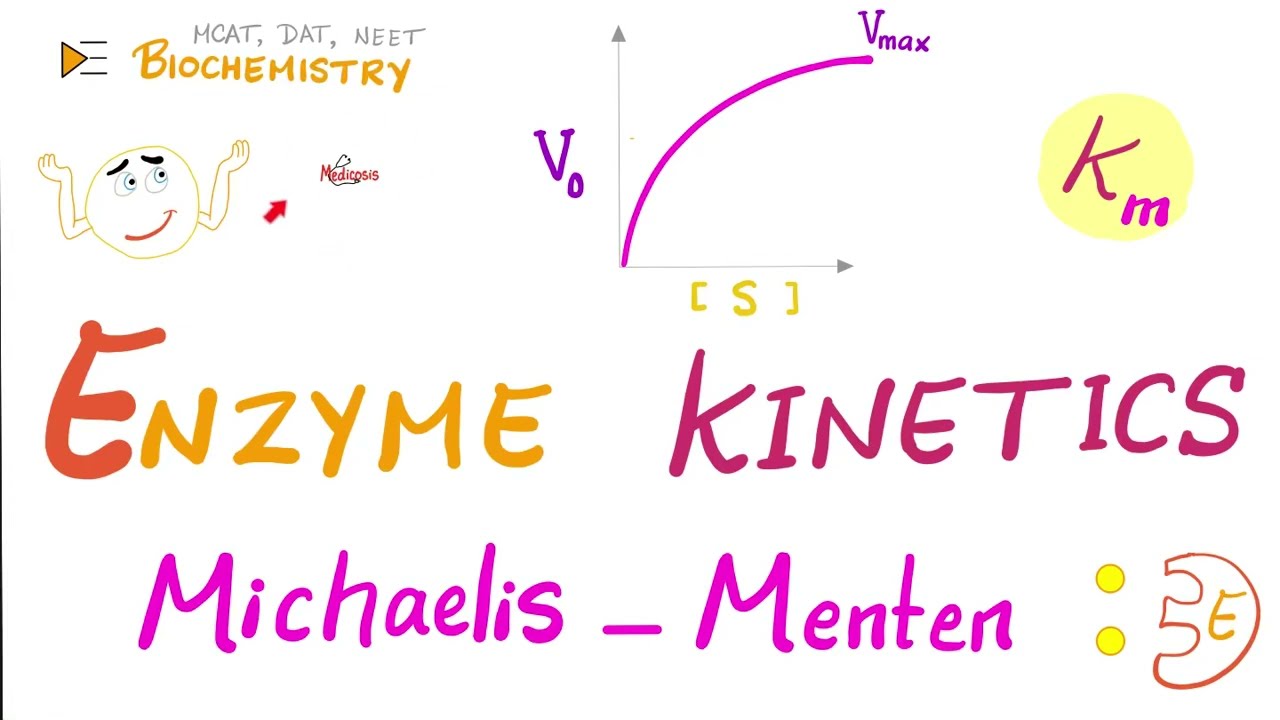
hello wonderful people what is going on it's meosis perfectus where medicine makes perfect sense welcome to the first video in my bio chemistry playlist where together we're going to study the chemistry of life what is your diet your diet has fed protein and carbohydrates your body uses metabolism to deal with these foods you can build stuff up called anabolism or you can break down called catabolism utilizing lots and lots of enzymes enzymes are catalysts they increase the speed or the rate of the chemical reaction without being consumed by the reaction click the like button click
the Subscribe button and let's get started this is my biochemistry playlist please watch these videos in order for maximum understanding and retention I have a video for every biochemistry topic that you can imagine by the way I have two biochemistry playlists on this channel one short and one long if you're studying for the MCAT or the DAT or the neat exam Etc watch the short one but if you want to be a doctor a nurse a professional watch the long one the short one is titled biochemistry for the MCAT the long one is titled biochemistry
new as in neogenesis your diet has carbs has proteins has fat when you eat you eat big molecules and then through digestion you'll break down the big part into smaller parts so the carbohydrates whether starch or glycogen will be broken down into disaccharides and then monosaccharides can you give me examples of carbohydrates sure starch which is of plant origin and glycogen from animal origin the disaccharides include meltose sucrose and lactose and then you break down the disaccharides into monosaccharides so the meltose will give you glucose and glucose sucrose will give you glucose and fructose and
lactose will give you glucose and galactose then you're going to break down the big proteins into polypeptides oligopeptides dipeptides and amino acids or monopeptide so to speak so dipeptide has two amino acids tripeptide has three amino acids oligopeptide has a few amino acids polypeptide has many amino acids then you break down the fat which is big in the beginning triglycerides into smaller parts such as glycerol and free fatty acids it's not glycerol per se but two mono ail glycerol when it comes to digestion your pancreas is the hero because your pancreas can complete digestion from
A to Z your pancreas has digestive enzymes to break down carbohydrates to break down proteins and to break down fat can you give me an example of an enzyme that breaks down carbohydrate sure how about amas oh yeah that will do it next can you give me an example of an enzyme that will break down proteins a peptidase Trin can also help and to break down fat or lipid I need a lipase if it ends an a it's an enzyme metabolism came from the Greek meta or Metabo which means to change oh what type of
metabolism well I have two types there is anabolic to build up and there is catabolic to tear down let's build up okay building up is an endergonic process what do you mean endergonic means energy and enters oh so I need energy energy goes in energy goes in so that I can build up stuff so that I can build up small glucose molecules into a big glycogen molecule this is going uphill to build up which requires energy however catabolism is the opposite it's exergonic because energy will exit it is Flowing downhill because we are releasing energy
we are breaking down the glycogen into glucose for example this release is energy exergonic energy exits when do you build up stuff when I am fed when do you break down stuff when I am fasting who is the hero of the anabolic land it's insulin and who is the hero of the catabolic land glucagon and his friends what do you mean by his friends I mean epinephrine cortisol and sometimes thyroid hormone insulin alone is on the left and every other hormone that you can imagine is on the right so we say that insulin is is
a regulatory hormone whereas the rest of the bunch are counter regulatory hormones now let's talk about the most important topic in Biochemistry which is insulin Land versus glucagon land the feeding State versus the fasting State the land of abundance versus the land of scarcity this is the insulin World on the left and this is the glucagon World on the right again it's not just glucagon it's glucagon and his friends the epinephrine the thyroxin the cortisol Etc dear insulin when do you work I work in the feeding State because I am a builder I am anabolic
I will build up proteins I will build up glycogen I will build up lipids tell me more how do you build up proteins I convert amino acids into big proteins # protogenesis meaning protein synthesis proteo means protein Genesis means synthesis creation formation next glycogen anabolic I take the glucose molecules and then build them up to glycogen # glycogen synthesis or glycogenesis that's if you want to build up glycogen but if you want energy right now I can burn that glucose for you via glycolysis to give you energy in the form of ATP or adenosine Tri
phosphate speaking of fat it's the same thing I am fat anabolic because I will build up free fatty acids into big lipids big Tri glycerides # lipogenesis or lipid synthesis whereas glucagon and epinephrine and cortisol and thyroxin are destroyers they work in the fasting State when you're fasting you're not eating by definition see you need sources of energy what should I do you should break down what you stored for all these years so the protein will be broken down into amino acids # prois or protein breakdown then you can take those amino acids and make
glucose oh you're making glucose from noncarbohydrate sources oh you're making glucose from new sources gluco neo genesis Genesis means formation Neo means new oh new because this is non-carbohydrate Source gluco because I'm making glucose next glucagon is glycogen catabolic it will break down glycogen to glucose which is the exact opposite of this when you break down glycogen to glucose this is called glycogenolysis or glycogen breakdown glucagon and his friends is also fat catabolic it breaks down the big lipid triglycerides into free fatty acids and glycerol # liolis or lipid breakdown by the way anytime you
