today uh clearly i am a noble gas um and so thanks to is actually the ta's idea so and i was like of course that sounds awesome so um i hope you guys have a fantastic halloween obviously what you want to do is any kind of trick-or-treating or partying you know the drill you bring your periodic table because you never know and and that's what happened you know so on sunday right before the exam i got this and um okay so so this is uh charles and raymond and they're saying we wanted to celebrate the sox victory in boston but because the midterm was the next day we brought along our periodic table to study there were so many people moving around it felt like we were in a sea of electrons and of course the only thing that could have improved on that was actually like a little dance video but that'll probably come i'm sure they're on that next um but see they brought it not just to study for the exam because they didn't know if it was going to be important or not at the rally you never know right so so make sure you have your periodic table with you um now uh okay oh yeah they mentioned the exam so speak speaking of the exam um here are the results and um you can see that there's a pretty wide distribution um you can also see the average is a 77 and just to remind you that's that's about here just to remind you these uh these uh marks 85 that's the a range this is the b range right so the average was in the solid b range territory um and and then and so it goes um the standard deviation was 12. now i could tell you know exam one i think a lot of the topics of exam one many of you had already seen and i think in this exam some of you had not seen some of these topics especially like the crystal crystallography but also even the molecular orbitals the band structure and and i could tell that you know so some people were a little bit uh maybe uh you know you had to work a little harder maybe a little bit stressed and i could sense uh some of that uh but you know when i when i get stressed what i do is i need to kind of de-stress somehow and sometimes you know some people listen to music some of the other things i always like comedy as a way of of relaxing and so what i do is i'll google like for articles about about harvard um and i found and so i found this one this is actually an article in the crimson and you know and so some a few years ago and uh the most common grade at harvard is actually an a an a a solid a the most common grade um um and i like there's a okay suspicion that the college employs a softer grading standard than many of its peer institutions you think if everyone i mean if everyone's getting an a you think i i mean but the thing is uh so what i wanted to point out is like there's a difference right and it's not just about uh okay your privilege therefore you deserve an a um whereas we know that that actually takes work that's not the point i want to make the point i want to make is that we know also what to do when we miss things we know what to do when we don't get things right right that's when you do the work you know so wherever you are on this curve go back and figure out what didn't work what did you miss figure that out because that's where you learn right i mean there's a thomas edison who said i never failed i just did 10 000 experiments that didn't work right that you know you got to get through that you got to know that it takes hard work and that's the thing that we know here that's the difference so please make sure you do that with exam two we are all here to help you continue learning on the noble gas anyway okay and speaking of continuing to learn um right where were we x-rays so the last time now um uh on friday we had sort of you know some other things going on but i also was trying to teach you guys about x-rays and because in type and and what we did is we learned how they're generated right remember that the renkin experiments right um and so i want to pick up here with the kinds of x-rays that we have there are two kinds of x-rays right um and and we learned about those on on friday and i want to just remind you about those and i also want to um show you a video uh to kind of recapture what they are and the the the first kind of x-ray remember we plotted this as intensity and i'm not going to i'm not going to draw the cathode ray tube again and the experiment that renkin did and all that but but i'm going to just jump to the two kinds of x-rays that you get right and so if you plot the the wavelength of the x-ray versus the intensity of the x-ray right then one kind is the bremsstrahlung which comes from that that electron getting slowed down and and if it slows down it emits radiation in a continuous spectrum right and so so you might get you know remember we we sort of drew these these yeah okay and and this might be you know this would be like the incident electron energy right incident let's say incident um energy of the electron okay maybe like that's uh okay i don't know like 10 kav 10 kv and then up here the incident energy of the electron was you know something like maybe 30 kev right and so you see that it it as you as you hit that anode that remember what rinkin did he took a cathode ray tube and he upped the voltage so he really cranked up the voltage so those electrons coming off the cathode are going really fast right and then