[Music] hello and welcome back to immunology the war is over episode two where we'll be getting to know naive t helper cells and the t helper subset so the naive t cell graduates from the thymus and is released into the circulation from there it will travel to the lymph nodes or spleen and set up camp now of course we do have t cells whizzing around in our circulation moving between these sites all the time but you can expect in any one moment for the majority of your t cells to be chilling out inside your lymphoid
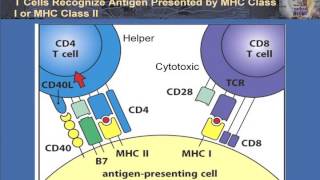
organs now we know that there are different types of t cell but the first t cell we're going to focus on is the t helper cell or the cd4 t cell remember that this t cell recognizes antigens presented by mhc2 molecules on antigen presenting cells such as dendritic cells b cells and macrophages and remember that because each t cell receptor is unique it will match to a very specific antigen and so if a dendritic cell has an antigen to present how does it find the right t cell that matches that specific antigen amongst all the
thousands of t cells in the lymph node if ever there was a needle in a haystack this is it so how on earth does the dendritic cell do this this is pretty amazing the dendritic cell takes the antigen and presents it within a nearby lymph node t cells are constantly moving around between the circulation and the lymph nodes but when they come across a dendritic cell they will stop to take a look and by take a look i mean t cells will literally crawl all over this dendritic cell it is touchy-feely and it is an
absolute free-for-all you could think of this as a sort of antigen presentation cuddle party between the dendritic cell and numerous t cells and this cuddle party continues until a t cell comes along with the t cell receptor which specifically matches the antigen being presented and when this match occurs the dendritic cell and t cell become exclusively connected and this sparks off a whole chain of events that i'm going to show you now the chain of events is known as signal 1 2 and 3. signal 1 is simply the initial binding of the t cell receptor
to the antigen mhc complex it's that moment in the cuddle party when the dendritic cell finally finds the t cell receptor it's been looking for and now instead of just touching these cells are interlacing hands with each other once a match occurs the t cell and dendritic cell acknowledge that it wasn't easy to find each other and so they place adhesion molecules on their surface in order to connect and hold on tight this is also known as a stop signal and so that's signal one recognition of the mhc antigen complex by the t cell receptor
which in turn generates a stop signal so that the t cell and the antigen presenting cell are now locked in position ready for more advanced communication which brings us to signal 2 co-stimulation and this step is absolutely crucial to invoking an immune response without it literally nothing will happen and this step is carried out by co-stimulatory molecules on the surface of the t cell and antigen presenting cell now it's worth pointing out that co-stimulation is just that it's a two-way conversation when these surface molecules interact with each other there will be changes inside the t
cell but also changes inside the antigen presenting cell and another thing that's worth mentioning is that although they are termed co-stimulatory molecules some of them have an important inhibitory function as well because just as much as we want to spark off a t cell response we also want to make sure that the response is not excessive and so these co-stimulatory molecules are designed to keep that t-cell response in check now there are a number of co-stimulatory molecules and we're going to be discussing them in great detail in the next episode and i've given them their
own episode because they lend themselves so well to exam questions but also there are numerous drugs in clinical practice which target these molecules so in the next episode we'll be diving into co-stimulatory molecules and the drugs which block them but for now we're going to stay focused on the broader aspects of t cell activation so for now what's important to know is that the end result of co-stimulation is the production of interlooking 2 by t cells now interlocking 2 is to t cells what caffeinated beverages are to me and to look into is t cell
activation fuel so just in the way that i have to go and put the kettle on and make myself a coffee t-cells have to make their own fuel as well so they make interlocking two it leaves the cell and then they use interleukin-2 to fuel themselves using interleukin-2 receptors also known as cd25 so if signal 2 happens successfully t cells will produce interlooking 2 creating their own activation fuel for what happens next if signal 2 does not go ahead interleukin 2 is not produced and instead the t cell becomes fully devoid of function and this
is known as t cell energy again without caffeinated beverages i can fully relate to this feeling of energy within the t cell already today i think i must be on my third cup of coffee and it's approximately quarter past 10 in the morning i should probably switch to decaf soon anyway the point is interleukin 2 is t cell activation fuel and interleukin 2 is the end product of a successful signal 2. now isn't it convenient that for signal 2 to be effective it requires interleukin-2 if that does not soothe your neurons i don't know what