break down fat like this Ketone bodies are going to emerge whether you like it or not so you only see Ketone bodies in the glucagon world you never see Ketone bodies in the insulin land because insulin is the major anti- ketogenic hormone in the entire body never ever forget this if there is insulin there are no ketones but if there is no insulin there are ketones and that's why some patients who have diabetic keto acidosis I.E they lack insulin guess what they do have a bunch of Ketone bodies and these Ketone bodies are what acetone
aceto atic acid and betah hydroxy acid have you noticed that these are acids for the most part so ketones are keto acids and that's why if I have a bunch of these in my body I have KET keto acidosis and since I have no insulin they call me diabetic so now I have diabetic keto acidosis because I lack insulin therefore I have a bunch of Keto acids in my blood see biochemistry makes so much sense once you understand what the french toast you're talking about for the pros please note that glycolysis belongs to the insulin
world but glucon neogenesis is part of the glucagon land if you want to download these doy colorful not go to medicosis perfection. if you want me to personally tutor you reach out to me on my website this is about half of your biochemistry textbook in one slide what is the central Hub in Biochemistry this is a silo a it's the sun around which all the planets revolve cuz whether you eat proteins or lipids or carbohydrates at the end of the day they become a COA the central Hub and then a c COA if you have
oxygen is is going to delve into the TCA cycle or the tricarboxylic acid cycle also known as Krebs cycle or citric acid cycle and then this will give you ATP moreover you're going to capture the H in the nadh and fadh2 all of these hydrogens will be captured in the electron transport chain which will give you even more energy both the TCA cycle and the electron transport chain occur in the mitochondrian when you take glucose and burn it it's called glycolysis and this is part of the insulin World glucose gets burned into pyrovate and then
pyrovate becomes a silico by the famous enzyme pyate dehydrogenase all of this started from Big carbohydrates like glycogen which is polysaccharide then you break it down into disaccharide and then into glucose and then glucose to pyruvate and then pyate to ailoa and you enter the TCA cycle how about proteins they get broken down into polypeptides oligopeptides a bunch of amino acids and boom a COA how about lipid or fat these triglycerides will be broken down into free fatty acids and glycerol the glycerol can give you glucose because glycerol is gluconeogenic the free fatty acids by
beta oxidation will give us aico a anytime you break down lipids you're going to get Ketone bodies Ketone bodies can also give you a silico a and a silico a can give you Ketone bodies when you are making Ketone bodies it's called a ketogenesis when you are bre breaking down ketogen bodies it's called ketogen olysis when you are making glycogen it's called glycogen synthesis or glycogenesis but when you are breaking down glycogen is called glycogenolysis when you take some protein sources and some fatty sources and convert them to glucose it's called glucon neogenesis some important
facts carbohydrates when you eat them are going to give you 4 kilo calories per gram proteins 4 kilo calories per gram fat 9 kilo calories per gram alcohol 7 kilo calories per gram notice that fat is nine which is the highest number here and that's why the favorite method for your body to store energy is in the form of fat and that's why if you eat too much you're going to grow more obese oh that's more fat why isn't it the case that when I eat so much I become more muscular or more sweet why
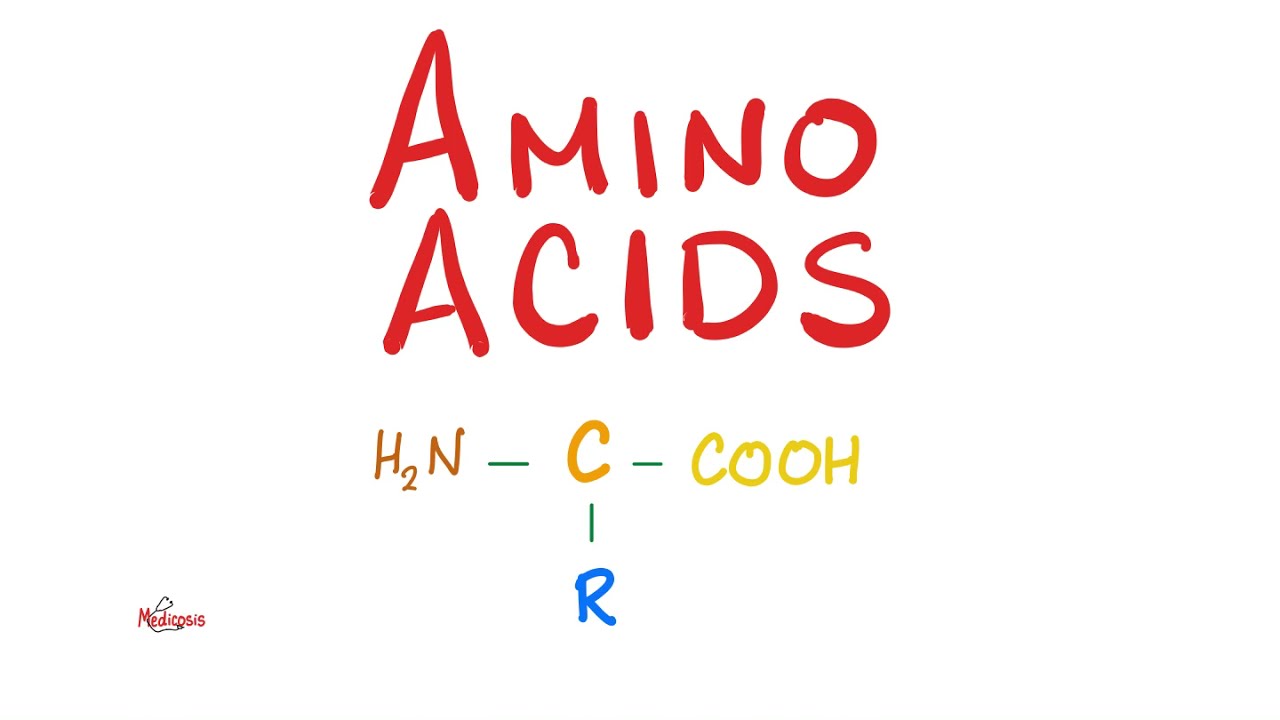
do I always become obese because fat is so dense from an energy standpoint it gives you 9 Kil calories per gram and that's why it makes more economic sense to store extra energy in the form of fat that's how your body does it please take a moment to pause and review next we'll talk about the amino acids start with this main carbon known as the alpha carbon if you want to know the difference between Alpha carbon beta carbon and Gamma carbon please refer to my organic chemistry playlist why do we call them Amino acids because
this is amino group and this is carboxilic acid group amino acid and this is the order by which they come out of the ribosome Amino comes before the acid that's why we say Amino before the acid and then there is a side chain or the R Group as you are familiar with from organic chemistry and this will determine the properties of the amino acid some amino acids have H instead of an R some have methyl oh just methyl yeah some have very long side chains some have branched side chains and some have a ring structure
please note that both the amino group and the carboxilic acid group are attached to the same Alpha carbon Amino is usually basic carboxilic acid is by definition an acid and that's why we say that the amino acid is amphoteric like an ampian it is half basic and half acidic by the way the amino could be written in many ways you can write it nh2 or nh3+ similarly the carboxilic acid group could be written as Co or Co negative when should we write it as Co versus Co negative was discussed in great detail in my video
titled titration of amino acids Hint it depends on the pH that you put the amino acid in and the pka of the amino acid next stereo chemistry Leo means left Dex remains right there is also s versus R don't forget that in your body you have D sugar but you have L amino acids sugars to the right but amino acids to the left what's the difference between this and this Leo versus dextro is performed via plain polarized light rotation but s versus R is done by a pencil and paper and they were discussed in great
