The plasma state is one of the fundamental forms in which matter can aggregate, and is the most predominant in the observable Universe. Plasma consists of a hot, glowing and highly ionized gas, to the point where it takes on unique properties that differentiate it from the gaseous state or from any other gas in particular. We see plasma scattered in the stars of the night skies.
As there are an endless number of stars in the Universe, as well as nebulae and other celestial entities, it is considered to be the most important state of matter. On Earth it is considered the fourth state, after liquid, solid and gaseous. The Sun is the closest example where we can appreciate the characteristics of plasma on a massive scale in a natural environment.
On the other hand, natural phenomena occur on Earth in which a momentary appearance of plasma is triggered, such as fire and lightning in storms. Plasma is not only associated with high temperatures (millions of degrees Kelvin), but also with large electrical potentials, incandescent lights, and infinite electrical conductivity. Characteristics of plasma Composition Matter is composed of particles (molecules, atoms, ions, cells, etc.
), which, depending on the effectiveness and the forces with which they are added, establish a solid, liquid or gaseous state. Plasma particles consist of positively charged atoms, better known as cations (+), and electrons (-). In the plasmatic state of matter we do not speak of molecules.
Cations and electrons vibrate at very high frequencies showing a collective behavior and not individual. They cannot separate or move without the entire set of particles being disturbed. This does not happen, for example, with gases, where their atoms or molecules, although they collide with each other, have minimal, negligible interactions.
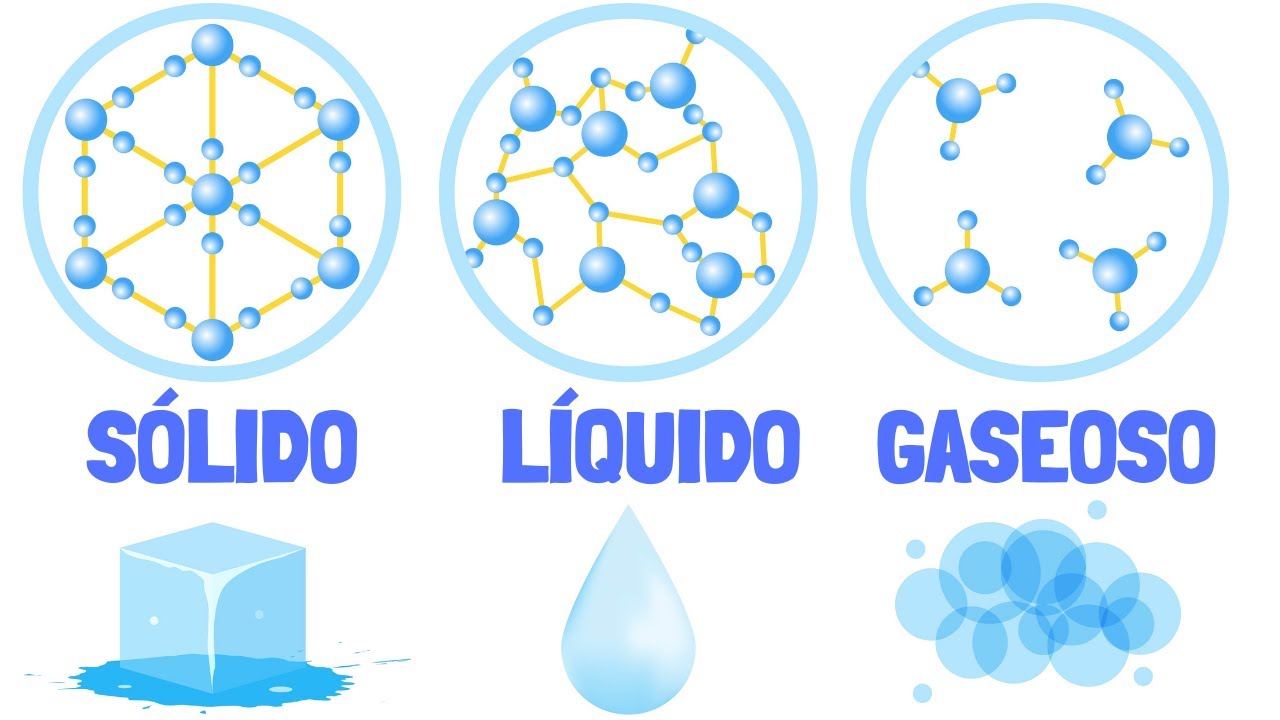
Formation The plasma state is formed, mainly, when a gas is ionized as a result of its exposure to very high temperatures. Let's start with an ice cube first. This is a solid.
If heated, the ice will melt into liquid water. Then, by heating to higher temperatures, the water will begin to boil and escape from the liquid as steam, which is a gas. So far we have the three best known states of matter.
If water vapor is heated to a much higher temperature, under favorable conditions there will come a time when its bonds will break to form free hydrogen and oxygen atoms. The atoms then absorb so much heat that their electrons begin to shoot out into the surroundings. Thus, oxygen and hydrogen cations have been formed.
These cations end up wrapped in a cloud of electrons, aggregated by the action of collectivity and electrostatic attractions. It is then said that a plasma has been obtained from the water. In this case the plasma was formed by the action of thermal energy.
However, highly energetic radiation (gamma rays), as well as large differences in electrical potentials, can also induce their appearance. Quasineutrality Plasma has the characteristic of being quasineutral (almost neutral). This is because the number of electrons excited and released from the atoms tends to be equal to the magnitudes of the positive charges on the cations.
Physical Properties Plasma generally has the appearance of a hot, glowing, highly electrically conductive liquid gas that is responsive or susceptible to electromagnetic fields. In this way plasmas can be controlled or enclosed by manipulating a magnetic field. Types of Plasma There are two types of plasma; partially ionized and fully ionized.
Partially ionized A partially ionized plasma is one in which the atoms have not lost all their electrons, and there may even be neutral atoms. In the calcium example it could be a mixture of Ca2+ cations, Ca atoms, and electrons. This type of plasma is also known as cold plasma.
On the other hand, the plasmas can be contained in containers or insulating media that prevent the diffusion of heat to the surroundings. Totally ionized A totally ionized plasma is one in which its atoms are “naked”, since they have lost all their electrons. Therefore, its cations have high magnitudes of positive charge.
In the case of calcium, this plasma would be composed of Ca20+ cations (calcium nuclei) and many high-energy electrons. This type of plasma is also known as hot plasma. Now let's look at some examples of plasma.
Plasma lamps and neon lights Plasma lamps are artifacts that adorn any bedroom with ghostly lights. However, there are other objects where we can witness the plasma state: in the famous neon lights, whose noble gas content is excited by the passage of an electric current at low pressures. Lightning Lightning that falls from the clouds is a momentary and sudden manifestation of the earth's plasma.
Solar storms Some "plasma particles" are formed in the ionosphere of our planet by the constant bombardment of solar radiation. In the eruptions or tail blows of the Sun we see enormous amounts of plasma. Northern lights At the Earth's poles, another phenomenon related to plasma is observed: the northern lights.
That fire of icy colors reminds us that the very flames of the stoves in our kitchens are another routine example of plasma. Electronic devices Plasma is also part, in smaller proportions, of electronic devices such as televisions and monitors. Welding and science fiction We also see examples of plasma in welding processes, in laser beams, in nuclear explosions, in the light sabers of Star Wars; and in general terms, in any weapon that resembles a cannon of destructive energy.