hello everyone welcome to this short tutorial from pathology made simple at tyler pathology the topic which i am discussing today is feroptosis this is a new identity this one is a form of regulated cell death in the earlier videos i had discussed about the other forms of regulated cell that like apoptosis pyroptosis and necrotosis if you are new to this channel you can just go back to my playlist and then have a look at those videos in the next 15 or 20 minutes i'll be talking about the basics of iron metabolism we will discuss in
detail about the mechanism of pharoptosis and then understand the concepts of pharaoh process in various diseases and some applications finally we will conclude with understanding the differences between other forms of cell death whether it be it necrosis or other forms of programmed cell death now what is feroptosis feroptosis is a recent entity as i told you it's discovered in 2012 the word is derived from greek again ferro means iron and ptosis denotes falling off so let us see the overview of feroptosis before we actually discuss the details of each one of the concepts so what
happens is whenever there is increased intracellular iron that leads to the production of increased reactive oxygen species that overwhelm you know the glutathione dependent antioxidant mechanisms it sort of strains these antioxidant mechanisms and once that happens there will be unchecked membrane lipid peroxidation sedation that leads to loss of membrane permeability finally leading to cell death so this cell death resembles necrotic cell death but unlike necrosis feroptosis is regulated by specific signals but has a morphological features of necrosis so what has been found that reducing iron levels can prevent the cell within these patients so let
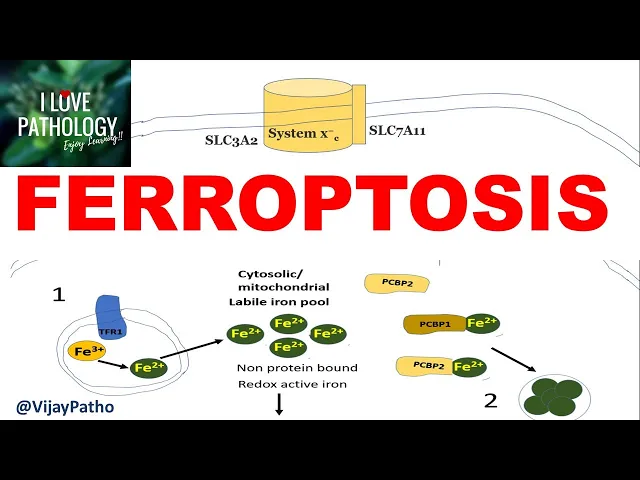
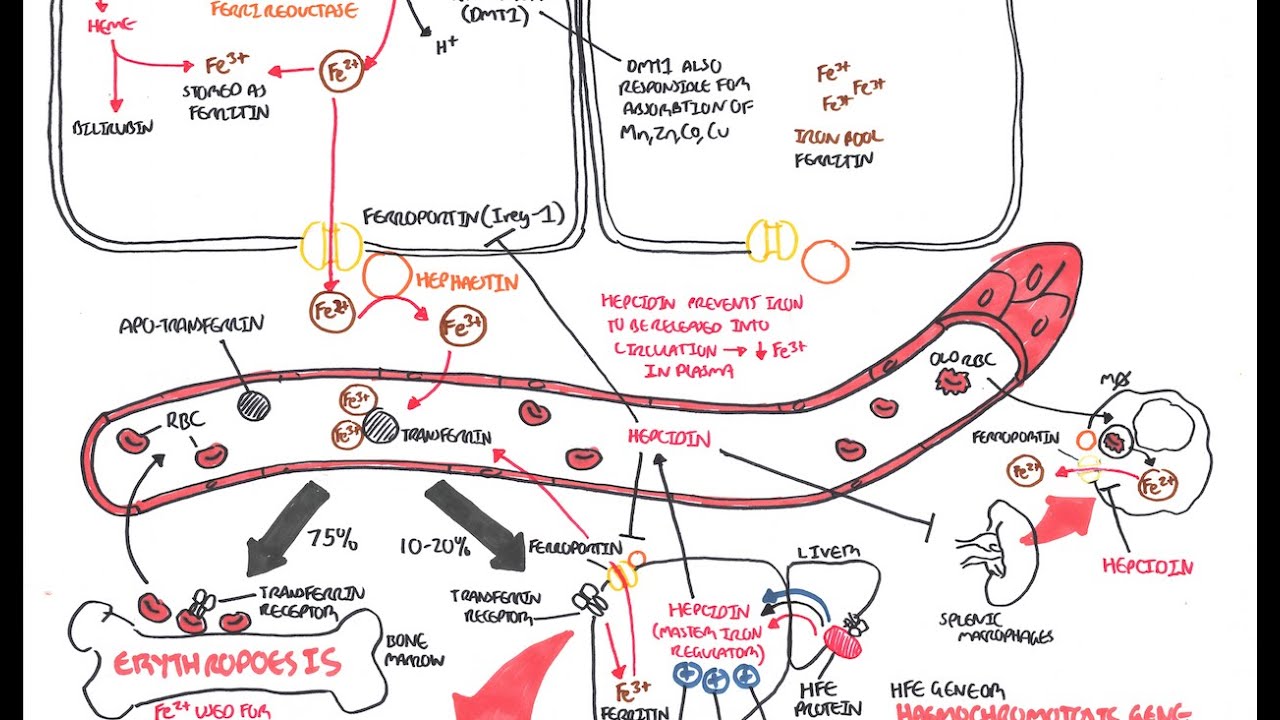
us understand this step by step first we'll talk about will understand the concepts of iron metabolism second we'll talk about how glutathione dependent antioxidant mechanisms work and thirdly we will talk in detail about the lipid peroxidation now coming to iron metabolism so this is a plasma membrane and this is a receptor called transferrin receptor this is a trans membrane receptor which binds the ferric form of iron and then results in a conformational change on the membrane resulting in endocytosis and that takes iron inside so basically this is an endosome where the ferric form of iron
is converted to a ferrous form of iron by the enzyme six transmembrane epithelial antigens of the prostate three or the steep three now from the endosomes these ferrous form of iron should be released outside into the cytoplasm by means of a protein called divalent metal transporter dmt-1 so within the cytoplasm many such ferrous forms of iron pool together to form a cytosolic or a mitochondrial labile iron pole this iron pool is non-protein bound and it is a very redox active iron that means they play a major role in redox reactions so this non-protein bound iron