detail in my organic chemistry PL look at this this is on the left so we call this Leo but this one is on the right we call it dextro keep in mind humans have D sugars but L amino acids so in your body right now your sugars are D for example D glucose not L glucose but your amino acids in your body right now are L amino acids not D amino acids do we have exceptions sure all amino acids in your body are chyal with l meaning to the left meaning Glo except glycine which is
not even chyal therefore it's neither L nor R and cysteine is another crazy exception look the acids in your body are L and S okay but cine is L yet R so when you use the plain polarized light cine is going to rotate the light to the left hence Leo but when you do it with pen and paper you find it R because it's clockwise and not counterclockwise all the amino acids have the amino group and the carboxilic group attached to the same carbon we call it the alpha carbon however keep in mind that we
have something called Gaba what is Gaba GMA I mean americ acid you know why we call it gamma we call it gamma because the amino group is bonded to the gamma carbon not the alpha carbon how many amino acids are there lots and lots of amino acids but hey my professor said that there are only 20 amino acids your professor lied to you look the 20 amino acids that you're talking about are the proteogenic amino acids however these are not the only amino acids in town because we also have nonproteogenic amino acids what's the difference
proteogenic amino acids get incorporated into proteins hence proteogenic they make proteins but nonproteogenic amino acids are not incorporated into making proteins moreover proteogenic amino acids are coded for by a genetic codone a piece of the DNA code for alanine for example or veine or glycine but you do not have a code on your genetic codons to make orane you do not have an order or a gene to make homosysteine or selenocysteine but these are still called amino acids and some of them are inside your body right now such as homosysteine now what are the famous
20 amino acids these are alanine and Arginine aspartic acid glutamic acid asperene glutamine phenyalanine tyrosine glycine lysine loine isoline cysteine veine histadine serine Proline tryptophan thionine and methionine each one of these 20 proteogenic amino acids have a name a three-letter abbreviation and a onlet abbreviation for example alanine is ala it's also just a lysine is l s it's also k isoline i l e it's also I histadine is h s also H take a moment to pause and review most professors most exams want you to know their names and the three-letter abbreviation however some professors
happen to hate their mothers they want you to memorize everything including the one letter abbreviation these people are sick next some amino acids can give you glucose we call these glucogenic some amino acids can give you keton bodies we call these ketogenic some amino acids can give you both glucose or Ketone bodies we call them both glucogenic and ketogenic what are the amino acids that are purely glucogenic these include alanine cine methine Arginine aspartic acid glutamic acid osine glutamine Proline veine glycine histadine which ones are purely ketogenic lysine and Lucine Lucine and lysine lysine and
Lucine which ones can give you both glucose or Ketone bit isosine alen tyrosine tryptophan thine and curine notice that almost all of the amino acids can give you glucose and these are all the amino acids except lysine and Lucine another classification of amino acids we have essential amino acids semi essential also known as conditionally essential amino acids and nonessential amino acids the essential amino acids what does that even mean essential means that your body cannot make them from scratch therefore it is essential that you eat them and consume them in your diet and these include
Lucine isoline phine tryptophan methine theine Hine and lysine on the other hand we have some nonessential amino acids it means that your body can make them from scratch therefore you do not have to eat them in your diet these non-essential amino acids include alanine aspartic acid glutamic acid aspine and cine then we have the semiessential or conditionally essential if you are a normal person you do not need to eat them in the diet they are not essential because your body can make them but if you suffer from a certain metabolic disease such as the famous
inborn errors of metabolism then one or more of these amino acids become essential because of the disease that you have for example there is a disease known as phal ketonuria in this phal ketua disease tyrosine which was normally nonenal becomes essential only in patients with phal ktina so if most people did not eat tyrosine in the diet they will be okay but if a patient with fenel kenua forgot to consume tyrosine in the diet this patient will suffer big time because for that pheno ketonuria patient tyrosine is essential others include arine cine glycine glutamine and
Proline to learn more about finel kenua and the other inborn errors of metabolism please refer to my biochemistry playlist now let's classify these amino acids in another way you can classify them into five groups as you see here first group non-polar non-aromatic side chains second group aromatic side chains and these will have a Benzene ring hence aromatic and these are beloved by your Chef Gordon Ramsey oh F me it's aromatic in my 30 plus years in The Culinary industry I've never seen so much arrogance Hey Big Boy listen get your head out of your sphin
pathetic number three polar nonaromatic side chain number four negatively charged or acidic side chain and last the amino acids that are positively charged or with basic side chain groups three and4 and five can form hydrogen bonds with water in aquous Solutions here are the five groups again group number one nonpolar non-aromatic side chain these include alanine glycine methine Proline and the branched chain amino acids including veine Lucine and isoline then we have the aromatic side chain they have a benzine ring these are the pheny alanine if you add hydroxy to it it become tyrosine and
