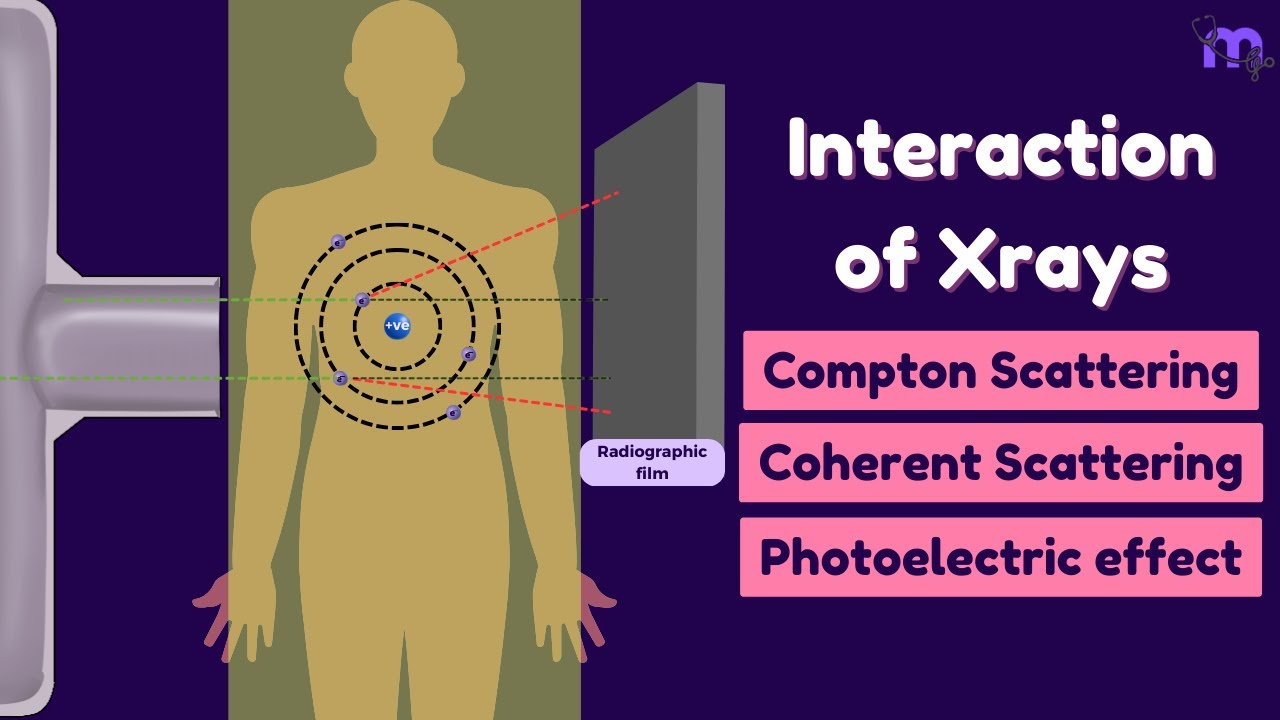
There are two major ways in which diagnostic x-rays interact with tissue. The first one is the photoelectric effect, and the second one is the Compton effect. The photoelectric effect is the process of a photon being absorbed by an inner shell electron.
All the photon energy is transferred to the electron so that the photon disappears. An energetic photoelectron is then ejected and this leaves a vacancy. To stabilize the atom, an outer shell electron fills the vacancy in the inner shell.
Each electron shell has a different energy level, with those closest to the nucleus being lower in energy than those farther from the nucleus. In order to move between shells, an electron must absorb or release an amount of energy. This is why if an electron drops down to a lower-energy shell, it will release energy in the form of x-rays.
This is also known as characteristic radiation. In Compton scattering, the photon transfers only a part of its energy to an outer shell electron. The photon knocks the electron out, but unlike in the photoelectric effect, the scattered photon still has a fraction of the energy of the incoming photon and can still travel through the patient.
These scattered photons may then proceed to generate further interactions both inside and outside the patient. This is why Compton scattering not only decreases the image quality but also increases the patiënt dose and is the predominant source of background radiation. So In the photoelectric effect, a single electron absorbs all the energy of an incident photon, while in the Compton effect, the incident photon only transfers a portion of its energy to an electron.
The likelihood of photoelectric absorption increases with the atomic number and decreases with increasing photon energy. Therefore, as the atomic number increases, the photoelectric absorption becomes more pronounced. This is why materials such as lead (Z=82) are widely used for shielding gamma and x-rays.
The photoelectric effect is dominant at the lower photon energies, the Compton effect takes over at medium energies and pair production at higher energies. Soft tissue is made up of low atomic number elements such as hydrogen, oxygen and carbon while bone is made up of higher atomic number elements such as calcium and phosphorus. Therefore photoelectric absorption is more likely to occur in bone than in tissue.
This effect is important in providing contrast. If all x-rays were to pass through the patient, the image would be uniformly dark and if all the x-rays were to be absorbed, the image would be uniformly white. Another important concept is that of the ‘k-edge’.
The k-edge represents the energy needed to eject a K-shell electron. You’ll see an abrupt increase in the photoelectric absorption of x-ray photons at the energy level just beyond the binding energy of the k-shell electrons. In other words, at energies just below the k-edge the photons don’t have enough energy to free the k-shell electrons.
As the energy increases to just over the required energy, the probability of the photon being attenuated by a photoelectric reaction significantly increases. Iodine and gadolinium-based compounds are the preferred contrast agents because they have K-edges around 30-50 keV, which is right in the middle of the x-ray beam spectrum. This is why tissues with even a small amount of iodine will have a much stronger x-ray stopping power than those without iodine.
Also, they both have high atomic numbers and the higher the atomic number, the more photoelectric absorption that will occur. So in summary, In the practice of nuclear medicine, where gamma rays between 50 and 550 kilo-electron volt are used, Compton scattering is the dominant type of interaction in material with lower atomic numbers, such as human tissue and air. The photoelectric effect is the dominant type of interaction in materials with higher atomic numbers, such as contrast agents, bone and lead.




![X-ray Interactions Photoelectric and Compton Scattering for Radiologic Technologists [Rad Physics]](https://img.youtube.com/vi/OaQHP4jD_hE/maxresdefault.jpg)