is used in various cellular processes now you have two more proteins which helps in transportation of these ferrous form within the hydroplasm so that this form of iron is stored in the form of ferritin whenever in need this ferritin can be utilized you know can be utilized by the degradation in the lysosome to release the parasite now this released ferrous iron binds with pcb p2 protein so with the help of a transporter ferroprotein it transport the ferrous iron outside the cell so this is in gist as to what happens to the iron within the given
cell we told that ferroptosis is a iron dependent programmed cell death right now the conditions where iron intracellular iron is increased means to say that they are more susceptible to proptosis now what are the conditions you can think of one if there is any defect in the transmembrane protein that is transferring protein or for example over expression of transferring genes that results in increased intake of iron into the cell now second if there is defect in these two proteins pcb 1 pcb 214 that means these cytosolic label iron pool will not be converted to a
storage form that is ferritin right that means there will be more and more availability of cytosolic or labyl iron pool the third one if the ferropotent is not working properly if there is any defect in this ferroprotein that means still there is chances that the more and more level ironpool within the cytoplasm so all these three entities are important reasons for increasing the intracellular iron and then susceptibility to ferrotosis so that's one part we understood the concepts of basic iron metabolism and then we will see how this results in increased intracellular species a bit later
now let's talk about the normal antioxidant mechanism again this is a plasma membrane you have amino acid trans anti-transporter called system xc which has two subunits one slc7a11 and slc a2 okay with the help of this amino acid antitransporter there is recruitment of one molecule of cysteine into the cytoplasm for an exchange of one molecule of glutamate okay this is in the ratio of one is to one one molecule of cysteine comes in one molecule of glutamate goes out now cystine gets immediately converted to cysteine which then gets converted to glutamate this system with the
help of gamma glutamy cysteine synthetase gets converted to glutamyl cysteine with addition of glycine it is converted to reduce glutathione we all know that this reduced glutathione is a very potent antioxidant right that so this reduced glutathione tackles all the reactive oxygen species produced within the cell and within the organelles now there is another enzyme called glutathione peroxidase a very important enzyme you know it's it's it's one of the master regulator of ferrotosis we'll understand as to why so this glutathione peroxidase is one which which converts a very important lipid peroxide to lipid alcohol this