you also have tryptophan phen alanine tyrosine tryptophan then you have polar non-aromatic side chain some of them are hydroxy containing amino acids the sorine and the thine some of them are amide side chain containing amino acid the S part Gene and glutamine some of them contain sulfur such as the famous cysteine negatively charge or acidic side chain well it's called acidic foras I can say aspartic acid or glutamic acid and they have an acid group remember the carboxilic acid is co if it's deprotonated it becomes Co minus this is minus hence negatively charged on the
other hand we have positively charged or basic side chains these include the Arginine the lysine and the histadine now let's start by talking about the first group which is the nonpolar non-aromatic side chain these include alanine and glycine the branch chain veine Lucin and isoline methionin as well as Proline please keep in mind that methionin and cysteine are sulfur containing amino acids you should never ever forget this methionin and cysteine are sulfur containing whereas cine and thionine are hydroxy containing I should be able to wake you up from a coma and you should still remember
that methine and cysteine contain sulfur whereas Serene and thine contain hydroxy now get some sheets of paper and a pen because I'm about to take you to class we will draw every one of the 20 amino acids and we will learn three facts about each amino acid starting with the amino acid glycine how do you draw glycine just like any amino acid with the alpha carbon in the middle then you have Amino on one side and acid or carboxilic acid on the other side you have H here and you have the r here except glycine
is different glycine has H instead of an r and that's why according to Legend glycine is the smallest amino acid look at this cue bouti why is it the smallest amino acid because it has H instead of un long R it's the simplest smallest amino acid what are the three facts that you need to know about gly fact number one it's the smallest simplest amino acid fact number two it is a chyal because this carbon is bonded into hydrogen and hydrogen making it not chyal because by definition a chyal carbon needs to be bonded to
four different species but these are not four different species that's why the C is not chyal therefore glycine is neither Leo nor dextro it's neither L nor d f fact number three is that glycine takes part in the formation of collagen which is a famous member of the extra cellular matrix of your body we find collagen in Bones carthagesavings you have Amino on one side carboxilic acid on the other side you have H here and instead of an H like glycine alanine is more complex it has an actual R which is just methyl alanine another
non-polar non-aromatic side chain amino acid what are the three facts that you need to know about alanine fact number one you can convert pyruvate into alanine and alanine into pyrovate the famous enzyme that does this is called Al trans aminase or alanine aminot transferase or simply alt which requires vitamin B6 pyro oxin as a co-actor never ever forget this alt is very important in your liver it's one of the liver enzymes or liver transaminases fact number two that alanine is part of something called a Cahill cycle this is the hell that alanine wants to die
on so now you know the three facts of alanine fact number one you can convert it to pyrovate and you can convert pyrovate to it fact number two this is the kahil cycle fact number three I need alt and B6 for this next we have methine still non-polar non-aromatic how do I draw meth Alpha carbon here you have Amino on one side carboxilic acid on the other side give me that hydrogen here and then what ch2 am I done nuh-uh ch2 am I done no methan is a sulfur containing amino acid don't forget your sulfur
and end with a methyl group this is methionin methyl methionin it is sulfur containing what are the three facts that I need to know about methionin fact number one it contains sulfur fact number two remember the St kudon on your beautiful mRNA remember AOG AOG codes for methionin the first amino acid to be put in the chain fact number three that methin can give you Uncle Sam Uncle Sam is the ultimate methyl group donor and when Uncle Sam loses its methyl group it becomes Uncle sa s adoil methin versus S adoil homocysteine here is glycine
the simplest amino acid here is alanine instead of theh you have methyl here is methionin which has sulfur and it has methyl methin then we'll talk about Branch chain amino acids we are still talking about nonpolar non-aromatic side chains but we're going to add a branch so let's draw veiling together how do you draw valine easy here is your carbon here is the amino and here is the acid and then what you have an H here okay but look at the branch that's going to happen oh look at this Branch this is a branched chain
amino acid and you have a methyl here and a methyl there this is veine what are the three facts that I need to know about veine fact number one it is a branched chain amino acid fact number two it is the smallest branched chain amino acid and fact number three there is a disease known as Maple curine disease where the patient has deficiency in the branch chain Alpha keto acid dehydrogenase these patients cannot metabolize vein Lucine or isoline so veine isoline and Lucine will end up in the urine giving the urine the classic maple syrup
odor this disease is known as maple syrup urine disease next Amino it is Lucine how do we draw this it is very similar to veine but a little more complex so here is the amino and here's the carboxilic acid this is the hydrogen and this is the r you have ch2 and then you're going to do CH then your branch oh so what is the difference between veine and Lucine the difference is just here that veine had CH and then your branch but Lucine has ch2 then CH then then New Branch so Lucine is more
complex what are the three facts that you need to know about Lucine fact number one it is a branched chain amino acid fact number two it is muscle essential fact number three because it's branched it is also implicated in maple serup urine disease then we have isoline ISO means what same or similar to Lucine oh so it's going to be similar to Lucine which means it's branched as well so here is amino on one side here is the carboxilic acid on one side and here is H then the r look at this the r is
