays were deflected by them in this way . cathode 2 rays were deflected by the presence of magnetic fields in the same way that negatively charged particles were 3 any metal used to make the cathode served equally well to produce cathode rays and their behavior was independent of the material these observations fueled the discussion about the origin of the rays The cathode ones who maintained that they were waves were based on the fact that cathode rays could travel in a straight line. In addition, this hypothesis explained very well the shadow that an intervening solid object cast on the wall of the tube and under certain circumstances it was known that the waves could cause fluorescence .