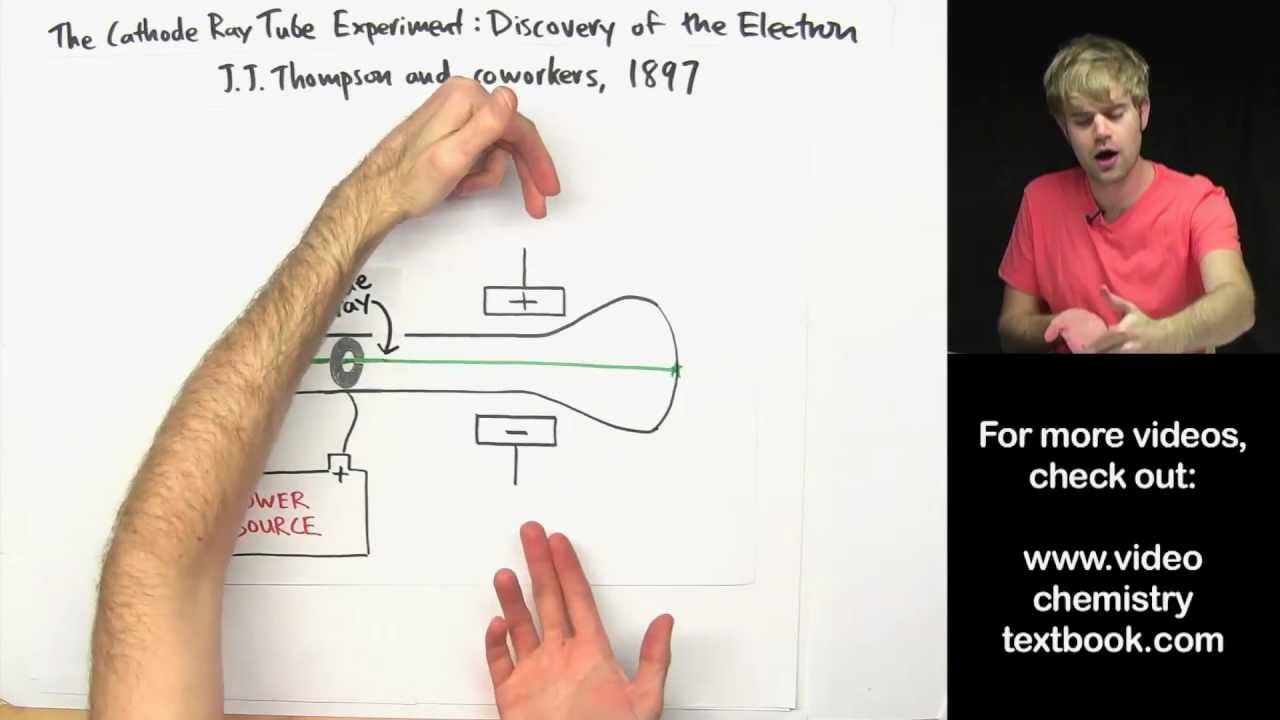
The Rutherford atomic model is the description of the atom created by the British physicist Ernest Rutherford (1871-1937) when he discovered the atomic nucleus in 1911, thanks to the famous dispersion experiments that bear his name. The idea of the atom as the smallest component of matter was an intellectual creation born in Ancient Greece, around the year 300 BC. Like so many other Greek concepts, the concept of the atom is developed based on logic and argumentation, but not on experimentation.