Branched in a different way you have the H here and you have the ch3 here and then you continue ch2 and ch3 the carbon that's attached to the amino and the carboxilic is the alpha carbon and it's going to be followed by the beta carbon is this carbon kyal well here is something something something something and all of these are different the species are different so so this is bonded to four different species so yes this is a chyal center how about this well I am bonded to h on one side ch3 on one side
ch2 on the third side and C which is bonded to all of these on the other so yes I am kyal meaning that isoline has two kyal centers so what are the three facts that we need to know about isoline fact number one it is Branched fact number two it has two kyal centers fact number three because it's branched it's implicated in Maple Sy urine disease these are the branched chain amino acids veine is the simplest one then Lucine then isoline isoline has two chyal centers to learn more about chirality please refer to my organic
chemistry playlist next Proline Proline is cyclic it has a ring so let's draw well this is the alpha carbon and here is the H here is co on one side and nh2 here and then what then this carbon is attached to ch2 which is attached to ch2 and which is attached to ch2 and boom before you know it we have this ring it is a five membered ring as you see and this five member drink has nitrogen as you see therefore we call Proline it's not just an amino acid but an Amo acid as well
what are the three facts that you need to know about Proline fact number one it is cyclic it has a five membered Rin fact number two Proline with a p is a pervert with a p why because this ring introduces kings in the peptide chain and that's why it's a pervert and because it's a pervert it's not only an amino acid but also an amino acid even the word Amino was perverted fact number three you need Proline to make the famous collagen which is found in bones carthagesavings blood vessel walls Scara and more here is
Proline again please pause and review this concludes the first group of amino acids which is the non-polar non-aromatic side chain the next group is aromatic side chain meaning that they have a Benzene rain these include tryptophan and phel Eline which will make tyrosine get your piece of paper and doodle with medicosis how do I drop tryptophan easy here is your Alpha carbon and you have Amino you have acid good don't forget the hydrogen don't forget the r this R has a ring because we're talking aromatic here so look at what's happening here okay look at
what's happening here okay look at what's Happening Here what is going on well you got to add a Benzene ring just like this this is just beautiful amazing how many rings does strip to Fan have the answer is two it is a double ring what are the three facts that I need to know about tryptophan number one it is the only amino acid that has a double ring fact number two you need tryptophan to make nasin nasin is vitamin B3 and this niin or nicotinic acid will give us the nad+ and if it gets reduced
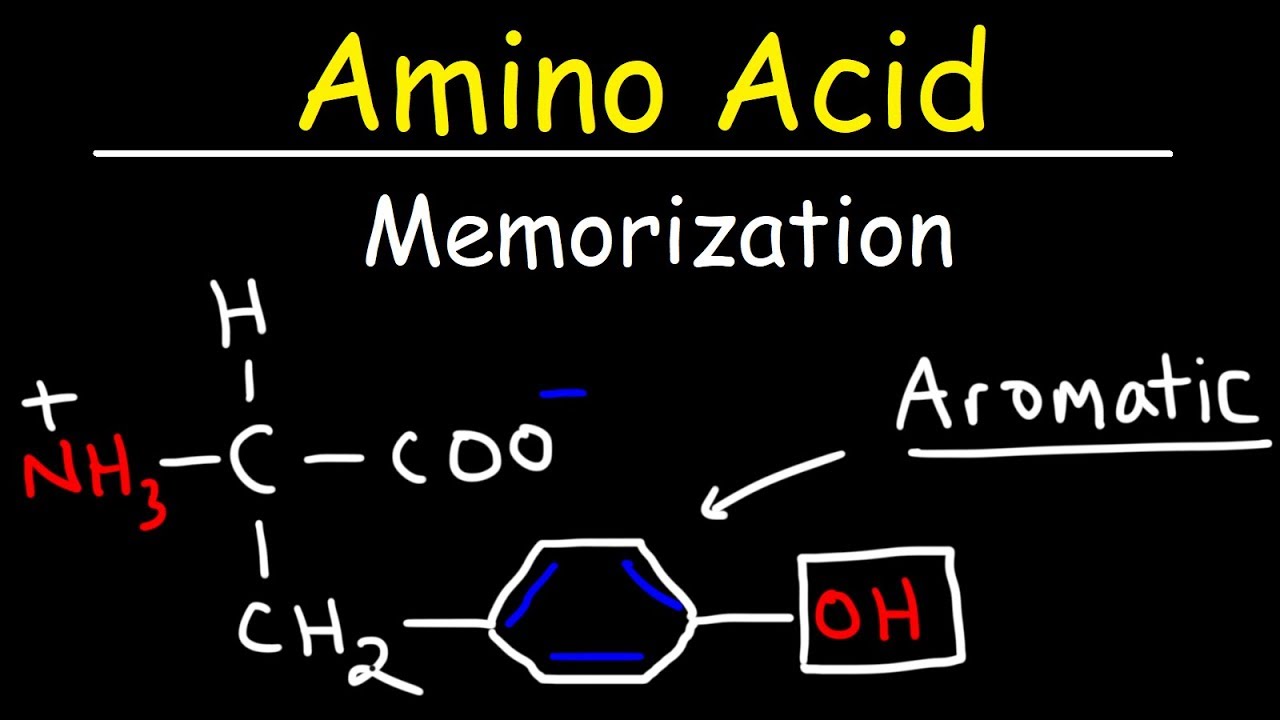
it becomes n88 and of course you're going to capture that H in the electron transport chain to make some energy fact number three about tryptophan you need tryptophan to make serotonin and and you need serotonin to make melatonin why do we call serotonin 5ht because it's five hydroxy tryptamine tryptamine yeah because it came from tryptophan and tryptophan is an amino acid tryptamine next pheny alanine also aromatic also has a ring this is my Alpha carbon here is amino on one side and carboxylic acid on the other side don't forget the H don't forget the r
this R is ch2 and you have your Benzene ring which is a six membered ring and don't forget the double bonds if you do not want to draw the double bonds you can draw a circle instead even better what are the three facts that you need to know about finel Aline fact number one it's an aromatic amino acid with a Benzene ring fact number two it is relatively nonpolar fact number three phel alanine will make you tyrosine if you just add a hydroxy group to it so pheny alanine plus hydroxy group equals tyrosine and that's
why tyrosine is a semi essential amino acid if you're healthy you do not have to eat tyrosine because don't worry your pheny alanine will make you the tyrosine that you want but if you have pheny ketonuria you cannot make tyrosine from pheny alen therefore tyrosine becomes essential for you which means you have to eat it in the diet see what happens when you get your head out of your sphincter you tend to understand biochemstry better next is tyrosine are you aromatic of course I am aromatic where did you come from I came from tryptophan which
is the previous amino acid if you just add a hydroxy group to tryptophan it will become tyrosine so let's draw tryp toan which looked like this the famous Benzene ring which is amazing like this and add to this o boom you have tyrosine what's the name of the enzyme that added the hydroxy group group to the pheny alanine to make tyrosine well guess what hydroxylase NOA and this pheny alanine hydroxylase enzyme is deficient in patients suffering from a disease known as phel ketonuria which was discussed in detail in a separate video in this biochemistry playlist
what are the three facts that you need to know about terine fact number one it's aromatic with a Benzene ring fact number two it is relatively polar why I added o making it relatively polar when you compare it to phenol Aline which was relatively nonpolar fact number three tyrosine will give us dopa and thyroid hormone and dopa can give us dopamine and dopamine can give us nor epinephrine and nor epinephrine can give us epinephrine nor epinephrine is also known as nor adrenaline and epinephrine is the same as adrenaline so phenol alanine will give you tyrosine
