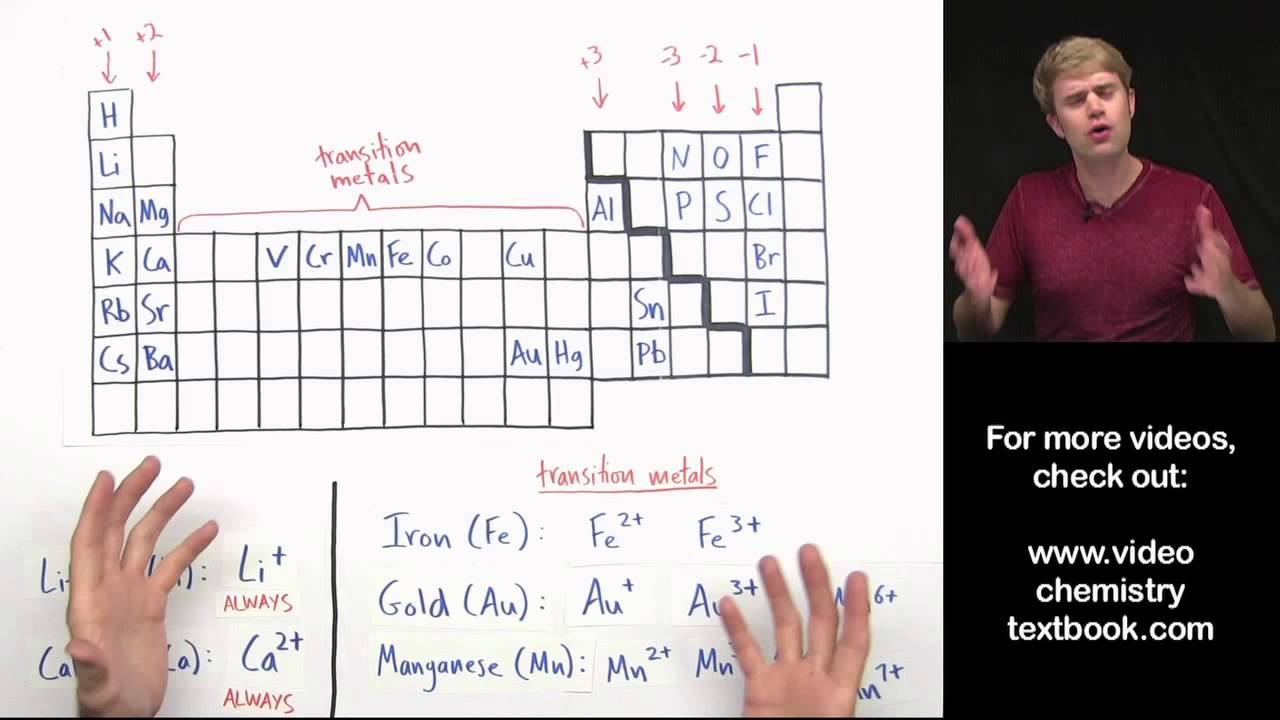
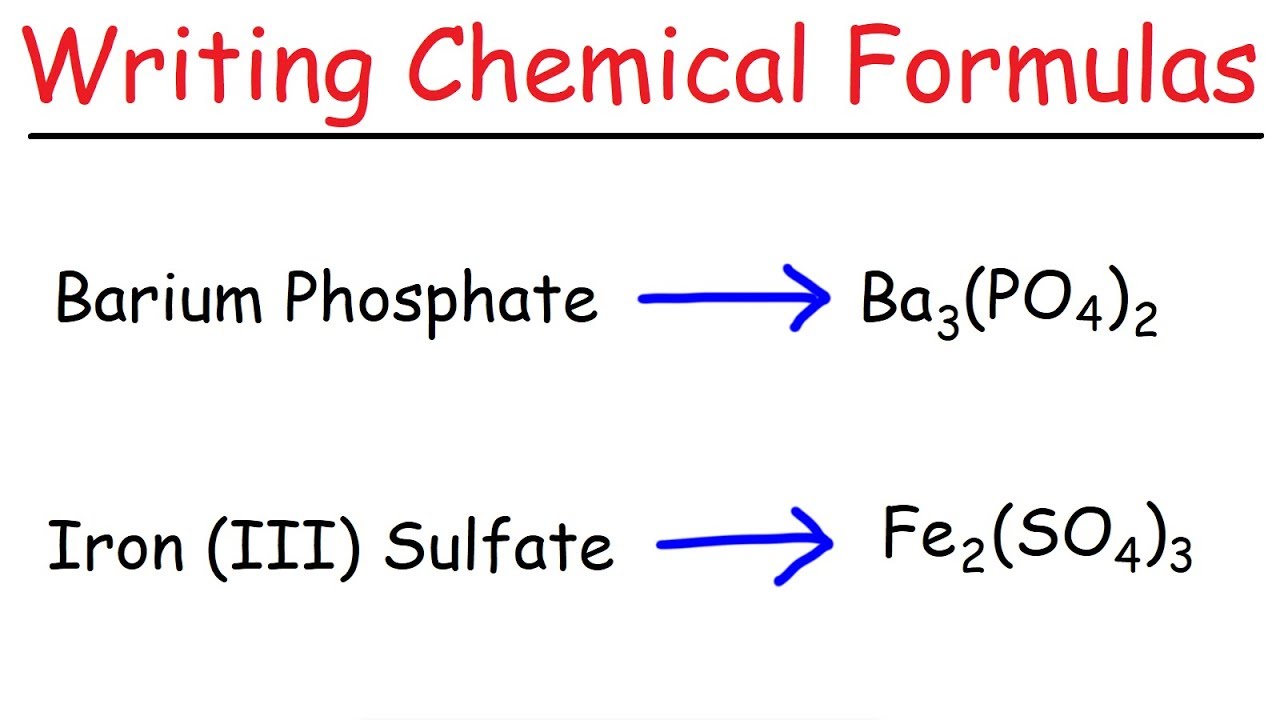
this video is writing ionic formulas with transition metals the transition metals are the elements in this part of the periodic table here and ionic compounds that have transition metals in them usually have Roman numerals in the name like this here so in this video we'll learn how to take a compound that has a transition metal in it and how to write a chemical formula for that now if this stuff is totally new to you you might want to get some background information from two of my videos the first is called writing ionic formula introduction and
the second is called transition metals in ionic compounds if you're all set with the background let's get started here's our first example chromium 2 iodide we want to write a formula for it now when we're writing ionic formulas the first thing we want to do is find out the charges of the ions in the compound and we often do that by looking at where they are on the periodic table so here we have chromium and iodide chromium CR here is a transition metal and transition metal ions can have a variety of charges so you can't
tell just by looking at the periodic table what the ionic charge is so that's where this Roman numeral comes in this Roman numeral here is the number two and it tells us that in this compound chromium has a charge of plus two so that's chromium now we have iodide which is what we call iodine when it has a charge and iodine is in this column right here which means that in an ionic compound its charge is always minus one so now we want to add ions of one or both of the elements so that the
charges balance okay here we have two plus from chromium and we have one minus from iodide so to get this to balance We'll add one more iodide so now we have 2 plus here and two minus here now we write the formula we have cr we have one of them so we don't write anything after that and then we have two of the iodides so I do I 2 and that is the formula now here is a misconception that I want you to be careful of a lot of people see this two in Roman numerals
and they think that this means there should be two chromium in the formula okay that's not true as you know this Roman numeral 2 means that chromium has a two plus charge it's talking about the charge of the ion not the number that you have so be careful of that now let's write a formula for manganese 3 oxide first thing we'll do is figure out the charges on the ions so manganese here is a transition metal don't confuse manganese with magnesium which is not a transition metal so manganese is an element that can make many
different ions you can have MN 2+ 3+ 4 plus 6 6 Plus and 7 plus so the Roman numeral 3 here is telling us that in this compound manganese has a 3+ charge now oxide is what we call oxygen when it has a negative charge oxygen is in this column here which means that in an ionic compound it has a charge of 2 minus so we got mn3 + and O2 minus we want to add add one or more of these elements so that the charges cancel now when we have a three and a two
