aliphatic amino acids are nonpolar and therefore hydrophobic a simple mnemonic to remember the six aliphatic amino acids is glaciers in Alaska valiantly locate isolated prowlers glycine alanine valine leucine isoleucine prolene it is key to remember them in this order glycine alanine valine leucine isoleucine prolene because this makes it relatively easy to remember the structures let's start with glycine this is the simplest amino acid and the only non chiral one a chiral carbon is attached to four different groups glycine is attached to two hydrogen's since it's our group the group that changes among the 20 amino
acids is a hydrogen alanine has a methyl group instead of a hydrogen from here on in the amino acids look a bit like a germinating seed with the r group as the growing root in valine the root splits into two reutlitz both methyls in leucine the root grows longer and here the root analogy stops you can think of isoleucine as a conformational isomers of leucine prolene is the only ring structure but noted is not aromatic hence it is not one of the three aromatic amino acids and now for a brief review glacier's glycine in Alaska
alanine valine Li baleen locate leucine isolated isoleucine prowlers prolene aromatic amino acids fulfilled the three rules of aromaticity they are rings they must be planar usually sp2 hybridized with the unhybridized p orbitals overlapping to form a continuous ring of planar orbitals the ring must follow huckel's rule having 4n plus 2 electrons in its system of conjugated p orbital clouds or n is an integer here is a way to remember the three aromatic amino acids remember how glaciers nolasco valiantly locate isolated prowlers well why do people still go there it's because their aroma of fine pine
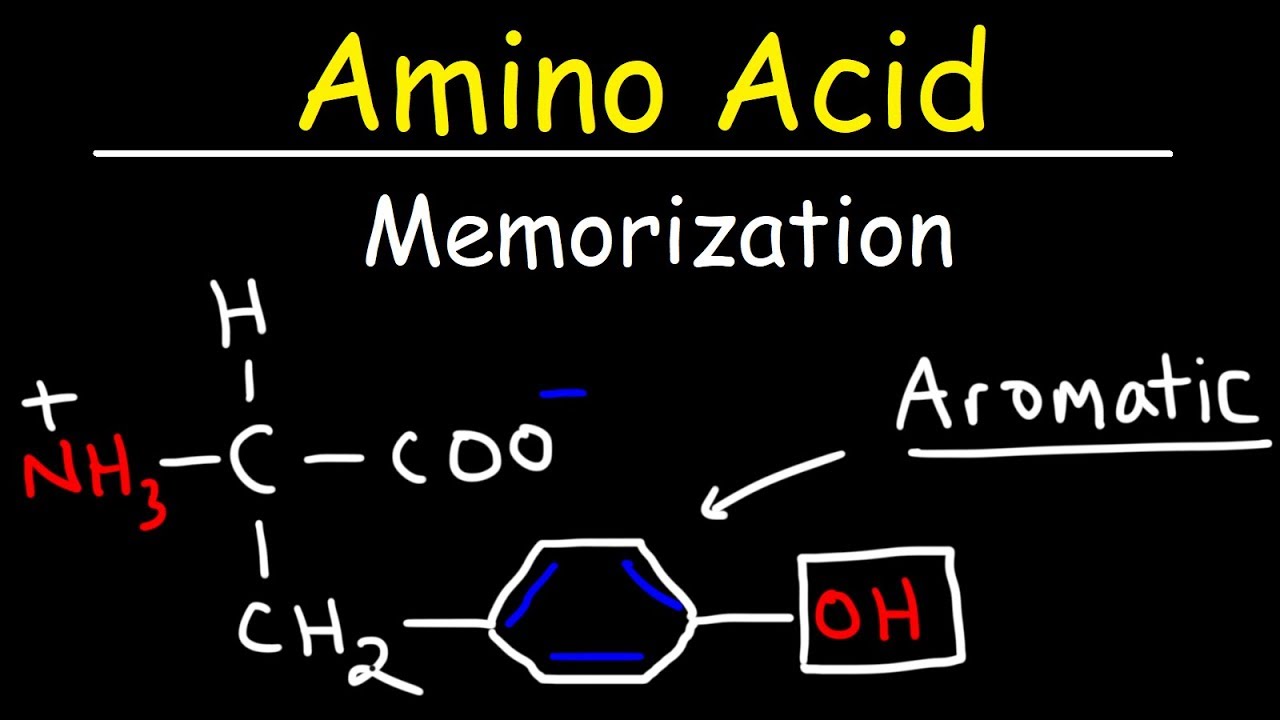
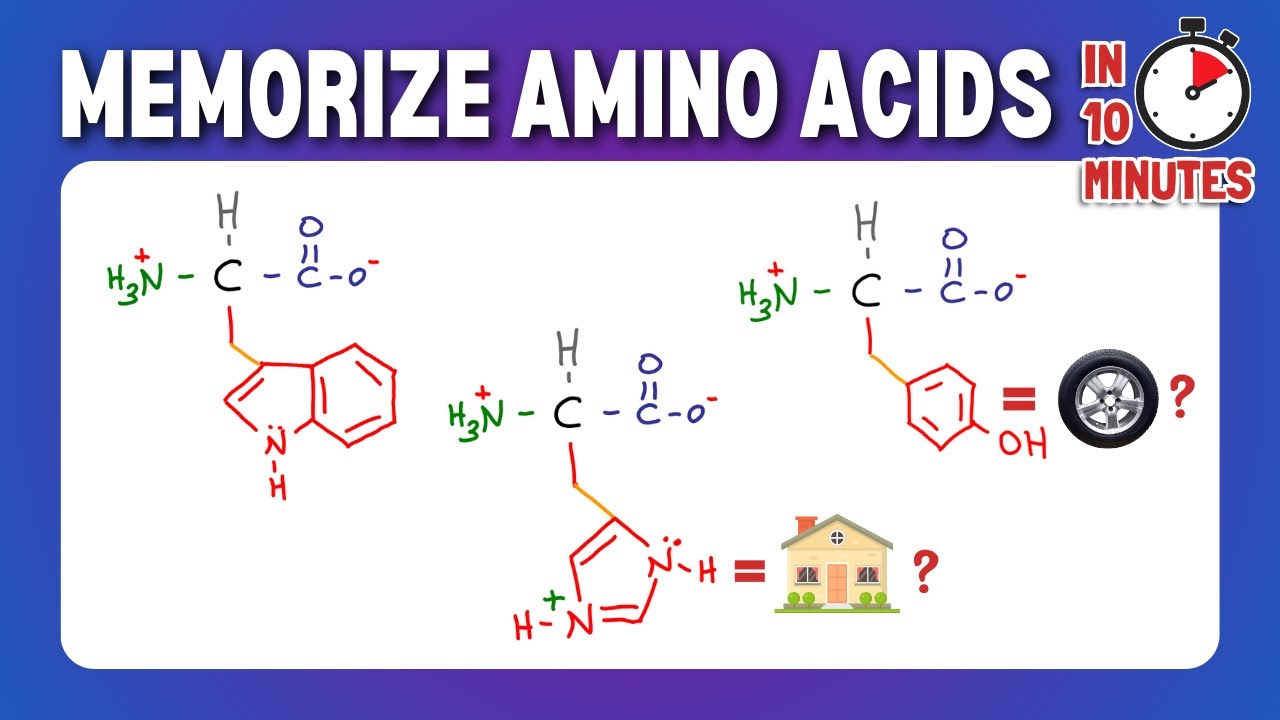
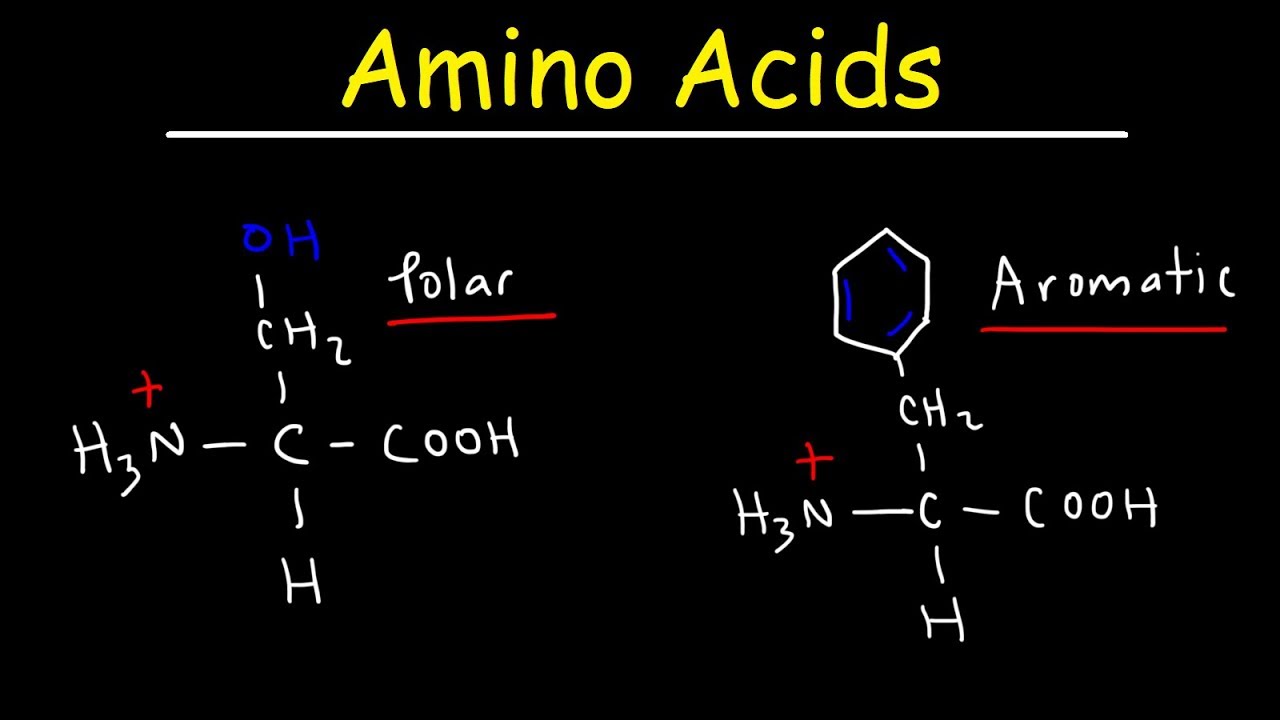
and yellow timber are worth the trip the aromatic compounds include phenylalanine tyrosine and tryptophan phenylalanine has a benzyl attached to it tyrosine is almost identical but has a hydroxyl group tryptophan has two aromatic rings one a five membered ring with a nitrogen and one a six membered ring the rings sort of look like a W if you think about it unlike with the aliphatic amino acids or the single letter abbreviations are all the first letter of the amino acid names the aromatic amino acids have weird single letter abbreviations you can remember them this way pine