will interleukin 2 is essential for signal 3 which is t cell proliferation this is when t cells do what they do best multiply into an army and go forth in their immune response activities signal 3 is drinking the interleukin-2 coffee which boosts the cell cycle and causes that t cell to multiply into an army of t cells now in order for t cells to proliferate they will need to enter the cell cycle and for this they're going to need some snacks so before t cells divide they place channels and transporters onto their surface so they
can take in more glucose and amino acids to provide sustenance for cell division okay so we got the coffee and the snacks signal 3 is well underway but to help t cells enter the cell cycle there are signaling molecules which lend a hand and conveniently these signaling molecules have been given names such as cyclin and cyclin-dependent kinases presumably because of their cell cycle promoting abilities and these cyclins are themselves promoted by various pathways one famous pathway which promotes cyclins involves the mtor complex so this emptor complex is known to promote entry into the cell cycle
and this might sound a little familiar you may have heard of the mtor inhibitors ceromis and everolimus these drugs limit entry into the cell cycle in immune cells but also in other tissues around the body so we use these drugs in clinical practice as both immunosuppressants and tumor suppressors so mtor and other molecules contribute to t cells entering the cell cycle and proliferating into a t-cell army but again we wouldn't want t-cell proliferation to go unchecked and what's super cool is that mother nature installed some cell cycle timers inside the t-cells that will blow your
mind one of these little timers is something known as mic when t cells are activated mic a signaling molecule is generated and wholeheartedly encourages the cell cycle to occur but mike has a short half-life and so when mike runs out the t-cell will stop dividing super clever right okay so these activated t-cells will proliferate into a t-cell army now everything i've mentioned so far about t-cell activation could roughly be applied to both naive cd4 and cd8-t cells so although cd8 t cells require antigens to be presented using mhc-1 and cd4 t cells require antigens to
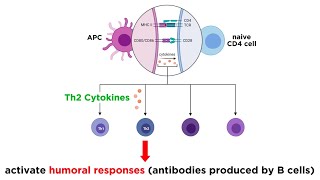
be presented using mhc2 their activation pathways are pretty similar but there is another key difference between cd8 and cd4 t cells activated cd8 t cells will without question become an army of cytotoxic t cells but cd4 t cells or t helper cells will differentiate into various t cell subsets with varied functions depending on the immune response that is required so what are these t helper subsets what do they do and how are they determined alti helper cells look alike and when they differentiate into their various subsets their appearance does not change what sets them apart
from each other is not what they look like but the cytokines they produce and different cytokine profiles will orchestrate a different immune response and similarly nurturing of t helper cells to differentiate them into a particular subset is also encouraged by particular cytokines i'll show you what i mean first up th1 cells chances are this is the t helper subset that you are most familiar with th1 is in the business of helping us fight viruses and other intracellular pathogens and its cytokine profile includes interferon gamma interleukin 2 and tnf alpha and the th1 lineage is promoted
by interleukin-12 and interestingly a key source of interleukin-12 is antigen presenting cells so right there next to the activated t cell is an antigen presenting cell capable of producing interleukin-12 and turning this basic naive t helper cell into the th-1 subtype interlocking 12 promotes the expression of a transcription factor known as t-bet also known as t-box binding transcription factor which promotes interferon gamma expression and the cytochrome profile necessary for the th1 phenotype the th2 subset has the cytokine profile interleukin-4 interleukin-5 and interleukin 13. this is helpful in orchestrating the fight against parasites in the extracellular
space but it also shows up in our clinical practice within allergic conditions such as asthma and the cytokine which promotes a th2 response is interleukin-4 interleukin-4 promotes the transcription factor gata3 which generates this characteristic cytokine profile and you can see how th1 cells are designed for intracellular pathogens whilst th2 responses are directed against extracellular parasites and so for a given organism you only need one of these responses to be activated and it's super clever that the transcription factors needed to create a th1 response will actively inhibit the production of th2 cells and vice versa so
you never get these two responses happening at the same time okay so th1 and th2 you're likely familiar with in some capacity but there are other subsets which have only recently begun to step into the spotlight and these are t helper follicular helpers t regulatory cells and th 17 cells let's take a look at these in turn t helper follicular helpers are the t cells which help b cells now we will be coming back to this in more detail a bit later in the series but just know that this t helper subset promotes b cell