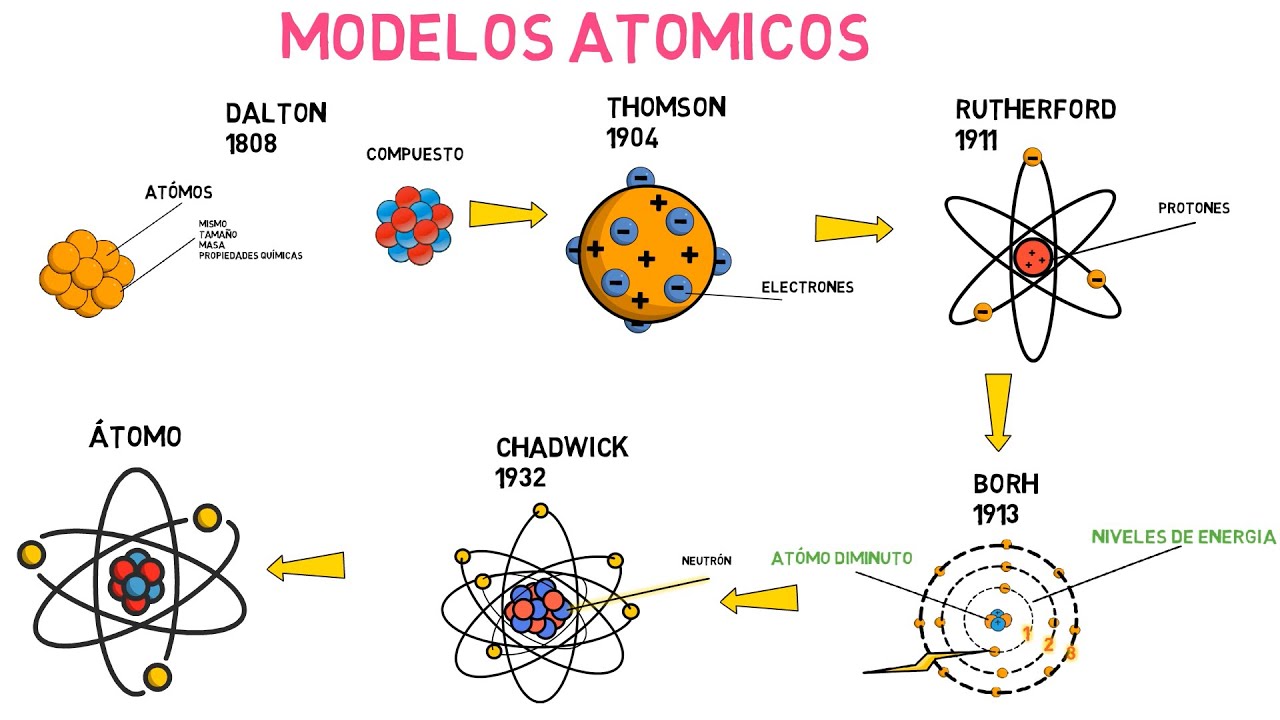
The ideas of the Greek philosophers about the structure of matter held true for hundreds of years, until an English chemist and schoolteacher named John Dalton (1776-1844) published the results of his experiments in 1808. Dalton agreed in which elements are made up of extremely small particles called atoms. But he went further by stating that all atoms of the same element are equal, they have the same size, the same mass and the same chemical properties, which makes them remain unchanged during a chemical reaction.
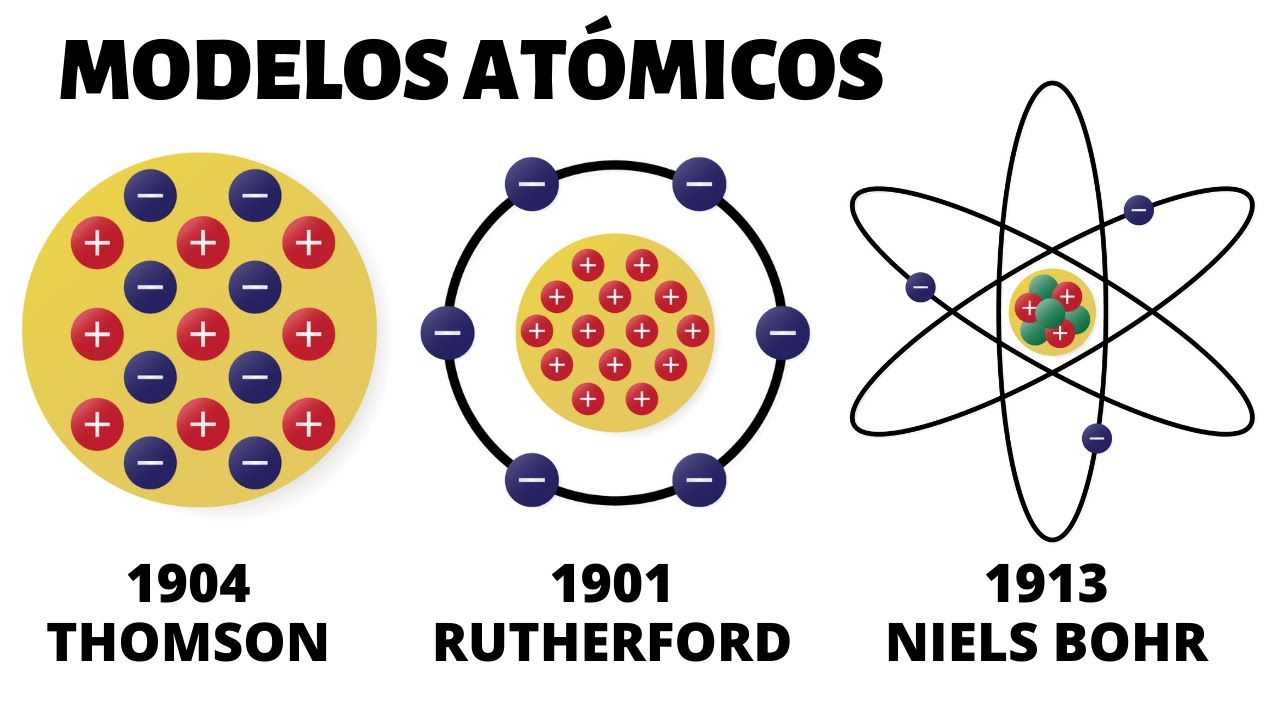
This is the first scientifically based atomic model . Like the Greeks, Dalton still regarded the atom as indivisible, therefore lacking in structure. But Rutherford's new model was very different from Thompson's.