trees phenylalanine have fine needles F timber tyrosine is yellow Y and the trip is worth it w-2 of the amino acids are alcohols while alcohol is a serious threat memorizing these amino acids will be a serious treat serine and threonine have very easy abbreviations the one letter abbreviations are just the first letter of each amino acid while the three-letter abbreviations are just the first three letters of each amino acid what a serious treat here is the structure of serine as you can see the structure of threonine just has an extra methyl group attached to the
carbon making it chiral three of their amino acids are bases here's a way to remember them basically as lost kid always returned the basic amino acids are histidine lysine and arginine and the one letter abbreviations were the last two are not the first letter of each while histidine is abbreviated as H lysine is abbreviated as K and arginine is abbreviated as R luckily the three-letter abbreviations are the first three letters of each amino acid let's look at histidine well not much we can do here but just memorize it but here is the cool bit once
we memorize histidine it is very easy to remember the other two basic amino acids we can change histidine to lysine thus first remove the two double bonds and the bond between the N with the lone pair and the carbon on its top left next remove the NH change the end with a lone pair to an nh3 this is lysine now let's change lysine to arginine switch the ch2 and nh3 plus erase the three plus and the H to dry in two NH 2's attached to the carbon since carbon makes four bonds one of the nitrogen's
will have a double bond to a carbon as a result it will have a positive charge and that's arginine now let's compare the three side-by-side his histidine lost lysine kid K always arginine returned R there are two sulfur containing amino acids and they are special because each can link to other sulfur-containing amino acids through oxidation of their salt hydral bonds to form sulfur sulfur bonds but that is outside the scope of this video the easy way to remember the sulfur-containing amino acids is to methodically check the path for sulfur methionine and cysteine are the two
