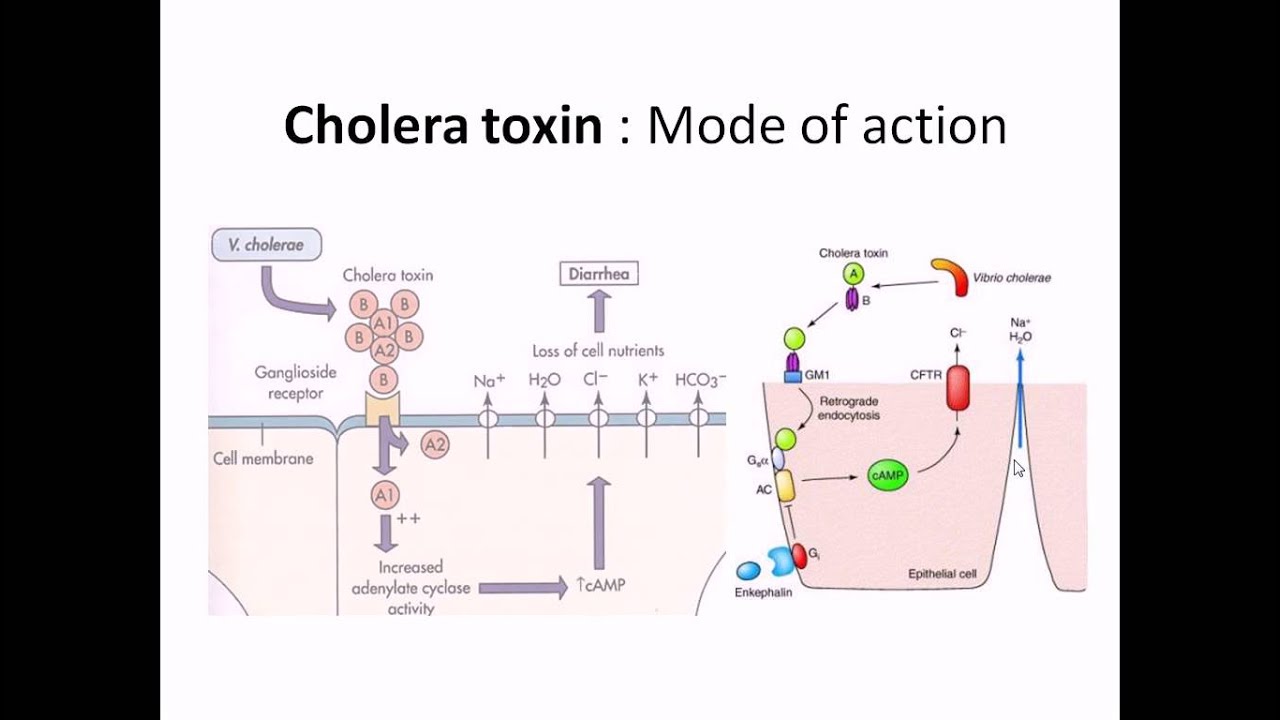
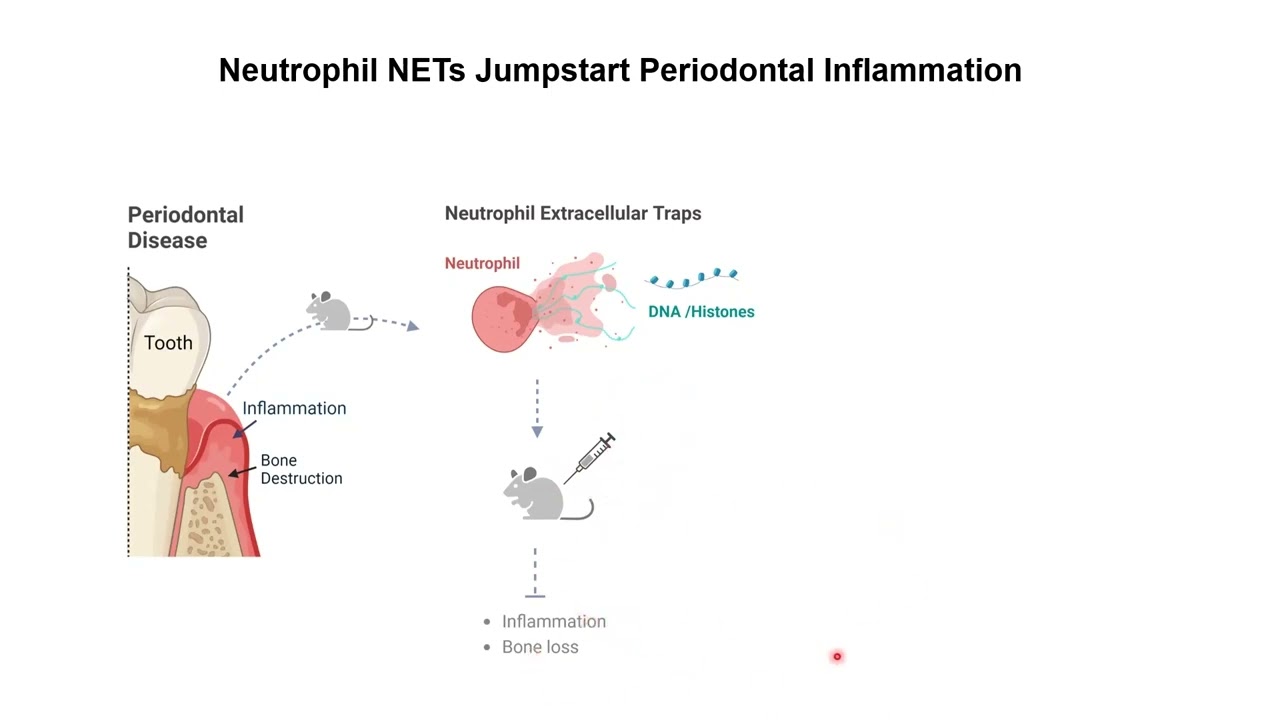
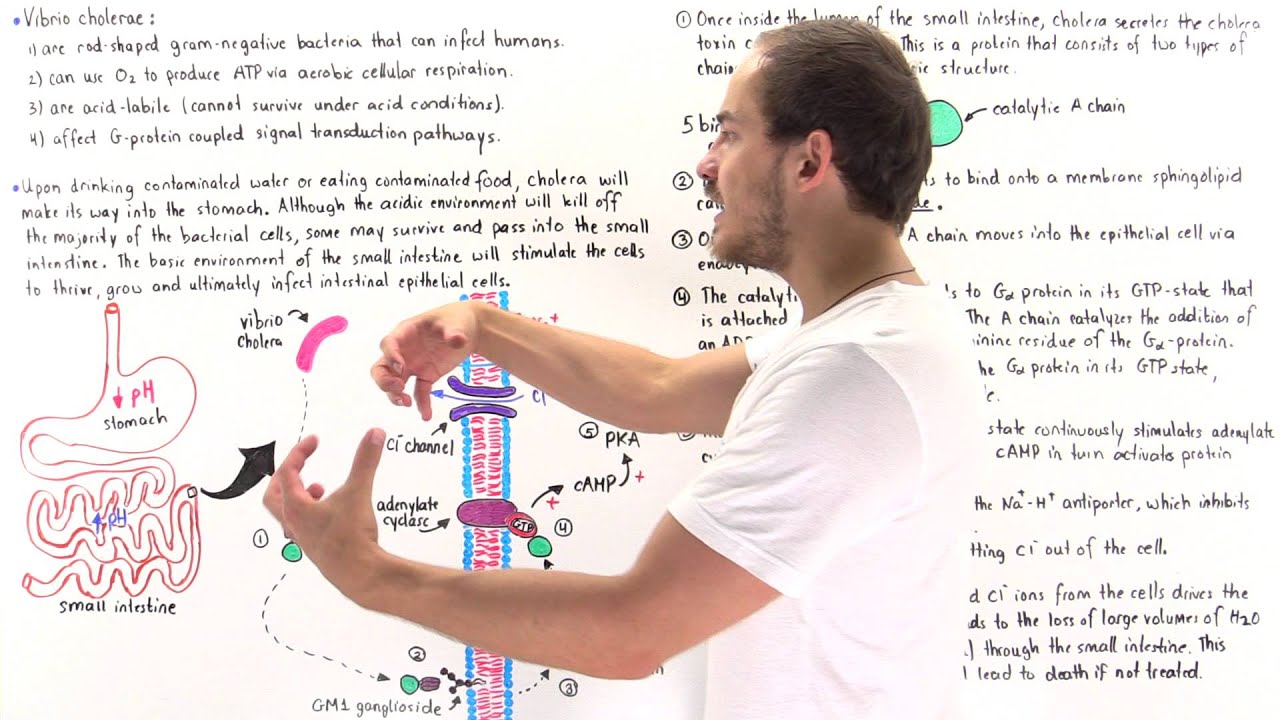
cholera a life-threatening disease marked by severe diarrhea is caused by the bacterium vibrio cholerae the diseases typically transmitted by the ingestion of food or water contaminated with human feces in the intestinal tract cholera Vibrio SCID here to the surface of cells the bacterium secretes cholera toxin which has two types of subunits five copies of a B subunit which functions in binding to host cell receptors and one copy of an a subunit which exhibits the toxic activity cholera toxin is structurally similar to many other bacterial toxins that are generally termed a B toxins because it specifically
affects the intestines cholera toxin is also called an enteric auxin each of the five B sub units but binds to a glycolipid called ganglia side gm1 on the intestinal cell membranes