survival proliferation antibody class switching as well as the differentiation of b cells into plasma cells which produce high affinity antibodies and these t helper follicular helpers have a characteristic cytokine profile which includes interleukin-21 and is promoted by transcription factor bcl-6 and the cytokine which promotes this t-cell subset forming is interleukin-21 you've probably noticed by now that there is an emerging theme where each t-cell subset appears to produce cytokines which reinforces its own development so t-cell subsets once initiated sort of back themselves up with positive feedback and become their own cheer squad next up are t-regulatory
cells or t-rex these are absolute champions for our health and well-being they are all about keeping the peace and making sure that the other t-cells don't get carried away they can literally suppress the function of effector t cells as well as other immune cells and this is a necessary maneuver after all we want the immune system to be effective in eradicating organisms but we also want to avoid excessive damage to our own healthy tissue and we also don't want autoimmune disease and t-regs are crucial in protecting us against the pathological potential of our own immune
system and in the setting of organ transplantation t-regs are thought to be helpful in establishing immune tolerance which means that over time we can lower the immunosuppression without necessarily having that result in rejection a proportion of tear eggs originate in the thymus and are termed natural tear eggs or n-t-regs but the t-regs which develop from naive t-helper cells following antigen presentation in the periphery are known as induced t-rex or i-t-rex in order to induce a t-reg we need tgf beta which promotes the transcription factor fox p3 this in turn results in the production of the
characteristic cytokines interleukin 10 and tgf beta these anti-inflammatory cytokines are all about bringing the calm and the healing and last but not least we arrive at th17 cells which secrete interleukin-17a and interleukin-17f which help in the defense against various extracellular pathogens now th17 cells are slightly more complicated in terms of how they develop there are various cytokines involved at various stages of this process at induction differentiation and maintenance so to transform a naive cd4 t cell into a th17 cell tgf beta combines with interlooking sex now remember that tgf beta could on its own promote
the development of t regs but when combined with interleukin 6 which is produced abundantly by the innate immune system these cytokines work together to induce a completely different transcription factor known as raw gamma t so interleukin sex dictates the balance between t regs and th17 cells but these cytokines alone are not enough to develop a fully fledged th17 cell we need another cytokine interleukin-21 to come along and take things up a notch and now the th17 cell can finally produce its classic cytokines interleukin-17a and f but it's still not over from there another cytokine interleukin
23 comes along and finishes the job making this th17 cell fully fledged and a fully fledged th17 cell secretes not only interleukin-17 but also interleukin-22 which has a further role in our immune defense and interleukin-21 which amplifies th17 production as well as having some other functions around the place now th17 cells seem to be important for fighting extracellular organisms and one way in which we do this is by using interleukin-17 to recruit neutrophils to the area and of course neutrophils are absolute bosses when it comes to fighting bacteria and fungi some of the organisms which
respond well to a th17 response include candida albicans citrobacter rhodensium klebsiella pneumonia and bacteroides species among others so following activation naive cd4 t helper cells will develop into various subsets under the direction of cytokine signaling the cytokine signaling may come from the innate immune response but also adjacent immune cells including antigen presenting cells within the lymph node and of course it's far more complicated than that the t cell's response to any cytokine is affected by the other cytokines around the place by the amount of cytokine receptors on its surface and by the length of time
that a cytokine binds to that receptor and the strength of antigen binding at the t cell receptor as well as co-stimulatory signaling is also thought to contribute to the development of various t-cell subsets and what's even more complicated is that there's thought to be some plasticity between these different t-cell types so you can take one subset influence it with a different set of cytokines and in doing so cause it to morph into a completely different t-helper subset so a th1 cell could become a t-reg a t-reg could become a th17 cell and so on and
so forth so that's a wrap for this episode thank you so much for joining me i hope you've enjoyed watching the journey of what happens when a naive t-cell meets its specific antigen and i hope you join me for episode 3 when we dive into co-stimulatory molecules and the drugs which block them this is sure to be mcq and clinical practice gold i'll see you there [Music] you