These were his postulates: The atom contains a positively charged nucleus, which despite being very small, contains almost all the mass of the atom. Electrons orbit the atomic nucleus at great distances and in circular or elliptical orbits. The net charge of the atom is zero, since the charges of the electrons compensate for the positive charge present in the nucleus.
Rutherford's calculations pointed to a nucleus with a spherical shape and a radius as small as 10 raised to minus 15 meters, the value of the atomic radius being about 100,000 times greater, since the nuclei are comparatively far apart from each other: of the order of 10 raised at least 10 meters. This explains why most of the alpha particles passed through the sheet with no problem or only a very small deflection in Rutherford's experiments. Seen at the scale of everyday objects, the Rutherford atom would be composed of a nucleus the size of a baseball, while the atomic radius would be about 8 km.
Therefore, the atom can be considered almost entirely as empty space. Because of its resemblance to a miniature solar system, it became known as the "planetary model of the atom. " The electrostatic force of attraction between the nucleus and electrons would be analogous to the gravitational attraction between the sun and the planets.
Limitations However, in Rutherford's model there were certain inconsistencies with respect to some observed facts: If the idea that the electron orbits around the nucleus is accepted, it happens that the electron should continuously emit radiation until it collides with the nucleus, with the consequent destruction of the atom in well under a second. This, fortunately, is not what actually happens. In addition, on certain occasions the atom emits certain frequencies of electromagnetic radiation when there are transitions between a state of higher energy to one with lower energy, and only those frequencies, not others.
How to explain the fact that energy is quantized? Despite these limitations, since today there are much more sophisticated models and according to the observed facts, Rutherford's atomic model is still useful for the student to have a first successful approach to the atom and its constituent particles. In this model of the atom, the neutron does not appear, another constituent of the nucleus, which was not discovered until 1932.
Shortly after Rutherford proposed his planetary model, in 1913 the Danish physicist Niels Bohr would modify it to explain why the atom does not is destroyed and we are still here to tell this story. Let us now look at Rutherford's scattering experiments that led to the discovery of the atomic nucleus and the proton. By 1898 Rutherford had identified two types of radiation from uranium, which he called alpha and beta.
Natural radioactivity had already been discovered by Marie Curie in 1896. Alpha particles have a positive charge and are simply helium nuclei, but at that time the concept of a nucleus was not yet known. Rutherford was about to find out.
One of the experiments that Rutherford carried out in 1911 at the University of Manchester, with the assistance of Hans Geiger, consisted of bombarding a thin sheet of gold with alpha particles, whose charge is positive. Around the gold foil he placed a fluorescent screen that allowed them to visualize the effects of the bombardment. Studying the impacts on the fluorescent screen, Rutherford and his assistants observed that: A very high percentage of the alpha particles passed through the sheet without perceptible deviation.
Some were deflected at fairly steep angles AND very few bounced completely back that responsible had a size much smaller than that of alpha particles. Rutherford himself said about it that it was ". .
. as if you fired a 15-inch naval shell at a sheet of paper and the shell bounced back and hit you. " This definitely could not be explained by Thompson's model.
Analyzing his results from the classical point of view, Rutherford had discovered the existence of the atomic nucleus, where the positive charge of the atom was concentrated, which gave it its neutrality. Rutherford continued with his dispersion experiments. By 1918 the new target for alpha particles was gaseous nitrogen atoms.
In this way he detected hydrogen nuclei and knew immediately that the only place these nuclei could come from was from nitrogen itself. How was it possible that hydrogen nuclei were part of nitrogen? Rutherford then suggested that the nucleus of hydrogen, an element already assigned atomic number 1, must be a fundamental particle.
He called it proton, a Greek word for first. Thus, the discoveries of the atomic nucleus and the proton are due to this brilliant New Zealander.