lipid peroxide is very very dangerous and that is the one which causes membrane lipid peroxidation and membrane damage okay so for this reaction to happen for the conversion of lyft peroxide to lipid alcohol you need glutathione peroxidase but at the cost of reduced glutathione where reduced glutathione is converted to oxidized glutathione but whenever in need this oxidized glutathione can be converted back to reduced glutathione for its antioxidant functions with the help of glutathione reductase of course that needs the conversion of utility of nadph where nadph is converted to nadp so what we need to understand
here is that this is the most dangerous thing we are interested in okay so this has to be converted to alcohol form lipid alcohol form because lipid peroxide is dangerous that is the one which results in lipid peroxidation and for that to happen glutathione peroxidase is very important enzyme for this enzyme to act it needs reduced glutathione and this reduced glutathione will be available only when the reactive oxygen species is not in excess because whenever the reactive oxygen species are in excess all the reduced glutathione will be utilized and if there is no reduced glutathione
this reaction does not happen and the lipid peroxide stays what are these lipid peroxides how are they formed they are formed either by enzymatic reactions or non-enzymatic reactions enzymatic reactions by means of lipoxynesis cyclooxygenesis and p 450 enzymes okay and non-enzymatic reactions where iron plays a major role again through a phantom reaction and enzymatic reaction let us see how it happens we all know that there is a polyunsaturated fatty acids in the lipid membrane right they get converted with the help of acsl4 and lp cat3 to polyunsaturated fatty acid phosphatidyl etherolamine so this bufa pufa
pe is the one which gets converted to lipid peroxide with the enzyme lypoxygenase where the ferrous form of iron acts as a cofactor now you understood right iron is an important component even in the conversion or even in the formation of lipid peroxide not only as a cofactor let us see how iron by itself helps in the formation of lipid peroxide so these are the two things which we studied right the concept of iron metabolism and then the reduced glutathione pathway now whenever there is increased intracellular label iron pool with the help of phantom reaction
