the discovery of radioactivity by Henry Beckel in 1896 marked the beginning of a new era in physics and chemistry many lives were altered by the discovery in the coming years and among the first affected was an upand cominging New Zealand research student by the name of Ernest Rutherford at the time Rutherford was studying at the Cavendish laboratory at Cambridge under JJ Thompson one year before Thompson published his findings on the discovery of the electric at Cavendish Rutherford was busy studying radio waves and at that time had held the record for the longest distance over which
these Wireless Waves were detected however upon hearing the news of this new form of radiation Rutherford quickly turned his attention towards uncovering its nature from 1896 until his departure from Cavendish 2 years later Rutherford devoted his time to the newly budding field and quickly found it to be very rewarding ing Rutherford's first successful experiment in radiation was actually rather simple and was set up as follows a sliver of uranium was placed in between two electrical plates one plate was charged by a battery while the other remained uncharged and was attached to what is called an
electrometer which is a device used to measure electrical potential difference and electrical current the radiation of uranium ionizes the gases around it creating positively and negatively charged gas ions the negatively charged ions move towards the charged plate and the positively charged ions move away from the charged plate this generates a current in between the plates which is picked up by the uncharged plate and sensed by the electrometer the more radiation present the larger the current measured by the electrometer for each time he made measurements he added on top of the uranium a sheet of Dutch
metal which is a form of brass composed of approximately 85% copper and 15% zinc these sheets were extremely thin around 0.00008 CM thick and were added to block some of the radiation from the uranium into the air between the plates Rutherford then made measurements of the rate of leak per minute for each sheet of Dutch metal he added over top the uranium the result result he got from this were somewhat surprising in this graph you can see how the decrease in radiation is not linear but rather exponential if all radiation from the uranium was the
same it would make sense that the radiation would decrease the same amount each time a new layer of Dutch metal was added on top of the uranium the unexpected results here pushed rufer to experiment further this time adding even thicker sheets to possibly uncover a SE separate layer of radiation Rutherford switched to 00005 CM thick aluminum sheets this expectedly did a much better job at blocking the radiation but it only did so for the first few layers added after adding four layers of aluminum foil the radiation level would remain the same for each new layer
of foil he'd add over top of the uranium it would take 12 sheets before Rutherford would see any more decrease in radiation which ultimately led him to conclude that there were two different types of radiation from the uranium the first type which he called Alpha rays were the easiest to block and the second type which he called beta Rays required much more strength to block Rutherford then studied beta Rays further by continuing to add foil sheets and he recorded a data set that showed the decrease in radiation without the presence of the alpha Rays this
chart was much more linear than the previous data that contained both types of rays although not entirely perfect ruer concluded his experiments there ruling that there was possibly a third type of radiation emitted from the uranium but that it must have been either so small or so penetrating that his experiment could not detect it this third type would be confirmed a few years later in 1903 and would be labeled as gamma rays the most penetrating form of radiation after his discovery of Alpha and beta particles Rutherford moved to Mill University in Montreal Canada and teamed
up with a mill chemist Frederick Saudi to further work on radioactivity during the years of 1902 and 1903 they made another significant breakthrough their experiment used a similar setup to that of the one that detected Alpha and beta rays but instead of measuring uranium it measured an isotope of radon called thoron while while studying thoron they noticed an interesting Trend that after identical increments of time the amount of radiation detected by the thoron at a specific step would be half of what it was at the previous Step in Time as this was happening they noticed
a seemingly spontaneous buildup of other elements most notably LED in their setup from these discoveries They concluded that over time radioactive elements must Decay into other elements and introduced the concept of half- lives to the scientific Community Rutherford and Saudi both exploded into scientific Fame after these discoveries sparking debate and controversy over the nature of these results and their implications almost immediately after the discovery of radioactive decay a quest to calculate the true age of the earth based on half lives was ignited and would make significant leaps in the coming decades both Rutherford and Saudi
were just getting started too Rutherford won the Nobel Prize in chemistry in 1908 for the discovery of alpha particles beta particles radioactive decay and half- lives and would go on to make many more Monumental experimental discoveries that I'll save for future videos Saudi although not being credited in the 1908 prize would later get a Nobel Prize of his own in 1921 for discovering Isotopes another topic I will also C in the future Rutherford and saudi's pioneering work in these areas set the wheels in motion for a new era of nuclear physics opening up an entirely
new can of worms and just scratching the surface on what was possible with the elements based on the newly discovered phenomenon of radioactivity if you enjoyed this video please consider liking and subscribing click here if you want to see more scientific progress made during this time period thank you for watching and I will see you in the next video










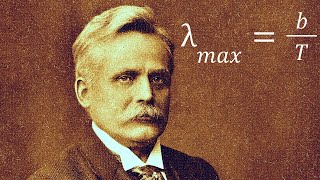
![Alpha Beta Gamma: History of Radioactivity [CC]](https://img.youtube.com/vi/-G_CtD2UnAc/mqdefault.jpg)