sulfur containing amino acids and fortunately the abbreviations are just the first letter of each and the first three letters of each here is methionine to get cysteine you check between two rocks the methyls get rid of them and all you're left with is sulfur desperately trying to hide behind the one rock left there are two acid amino acids as Peter digested the glue his stomach became acidic the two acid amino acids are aspartate and glutamate these are actually two excitatory neurotransmitters of the brain in other words they make postsynaptic neurons more likely to fire anyway
the three-letter abbreviations are just the first three letters of each but the one letter abbreviations are not here is the structure of aspartate just blunt memorization unfortunately glutamate is easy to remember though you just glue on another carbon tada the amede amino acids are so named because they have an immediate amana is amid this neatly stacked asparagus is a gluttonous quail the to a meat amino acids are asparagine and glutamine the one letter abbreviation for asparagine is N and for glutamine it's q since the three-letter abbreviations can't be the same as for the acid amino
acids they are a SN for asparagine and GL n for glutamine attention be careful and don't confuse the acid amino acids with the Amidah meno acids you can remember that the amide amino acids are the ones ending in e because amines and amines are often confused as the two functional groups with nitrogen's here is asparagine structure unfortunately blunt memorization is needed here but it is easy to remember glutamine because what happens to those who are gluttonous they get bigger and that is exactly what happens to glutamine it gets an extra carbon now I wonder if
any of you have noticed a cool pattern between the acid and Amade amino acids well aspartate and asparagine both have two carbons and there are groups and glutamate and glutamine both have 3 carbons and there are groups the only difference between the pair of acids and the pair of a means is the following just the last point about amino acid properties acid and basic amino acids are charged amino acids all aliphatic amino acids as well as methionine a sulfur containing amino acid and phenylalanine and tryptophan which are aromatic amino acids are nonpolar amino acids all
alcohol amino acids and a mead amino acids as well as cysteine which is a sulfur containing amino acid and tyrosine which is an aromatic amino acid or polar amino acids if you liked this video please like and subscribe it would help me make more videos and make sure to comment with any topics you'd like me to cover in future videos also it would be really nice if you could support me on patreon thank you