tyrosine will give you dopa dopa will give you dopamine dopamine will give you nor epinephrine nor epinephrine will give you epinephrine so it goes like this phen Eline tyene dopa dopamine nor epinephrine epinephrine phine dopa dopamine nor adrenaline adrenaline all of this happens in the adrenal midle the last three dopamine nor epinephrine and epinephrine are famously called the camines amines yeah because we're talking amino acid here these are the aromatic side chains which have Benzene Rings you have tryptophan you have phen Aline and if you add hydroxy to it it becometh tyrosine what's the name
of this enzyme pH alanine hydroxy and this is the same song that we just did pH alanine tyrosine dopa dopamine norepinephrine epinephrine or pH alanine tyrosine dopa damine nor adrenaline adrenaline all of this happens in the adrenal midle do amine nor EPI and Epi are collectively known as ketamines tyrosine can give you dopa it can also give you thyroid hormone in your thyroid gland pheny alanine is converted to tyrosine through a famous enzyme known as pheny alanine hydroxy which is missing in patients with pheny kenua then from tyrosine to dopa there is a bunch of
enzymes and there is a cycle enzyme deficiencies will give me tyrosinemia dopa can make melanin and if I have a problem or deficiency in this enzyme tyrosinase I will not be able to make melanin and I will suffer from a disease known as albinism an albino is a person with a very very very very light and pale skin because this patient cannot make melanin because this patient is lacking tyrosine this concludes the second group of amino acids onto the third group polar non-aromatic side chain these include serine and theine both of them have hydroxy group
we have cine which has sulfur what else had sulfur today if you said methionin you're absolutely correct then we have as sparine and glutamine and they have amide notice that amide I had a circle that is blue you know why because Amite has oxygen in it and this is the O that I drew inside the D let's draw Serene remember that this Serene has hydroxy yes it does have hydroxy but does it have a Benz ring no these are nonaromatic amino acids so I do not have a Benzene ring but I do have a hydroxy
group so let's draw Serene where should we start we should always start with the alpha carbon give me an amino here and a carboxilic acid here don't forget your H and the r where here's a carbon I will not forget the o because Serene is hydroxy containing and then on the other side H and H is this a chyal carbon well since it's bonded to four different different species yes indeed it is chyal however look at this carpon is it chyal well here is H here is H these are not different species so the answer
is no this second carbon is not chyro which makes it liable for all the damages that happen next next is thionine another hydroxy containing amino acid so here is the alpha carbon here is amino on one side and carboxilic acid on the other side don't forget the H and look at this there is c o here and H there but instead of an H as in Serene here you have ch3 what are the three facts that you need to know about thine number one it's hydroxy containing as you see number two it has a methyl
group instead of an h and the way I remember this is that I think of thionine with a th as methyl group with th and number three unlike serene theine actually has two chyal centers Serene versus thionine both are hydroxy containing the difference is that thionine has a methyl group Next we have cine this one has sulfur so I got to find a way to put the sulfur this is amino this is acid this is the H and then ch2 now you add the sulfur sh what are the three facts that you need to know
about cine number one it is polar and nonaromatic number two it has sulfur and because it has sulfur we can use it to make disulfide bonds or disulfide Bridges and we need this in the primary and tertiary protein structure to learn more about the difference among primary secondary tertiary and quary protein structures there is a video on this topic which you can find in this biochemistry playlist here is suine and it has sulfur next is a spar Gene okay a spar Gene is one of those that has amide what's an amide group a group that
has nitrogen and oxygen so let's just start by Amino on one side and acid on the other don't forget the H and then you have ch2 here and where is the amide the amide is C double bonded to oxygen and the same carbon is bonded to nitrogen this is amide the three facts that you need to know about aarene include the fact that it's polar the fact that it has non aromatic side chain and the fact that we use it for strong hydrogen bonding in the secondary protein structure next is glutamine also have an amide
group so it's going to be very similar here's the carbon here is amino on one side and the acid on the other side here is the H and then ch2 repeat it again ch2 and then give me that Amite Group C double bonded to O and bonded to nitrogen so simply glutamine is slightly longer compared to asper gen what are the three facts that you need to know about glutamine number one it is polar number two has nonaromatic side chain and number three it is important as a transport of nitrogen throughout the body and this
is going to be super important when you learn about trans amination deamination and Thea cycle later in this playlist here is a spar Gene which has an amide group and this is glutamine another amide group this concludes group number three next group number four negatively charged or acidic side chain aspartic acid has an acid Co this is an acid what if you remove the H what if the proton left the chat the remainder will be Co negative we call them acidic because this is an acid we call them negatively charged because Co negative aspartate came
from aspartic Acid after it lost a proton but when glutamic acid loses a proton it becomes glutamate and if you are a chem buff you would recall that if something ends in eight odds are it is negative does anyone remember bicarbonate negative because it ends in eight note that aspartate and glutamate contain Co negative but aspar and glutamine contain amide side chain how do we draw is part8 which is negative this is how we draw it this is the carbon Amino on one side and the carboxilic acid on the other and here's the H ch2
