The Compton effect or Compton scattering is a phenomenon that happens when a high energy photon interacts with an electron. Upon colliding, the photon transfers some of its energy to the electron, ejecting the electron from its veilent shell and also scattering the photon. Since the scattered photon has less energy than before the interaction, it also has a longer wavelength than before the interaction.
Before this effect was known to exist, the prevailing theory of light scattering was known as Thompson scattering, named after English physicist JJ Thompson. Thompson scattering uses classical electromagnetism to explain interactions between light and electrons. According to this model, when light interacts with an electron, the electric and magnetic components of the light exert a lent force on the electron.
The electron absorbs the light and sets in motion and as a consequence of acceleration emits radiation which is the same wavelength of light as the incident beam of light. This model of scattering dominated the scientific community ever since its proposal in 1906 but did have a couple of holes. The first hole was that Thompson's model predicted the scattering to be symmetrical along the incident axis.
But this was contradicting future scattering experiments where high energy light beams like X-rays and gamma rays were not in fact scattering symmetrically. The second hole came from experiments headed by English physicist Charles Barka which showed that the light with low wavelengths like X-rays and gamma rays was being absorbed during scattering at a much lower rate than predicted by Thompson scattering. In the late 1910s, an upand- cominging American physicist by the name of Arthur Compton also began to investigate these anomalies.
He initially tried to reconcile the problem of asymmetry with classical physics, going so far as to even propose the idea of a ring-shaped electron with a radius of two pometers, which is about 1,000 times as large as today's accepted radius. However, upon trying to reconcile the second issue of low absorption, he ran into an additional problem that led to him eventually abandoning classical mechanics entirely. In 1921, in a lab in the basement of a building at the Washington University in St.
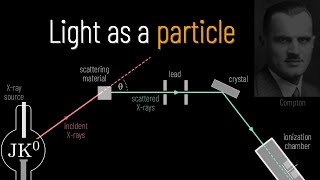
Louis, Compton made the choice to observe the spectrum of scattered X-rays rather than the intensity. His setup began with a lead box that housed an X-ray source from the element malibdinum. The box also contained a chunk of graphite, and the X-ray beam would reflect off of this graphite at a 90° angle.
After this deflection, the X-ray beam would exit the lead box through a small slit and then go through a Bragg spectrometer. A Bragg spectrometer works as follows. First, the beam defracts through a calite crystal.
Then, the X-rays that have been scattered and now also defracted would be collected in an ionization chamber attached to an electrometer to analyze. Using Bragg's law, he could determine the wavelengths of the X-rays based on the angle of incidence of the X-rays upon scattering through the calsite crystal. He then plotted intensity versus scattering angle graphs for both non-scattered X-rays shown as the dotted line and the scattered X-rays shown as the solid line.
As the glancing angle or angle of incidence increases, so does the wavelength that the Bragg spectrometer is sensitive to. As you can see, the scattered X-rays underwent a wavelength shift, which Compton initially attributed to a Doppler shift in an attempt to reconcile classical mechanics. He blamed this Doppler shift on rapidly recoiling secondary electrons.
But if there was a Doppler shift, this effect would virtually disappear at low light intensities regardless of the wavelength. And it's clear here that the wavelength shift happens at all intensities. Compton was left with no choice but to rework his theory entirely, abandoning classical mechanics in favor of quantum mechanics.
Instead of looking at X-ray scattering as a high energy wave being absorbed by an electron and remitted, he looked at it as a collision between a light particle and an electron. Then in his math, he was able to conserve energy and momentum and then use a lot of clever algebra to arrive at his equation describing the change in wavelength of a photon after it scatters off of an electron. With this equation, suddenly the holes in Thompson scattering were fixed through quantum mechanics.
He published this paper in 1923 and by the end of the year, Charles Wilson observed these recoiling electrons in his cloud chamber. The two of them shared the Nobel Prize in physics for this work in 1927. Compton's work on X-ray scattering has been recognized by many as the pivotal moment in history when scientists left classical theory in favor of quantum physics as it showed complete concrete evidence of the wave particle duality of light proposed by Einstein way back in 1905.
As Compton himself put it in his paper, there was now quote little doubt that the scattering of X-rays is a quantum phenomenon end quote. If you enjoyed this video, please consider liking and subscribing. Click here if you want to see more scientific progress made during this time period.
Thank you for watching and I will see you in the next video.