in this way cholera toxin binds tightly to intestinal epithelial cells the a subunit consists of two domains the a1 domain which contains the toxins activity and the a2 domain which tethers the a subunit to the B ring a protease clips the a subunit into two polypeptides which remain together by virtue of a disulfide bond The Binding of cholera toxin to gm1 triggers endocytosis the resulting
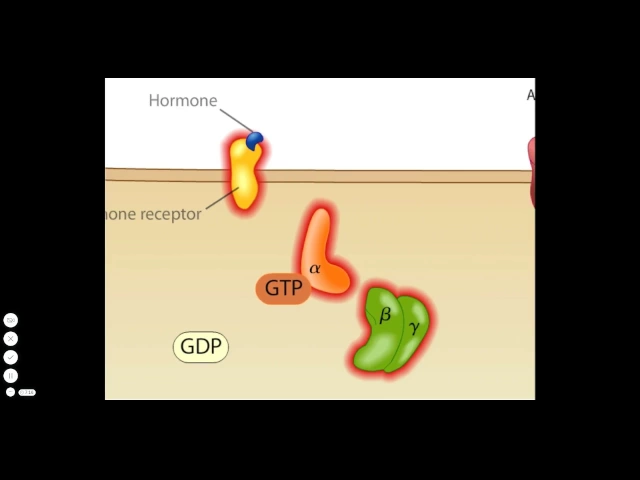
toxin containing vesicle is transported to the endoplasmic reticulum the a1 peptide is then released from a2 and the rest of the complex by a reduction reaction at the disulfide bond from the lumen of the endoplasmic reticulum the toxin is then transported into the cytoplasm the mission of the a1 peptide is to modify a membrane associated protein called a g-protein that binds to the enzyme adenylate cyclase and controls its activity a g-protein normally functions in the following way an inactive g-protein resides in a complex made up of alpha beta and gamma subunits the inactive alpha subunit