is here and then where is the co o it is co o but I lost the H so instead of Co o it is co negative it is a negatively charged side chain the three facts that you need to know about aspartate number one it came from as spartic acid after it lost a proton number two it has a negatively charged side chain number three it helps eliminate nitrogen in diarrhea cycle later when we talk about the Ura cycle you will know that Ura has two nitrogen groups one of those nitrogen groups actually came from
aspartate never ever forget this next is glutamate similar to aspartate but longer so here is the C and here is amino on one side carboxilic acid on the other here's the H the side chain is longer so ch2 another ch2 and then you have the carbon bonded to oxygen and O Negative this would Co and we called it glutamic acid after losing a proton it became Co minus the three facts that you need to know about glutamate number one it is derived from glutamic Acid after a proton was lost number two it has a negatively
charged side chain number three glutamate is actually an excitatory neurotransmitter in your nervous system remember that Gaba is inhibitory but glutamate is excitatory here is as pate and they longer glutamate pause and review this concludes group number four last group number five positively charged or basic side chains we have Arginine which has three nitrogens we have lysine and we have histadine with the famous imidazol ring pneumonic to help you remember these three amino acids is the following her leggings are so basic basic because these are basic side chains HLA histadine lysine Arginine for the pros
you know the histones oh DNA yep we wrap your DNA around histones to make chromosomes these histones are nucleo proteins nucleic acid and proteins these histones are rich in arginine and they are rich in histadine histadine histones arginine has three nitrogens look at all the ends in the word here is the alpha carbon which is bonded to Amino on one side and carox IC acid on the other don't forget the H don't forget the r what is the r very long ch2 repeat it ch2 again ch2 then a nitrogen bonded to hydrogen here and to
a carbon here carbon is double bonded to nitrogen and single bonded two nitrogen here is a nitrogen here is the second nitrogen and the third nitrogen what are the three facts that you need to know about Arginine number one it's positively charged it has a basic side chain number two AR can give you something called nitric oxide which is a potent vasodilator it dilates your blood vessels number three is the fact that Arginine has three nitrogens next we have lysine similar to Arginine but less drama this is the alpha carbon and this is my Amino
and this is my acid don't forget the hydrogen don't forget the r ch2 ch2 repeat it again ch2 repeat it again ch2 and finally the nitrogen is this basic of course this is basic is this positively charged yep what are the three facts that you need to know about lysine number one it is basic it is positively charged side chain as you see number two it has a terminal primary NH3 group number three later in fatty acid metabolism you'll learn about the carnitine shuttle this lysine is going to be converted to carnitine to help fatty
acid transport in the carnitine cuttle to learn more about fatty acid metabolism please refer to my videos in this biochemstry playlist the last amino acid for today is histadine histadine has nitrogen and it also has a ring so this is the carbon it's called the alpha carbon this is nh2 and this is the carboxylic acid and don't forget the H don't forget the r here is ch2 and then we're going to start building the ring here is carbon double bonded to another carbon single bonded to nitrogen which has hydrogen here double bonded to carbon which
has hydrogen here and single bonded to nitrogen and then you continue the ring and this is called aidasol ring or imidazol what are the facts that you need to know about histadine number one it is positively charged basic side chain how come this is the positive end here and of course this end could be positive if you protonated but this depends on the ph and I've talked about this Topic in my video called titration of amino acids fact number two about histadine it has an amidol ring and of course amide has nitrogen as you see
fact number three histadine is a physiological buffer in your body because you know what the pka of this group is it's about six and what's the pH of your body it's about 7.4 since the PK is similar to the pH this makes histadine a very good buffer and don't forget the that you need histadine in histones here are the positively charged basic side chain amino acids you have arginine lysine and histadine the pneumonic is her leggings are so basic and here are the five groups of amino acids again please pause and review amino acid derivatives
pH Aline tyrosine dopa dopamine nor epinephrine epinephrine glycine will give me protor which gives me H which is part of the hemoglobin which carries oxygen and carbon dioxide in your blood tryptophan gives me serotonin which gives me melatonin it also gives me niin and n and gives me nad+ if you reduce it it becomes nadh if you capture this H in the electron transport chain you're going to end up with energy how many atps about 2 and A2 approximated to three ATP molecules per one molecule of nadh histadine gives me histamine by decarbox ilation decarbox
silation meaning removal of carbon dioxide also glutamate will give me Gaba when carbon dioxide leaves the chat the moment you say decarbox vitamin B6 is going to help you as a co-actor in order to make collagen in your bones in your tendons in your cartilage in your blood vessels in your basement membrane in your hair in your Scara Etc you need three amino acids don't forget them glycine Proline and lysine quick note on protein metabolism again metabolism has two parts anabolism and catabolism don't forget that anabolism is insulin land but catabolism is glucagon land you