there is lots of free radicals being generated more and more hydroxyl ions are being generated and whenever there is more and more hydroxyl ions this glutathione comes in to help and they neutralize these free radicals now now because of increased availability of intracellular iron more reactive oxygen species are formed and more utilization of reduced glutathione right so what does that mean so increased amounts of free radicals overwhelms the glutathione dependent antioxidant mechanisms this hydroxyl ion also helps in conversion of polyunsaturated fatty acid pe to lipid peroxide earlier we saw that lipid peroxide was formed enzymatically
with the help of lipoxygenase right so again free radical also results in the conversion of lipid peroxide now once there is more and more lipid peroxide because lipid peroxides accumulate lipid peroxides accumulate because this reaction does not happen the lipid peroxide is not converted to lipid alcohol because this glutathione peroxidase cannot function without the presence of reduced glutathione because all the reduced glutathione has been utilized to tackle these reactive oxygen species right now what happens when there is increased lipid peroxide that results in lipid peroxidation now what is this this is basically a chain of
reaction of oxidative degradation of lipids in the membrane the membrane can be a membrane of an organelle like for example mitochondria or membrane of the cell itself now once there is lipid peroxidation there is loss of membrane permeability so there is severe alteration in physical and biochemical properties of the cell membrane resulting in loss of membrane permeability ultimately leading to cell death so this is the basic mechanism of ferroptosis as we said morphologically it resembles that of a necrosis now what is the ultrasexual finding which differentiates ferrotosis from other forms of programmed cell death like
apoptosis so the most important to finding what we see in feroptosis is loss of mitochondrial cristae and there is ruptured outer mitochondrial membrane now why mitochondria the reason is mitochondria are the main intracellular producers of reactive oxygen species and two mitochondria are also you know focal labs in iron metabolism and homeostasis so that is the reason more damage is found in mitochondria of course the damage can be found in other organelles as well as the cell membrane itself but the predominant and the most important finding is found in mitochondrial membranes where there will be reduction
in the volume of mitochondria there will be loss of mitochondrial crystal and rupture of outer mitochondrial membranes now having understood the concept of ferrotosis let us see where all you can find feroptosis where all you can encounter serotosis ferrotosis is found in cancer where cancer cells you know accumulate high levels of iron as compared to normal cells so ferrotosis is also found or implicated in various neurodegenerative diseases for example like alzheimer's it is also implicated in ischemia reperfusion injury it's not just this ferrotosis involves various organ systems for example the gastrointestinal system the hepatobiliary system