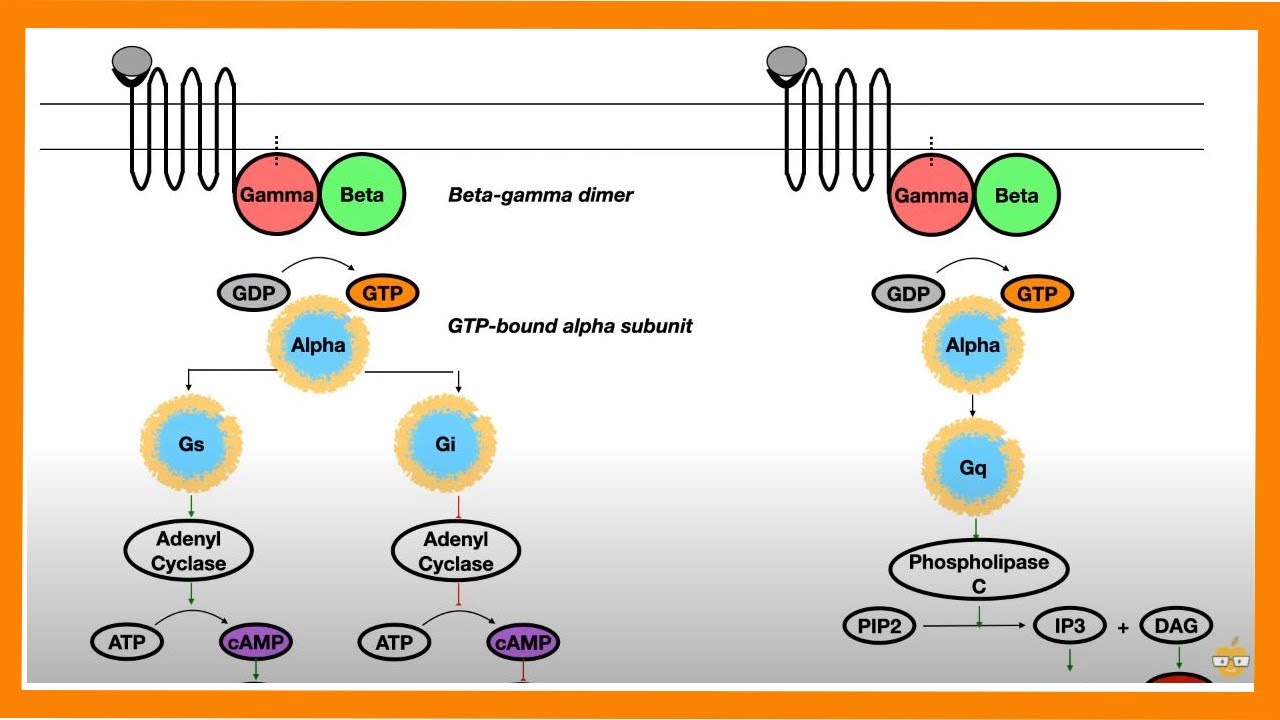
carries a gdp molecule when a hormone activates an appropriate receptor the activated receptor can activate the g-protein allowing it to release GDP and take up gtp the alpha subunit separates from the beta and gamma subunits the activated alpha subunit can bind to and activate adenylate cyclase activated adenylate cyclase converts ATP to cyclic a MP the g-protein alpha subunit has a gtp s activity and soon Cleaves gtp to form GDP thereby in activating itself and adenylate cyclase the g protein complex reforms and only a small amount of cyclic a MP hadn't made the a one peptide
of cholera toxin is an enzyme that uses a molecule of NAD+ which is made up of a nicotinamide group a ribose sugar and an ADP molecule and add to the ADP ribose to the g-protein alpha subunit this is an adp ribosylation reaction and it prevents the g protein from cleaving gtp into gdp and inorganic phosphate adenylate cyclase remains activated for a long period increasing the levels of cyclic GMP tremendously elevated levels of cyclic ANP stimulate a host enzyme called protein kinase a the catalytic subunits of the enzyme activate various ion transport channels including one called
CFTR which stands for cystic fibrosis transmembrane conductance regulator CFTR exports chloride ions as chloride and other ions leave the cell water leaves as well in an attempt to equilibrate the osmolarity ultimately the blood stream provides the water chloride and other ions that enter the intestinal lumen the influx of fluid results in diarrhea although devastating to the human diarrhea benefits the bacterium in large part by helping disseminate the species in the environment you