Hello, in this video we will see how surfactants reduce surface tension. First we will study structure of surfactant molecule. Then we will quickly see surface tension.
And then we will see mechanism of action of surfactants. Let's get started. First let's see structure of surfactant molecule.
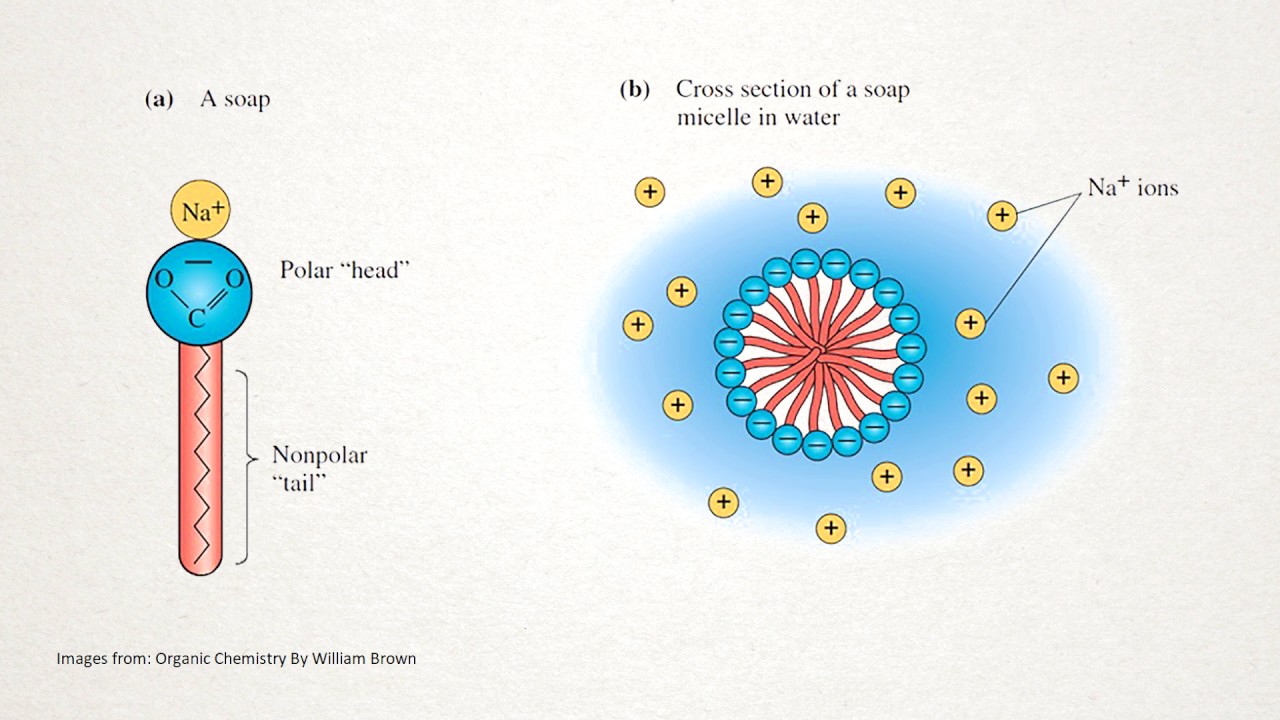
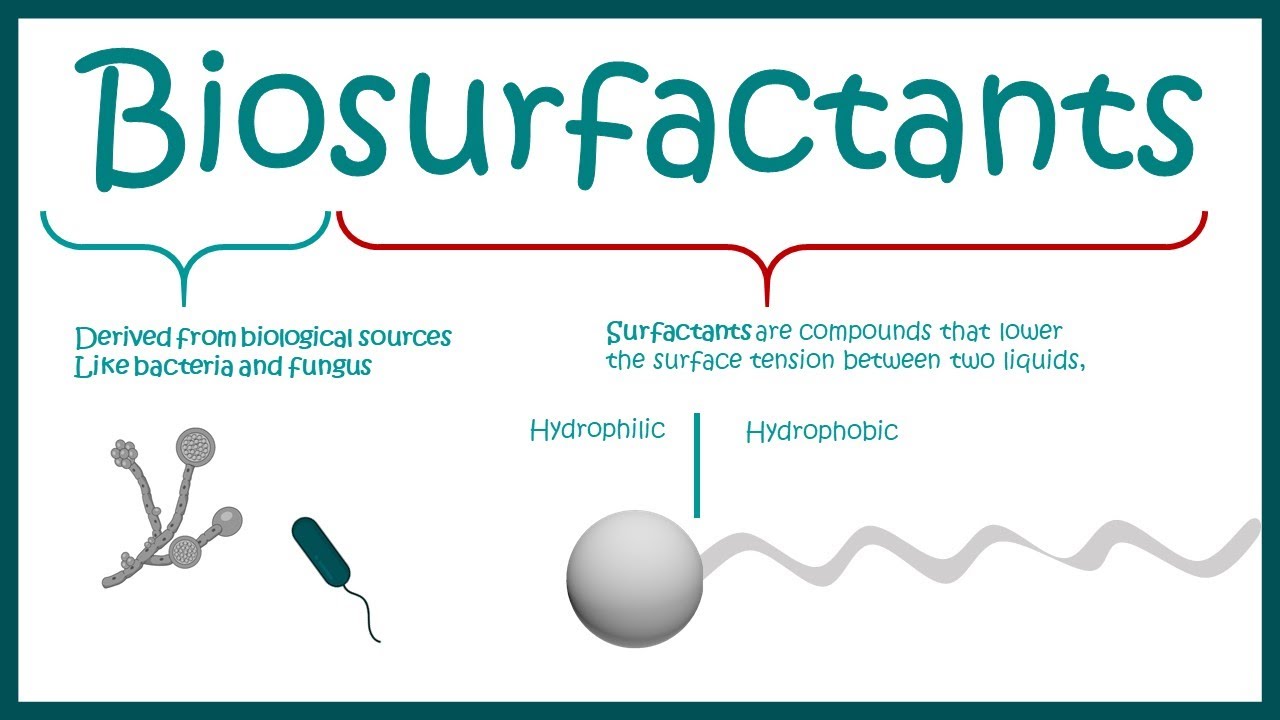
Surfactants are lipids or proteins. Their general structure is like this. They have a head and a tail.
The head has charged atoms. Due to this it dissolves in water or in other words it's hydrophilic. The tail on the other hand do not have any charged moiety.
So it's hydrophobic. This was about the structure. Now let's quickly revise what we have seen in video on surface tension.
We saw that water molecules attract each other. Molecules deep inside the bulk are attracted equally in all the directions. So there in no net force acting on them.
However, Water molecules on surface do not have any other water molecules upwards to pull them. So they experience net pull downwards by water molecules in the bulk. As they dive deep, the water molecules that stay on the surface develop surface tension.
This is how surface tension develops. The key point that I want you to focus here is this downward pull on surface molecules. It's this pull that's causing surface tension.
Now let's throw a surfactant molecule in water. In this molecule, this head is hydrophilic. So it wants to stay with water.
However this tail is hydrophobic. So it wants to stay away from water. One solution to make both of them happy is this orientation at the surface.
See this water soluble head is in contact with water. Due to the charged moiety, it is attracted by surrounding water molecules. The tail is out of water in air phase.
As it's hydrophobic, it pulls the surfactant molecule upward away from water. All these forces balance each other. So there is no net force acting on the molecule and it stays on the surface.
But, how does it reduce surface tension? One, as it occupies area on the surface, density of water molecules on surface decreases. The lesser the water molecules on the surface, the lesser the surface tension.
Second, the head portion of the surfactant molecule attracts the water molecules near surface. This attraction opposes the downward pull on these water molecules. Here, surfactant molecule is prevented from going deep by hydrophobic tail.
Now look at the downward pull on the surface molecules. It's lesser as compared to that without surfactant. Thus surfactant reduces tendency of surface molecules to dive deep in bulk water.
This results in reduced surface tension. The more the surfactant molecules on the surface, the less the surface tension. I hope now it's clear how surfactants work.
We will see specific problems solved by surfactants in a separate video. If you struggle understanding medical concepts, subscribe to the channel and also follow me at other platforms. At nonstop neuron.
com, learning medical concepts is as easy as watching cartoons. Thanks for watching, see you in next video.