what he did is he put a piece of metal in their way and and what happened is those really high energy electrons they see those metal atoms and sometimes they get inside the electron cloud and they turn and that's when they give off this continuous radiation right but but but we also know that there's a limit here we right remember that we talked about that and that this limit limit is set by this maximum this it's just the the maximum that you could the maximum amount of energy that you could get out of a photon being emitted this way would be equal to the incoming electron the incoming electron transferred all of its energy to the photon so that's why there's a maximum and and it's also why it increases remember right shorter wavelength higher energy right so it increases as you increase the energy of the incident electron okay that is brem strolling let's watch a video because i this is actually a very nicely done uh video that captures it with animation so okay so here's your crt now okay here they come those are those electrons in there voltage is high so they're coming out look at them coming out really fast right lots of kinetic energy okay and then there they go and they put and this is what rincon did he put a little piece of metal in there so now those electrons are hitting the metal okay so those are the two things that you differently remember the room lit up even when all the lights were off okay there they are nice good electrons oh and here they go and there's the metal atom okay and oh look at that now what are these those are x-rays coming off those are the electrons hitting the metal and here's the metal atom and as the electron comes in remember it sees the charge of the nucleus and it gets deflected and that deflection loses energy and that loss of energy goes into a photon now because these electrons have such high energy to start with because they have such high energy to start with um you know the energies of the photons are very high and they're x-rays right they're x-rays um and so okay and so here's the wavelength and you can see that uh well it's sort of a little hard to see in this blue range but see higher energy shorter wavelength lower energy longer wavelength very nice okay so that's the animation of brem stalling now okay good now the thing is what we also learned is that there's another type of x-ray there's another type of x-ray and and in fact if you crank this up high enough you get that you can get that other type so now we go higher oh we didn't see it here it just looks like that and that and that and then all of a sudden we go up to 40 kev and we see this why right what happened here and what happened here is a totally different mechanism for generating x-rays right and that was the second type that we talked about on friday those peaks are called characteristic and and the reason is that remember we have these levels which now that we are talking about x-rays we give them letters k l m n but it's just the quantum numbers n equals 1 n equals 2 right okay n equals 3 n equals 4. and what we said is that the way characteristic x-rays are labeled is that if an electron is excited from this gazentite from this lower level right if an electron is is knocked out of here then there's a place for an electron from here to go down maybe the electron is excited maybe the electron is kicked out and then something here can come down and when that happens just like just like in the bohr model you get radiation right but now unlike hydrogen 13.
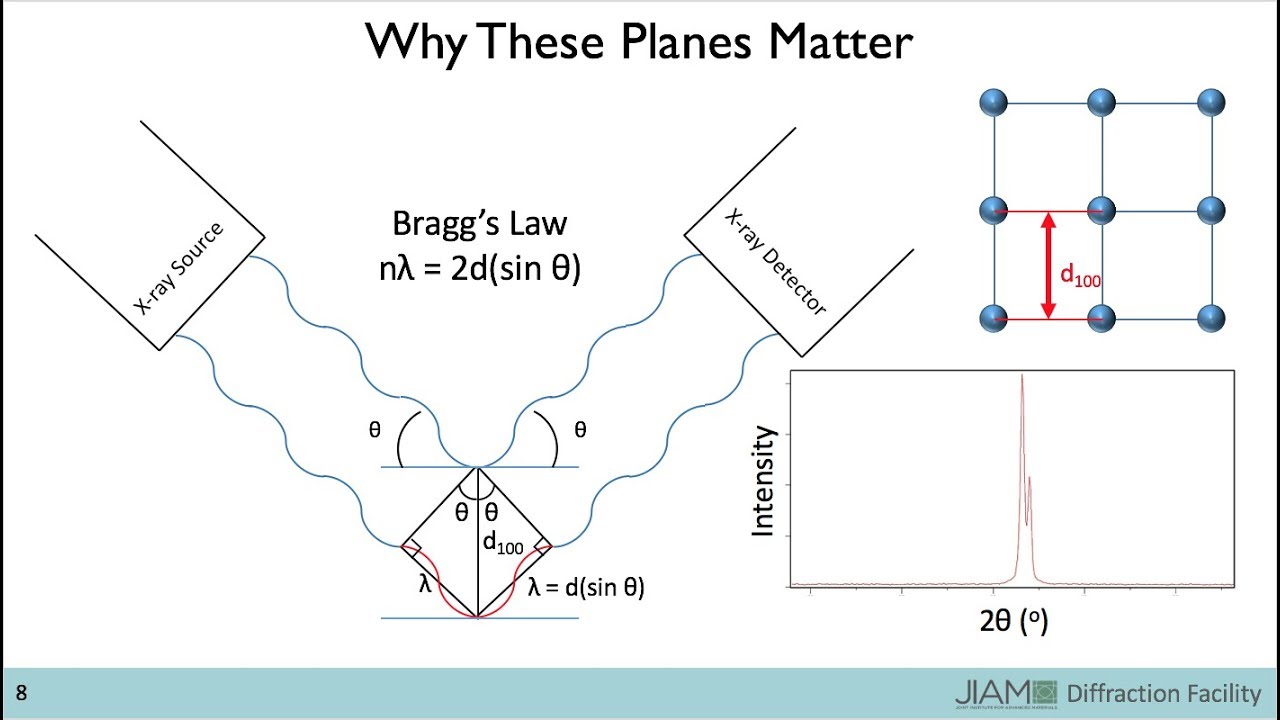
6 electrons these are kev of energy these are very high energies why because it's the 1s electron and we know that once you get down to those metals right wait those 1s electrons you go down further down the periodic table those 1s electrons are seriously tightly bound yeah and and those levels have a have a difference down there that is pretty high in energy it's x-ray high in energy that's the point so now when that cascade happens we call that a k alpha and if it were to have come from here it would be called a k beta right and and those are transitions that unlike this continuous energy those transitions only happen at very specific energies delta delta energies right the change in energy from l to k or or for k beta would be going from m to k you say it's k alpha k beta you use the k because that's the final place the electron goes when it it decays down okay so if you had to if i just showed you these and say well that would be like k alpha and that would be like k beta because you know that k beta is going to be a higher energy photon right because it came up from a higher level okay well you would also have on here some other peaks right you would have the l peaks right so you'd have like l alpha l beta right see so you see so so as you crank the energy up right then you can knock out those core electrons and these cascades happen and you get these discrete peaks notice they will only come when you have enough elect enough energy in that incident electron to knock this electron out from the core so that's why they don't appear until you get to a certain incident electron energy right they don't appear until you get to that certain energy right and so we have a video on that which also i will narrate but is this it there we go okay so there it is it's a metal atom