take those amino acids and then bind them together oh dipeptides then tripeptides then oligopeptides many polypeptides bigger proteins amino acids just one amino acid get two together bind them you have D peptide get three together tripeptide get between four and 20 oop peptides more than 20 polypeptide what are you trying to achieve you should begin with the end in mind but what end are we talking about are we talking about the N Terminus or the C Terminus haha very funny that's the funniest that you can get with biochemistry of course the N Terminus is this
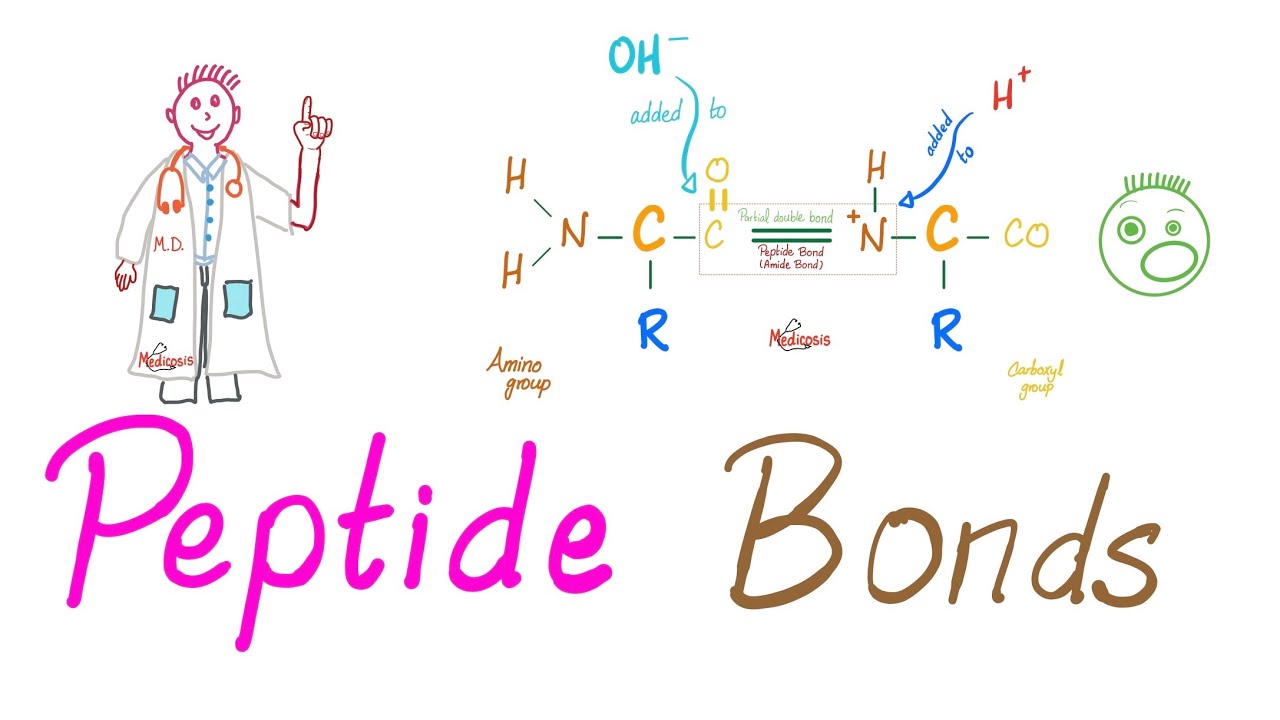
and the C Terminus is this amino acid what I was trying to say is what are you trying to achieve are you trying to build up this is anabolic or to break down catabolic if you want to build up let's lump some amino acids together combine them together condense them together to make bigger proteins how do you condense by removing water it's a dehydration reaction so making peptide bonds is a dehydration or condensation reaction but breaking down proteins into amino acids requires the opposite instead of removing water you need to add water and this is
called hydrolysis so forming peptide bonds is a condensation dehydration reaction but breaking down peptide bonds is a hydrolytic reaction performed by hydrolytic enzym such as your digestive enzyme digestion is catabolism so here is one amino acid here is another amino acid let's lump them together to form a peptide bond this used to be Co the O is going to leave the chat this used to be nh2 one of the H's will leave the chat h plus o h boom water water is leaving # dehydration # condensation condense them together what's the name of this Bond
peptide bond how does it look like it's an amide group it's a carbon double bonded to oxygen and the same carbon will be single bonded to that nitrogen and that same nitrogen is bonded to hydrogen this is amide for you amide has oxygen let's make a peptide bond here is one amino acid here's the second amino acid remove water # condensation # dehydration before you know it you have a DI peptide you have a peptide bond the pepti bond has an amide group can you call this condensation sure can you call it dehydration sure can
you call it asil substitution also sure to learn more refer to my organic chemistry playlist here's one amino acid here's another amino acid let's make a peptide bond make a peptide bond before you know it I have a dipeptide what is this carbon double bonded to oxygen and the same carbon is bonded to nitrogen which is bonded to hydrogen this is aide Group which is the essence of the peptide bond remember if you want to make a peptide bond if you want to build up proteins this is anabolic insulin world but if you want to
break down the peptide bond this is catabolic glucagon World peptide bond formation is condensation or dehydration but peptide bond breakdown is hydrolysis remove water add water now let's break down proteins into amino acids you do this every day you eat proteins when you eat proteins your body is going to add water to them during digestion in your intestines the proteins become dipeptides when you add water it's called hydrolysis the enzymes are hydrolases such as the peptidases such as the trein the kyot trein the carboxy peptidases ETC add another water before you know they have amino
acids this is hydrolysis which is breakdown the Tron and the kyot triin are hydrolytic enzymes or hydrolases tripsin breaks the carboxilic end or the C Terminus of Arginine and lysine but kimot tripson breaks the carboxy end of phenol Aline tyrosine and tryptophan the way I remember which is which is that I think of Kimo as chemical and chemicals can smell like perfumes and these are what aromatic amino acids they smell how do you break a peptide bond you add water add o here is going to attack on this side and add an H here to
attack on that side this is basically the opposite of peptide bond formation instead of condensation we're performing hydrolysis today you need to hydrogen the nitrogen and hydroxy the caroy hydrogen the nitrogen and hydroxy the caroy hydrogen the nitrogen hydroxy the caroal before you know it I'm breaking the peptide bond # hydrolysis and I have two separate amino acids now to learn more about all of these different digestive enzymes please refer to my physiology playlist especially the gastrointestinal physiology series during periods of prolonged starvation your body has to do something break down sugar we did this
a long time ago break down fat done what else should we do break down proteins your body does not want to do this but your body was forced to do this with its back against the wall because you're starving and I better break down my muscles than die so I'm going to break down the proteins including the proteins in the muscle so catabolism of proteins will happen is this insulin land or glucagon land glucagon land of course when you break down proteins they become amino acids by transamination and the amination amino amination nh2 will leave
okay and then what this Amino will enter the Ura cycle and the ammonia will be converted to Ura and Ura will leave the body through the kidney as for the remaining carbon skeleton we can use this to release energy if the amino acid was one of those glucogenic amino acids voila we can make glucose if it was a ketogenic amino acid we can make Ketone bodies and this was the story of biochemistry in a nutshell I hope you learned something today if you value what I do help me make more videos by supporting the channel
go to buy meac coffee.com medicosis there are more than 600 premium videos available on this channel when you click the join button and choose the highest tier Please Subscribe hit the Bell smash like support my my channel on patreon PayPal or venmo go to my website to download my courses notes and cases and if you would like me to tter you be safe stay happy study hard this is medicosis perfectionalis where medicine chemistry math and physics make perfect sense