the cardiovascular system the nervous lung name it any system almost all systems are involved in feroptosis okay so this is a wonderful article from the cellular disease and this image is from this article where you can easily see where you can make out that you know feroptosis is implicated in so many disease conditions what is the utility of knowing feroptosis so feroptosis is being tried as a treatment modality because you know executing pharoptosis as a non-apoptotic cell modality is one of the novel therapeutic opportunities to eradicate cancer cells now one such example is anti-tumor therapy
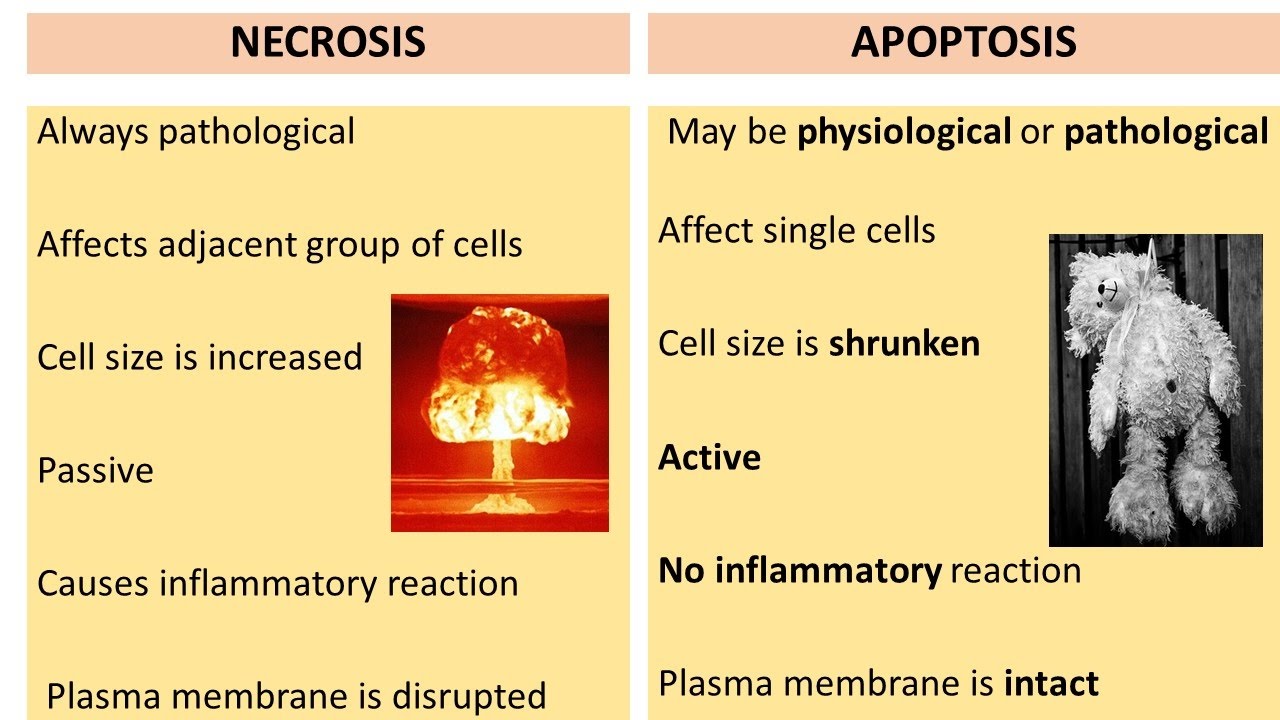
or radiotherapy you know where the anti-tumor effects of irradiation are attributed to the micro particles released by these irradiated cells which shown to induce immunogenic death mainly by ferrotosis so one thing we need to tell one thing we need to understand that the research on ferrotosis is still in infants actually this is just the beginning of understanding ferrotosis in various diseases and their utility in various disease processes so before i conclude let us see the differences between various forms of cell death i mean we we know the differences between necrosis and apoptosis right where the
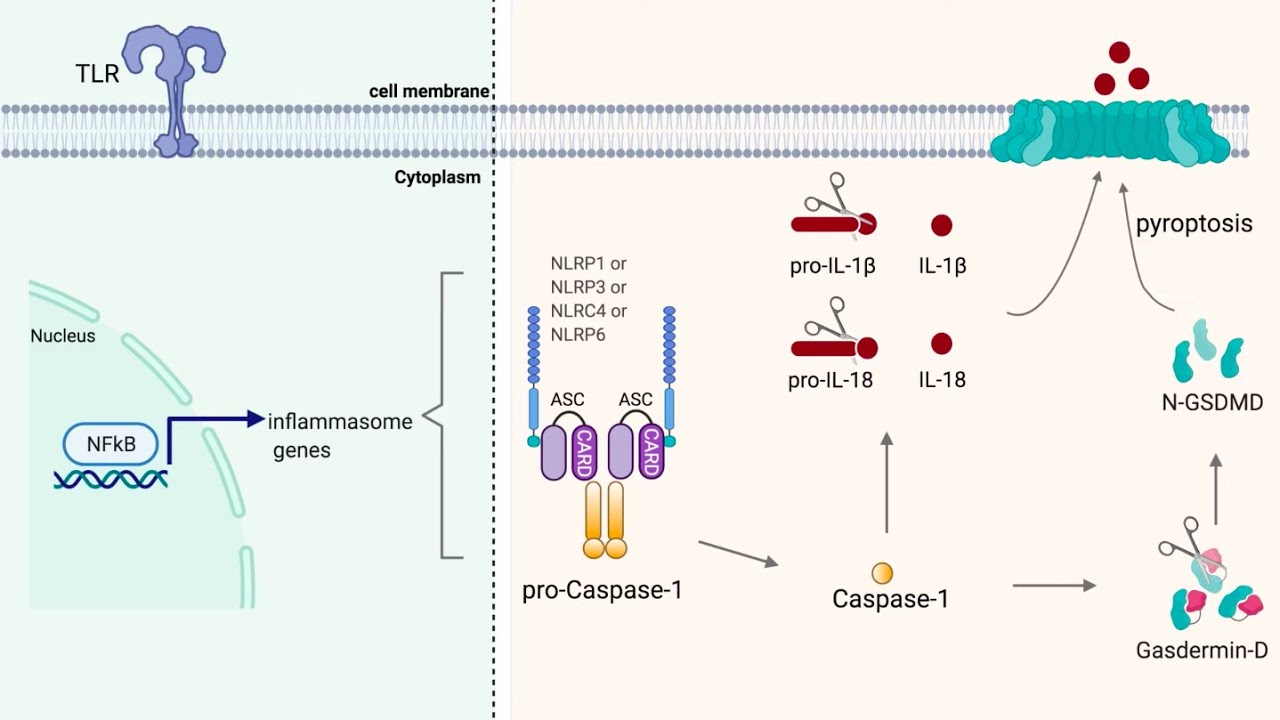
cell size increase the necrotic process is very passive the plasma membrane is disrupted it causes inflammatory reaction unlike apoptosis okay where there will be no inflammatory reaction and this is an active process it is a program cell death but the cell size is shrunken and the plasma membrane is intact you find lots of apoptotic bodies also right so let us see the differences between necrotosis pyrophosphates and feroptosis necroptosis is rip 1 and 3 dependent it is also a programmed type of cell death it's called programmed necrosis hydroptosis is by activation of inflammasome it's a caspase
one dependent it also has features of necrosis ferrotosis we all know that this is iron dependent and it also has features of necrosis but then there is predominant mitochondrial involvement so with this we conclude the understanding of ereptosis we discussed about the basics of iron metabolism and the role of iron we talked about the mechanism of seruptosis and how increased intracellular iron you know leads to the production of reactive oxygen species and that overwhelm the antioxidant mechanisms right and thus increasing in lipid peroxidation by lipid peroxides we also understood the various diseases associated with ferrotosis
and a few applications of erectors and finally we concluded with understanding of differences between other forms of regulated or programmed thank you for watching if you have liked this video please hit the like button do comment don't forget to subscribe if you're new to this channel and do share if you find this video useful thank you