there's a little strategy here that you might know here's how we do it to get the charges to balance we add another one of these manganes and now we have 3+ and 3+ so we have a total of six plus on this side and to balance that with the oxides we add another for four minus and then we add another for 6 three oxides balance out two manganese 3+ so our formula is MN we got two of those two o one two three of those and as before don't be confused by this Roman numeral three
the three does not refer to the number that should be after manganese in the formula the three refers to the ionic charge on maganese let's do one more lead for nitrate so this Roman numeral IV here is a Roman numeral for four believe it or not it's important to know the Roman numerals in order to deal with transition metals so you want to make sure you learn these at least 1 through 7even which are the most common now to write the formula for lead for nitrate we'll figure out the charge of both of these ions
lead PB here isn't technically a transition metal but lead is like the transition metals because it's able to make multiple ions with different charges so that's why we use the Roman numerals anyway to show what charge lead has in this compound so we got lead 4 PB 4+ and then we got nitrate now nitrate isn't on the periodic table because nitrate is a polyatomic ion it's a big clump of atoms that together have a charge so for this we need to take a look at our list of polyatomic ions how did I know that nitrate
was a polyatomic ion you just got to learn a list of of the polyatomic ions it's a pain but you just kind of got to do it anyway nitrate is right here N3 and that whole clump of atoms has a one minus charge so now we want to add one or both of these to make the charges balance we have four plus from the lead and we have one minus from the nitrate so I add another one of these now I have 2 minus now I have 3 minus and now I have four minus so
my formula is going to be PB I have just one of those and then I have four of these nitrates and since this is a polyatomic ion I'm going to use parentheses because I want to show that I have four of this whole Clump so I put NO3 in parenthesis and then do four outside those parentheses so keep in mind again this four refers to the charge of lead not to the number that should be after it and remember that since this is a polyatomic ion we use these parentheses and then a number outside of
it to show that we have four of this entire group of atoms so that's how we write formulas for compounds that contain transition metals in the next video we'll learn how to go in the other direction how to start with a formula contains a transition metal and then write a name for it this is a little bit more challenging because transition metals don't always have the same charge so we have to do some math and figure out what the charge of the ion is and then we use that to write the name so check out
that video called naming compounds with transition metals