i don't know which one um and what you're okay there's the incident electron you fired it and look at that it knocked out a core 1s electron because it had enough energy very high energy that's what rinkan did crank up the voltage higher kb and there it is a cascade down and an x-ray comes out you see that okay so so that uh very uh is that all oh yeah and then it's gonna draw because of those you get these characteristic peaks now we call them characteristic because now you can see why so unlike the continuous radiation right these peaks depend on the atom because they depend on the energy levels of the atom right and so that's why like if you look this up you say well okay let's look at the k alpha radiation let's look at the k-alpha peaks right that come out of of different atoms they're going to be different okay like so so you have so you have very sharp lines of of x-rays at very specific energies for copper it's 8 kv for for molybdenum it's 17. 5 right silver tungsten it changes and you can see that it goes up right as you as that 1s electron is lower and lower in energy because i'm adding all these protons so it all makes sense from the concepts we've learned oh but see now now that's really useful that is really useful because now i've got a way to have a source of x-rays that is super well defined it's super clear it's always this ah that's so cool it as long as i have the same metal it's always the same right i can increase or decrease well i can't go below the threshold but i can go above it and that peak is characteristic of the metal so it doesn't change that's really useful because i've got now a flashlight i've got an x-ray flashlight where the energy that i'm sending out is always exactly what i know it to be i can predict what it is and it always will be that depending on which metal i put in there right so so that's a useful that's a useful thing why is that useful well that gets to the the topic that is the topic of today and a friday which is what are we doing with these x-rays well first we're generating them so that's what we've talked about so far right but now because in tight we're going to actually use them to determine the crystal that we have we're going to actually use them we're going to use that flashlight okay and and so you know so you can see why this would be useful because here is the this is the range we've showed this before right of x-rays so they they have these energies of kev and they have wavelengths right around a few angstroms that's a little less than an angstrom maybe two angstroms right see where those wavelengths are well those are atomic spacings those are like distances between layers right and so if we could shine these on a crystal and and somehow figure out what it is with that light oh there's a way we can do that it's called diffraction because what happens is and and we know this from from many fields this is you can think about this just as a water wave any wave okay if the wavelength is similar in size to the features then you get constructive and destructive interference as a result of the interaction between the wave and the features so that's called diffraction right and you can see it here with this very simple uh picture of say a water this could be like a water wave a sound wave you know and there it is and it's it's interfering uh both constructively and destructively along these lines right you can do this test yourself you could take um i highly recommend this take a laser pointer and uh now if i is there now if i just had a piece of metal i don't have a piece of metal but if i did then i would shine it on it and what you'd see is that the dot would just reflect off the piece of metal okay so i just would get the dot back right but now i've got this thing here this we many of you may not know what this is um this is called a cd okay and oh but it turns out that a cd has it has features and it's got trenches that are like you know a little less than a micron apart 800 nanometers and this is 500 nanometer light so you would expect there to be diffraction you would expect there to be constructive and destructive interference and when i bounce it off of this look at that there it is this never gets old i'm not getting i'm not getting just one reflection here i'm getting a whole scatter of them that have constructively interfered because of the feature sizes being the same as the as the wavelength okay but now i want to do that with x-rays and i want to do it onto onto crystals so how do we do that okay so let's think about that and we're going to think about it in terms of what the bragg father-son pair who won the nobel prize and are on a stamp that's what you get uh when you win a nobel prize i guess um and um and what they did is they figured out how to do this okay so let's let's go through that just so we understand it conceptually um okay so if i have i'm going to say that i have a set of atoms now right now i'm not going to worry about what they are um but i'm just going to say that there's some plane of atoms here and there's another plane of atoms beneath that okay so there's another one and so on and so on and now these these would be miller planes right these would be miller planes in the crystal and let's just assume they're very very simple this plane in that plane and now what i'm going to do is i'm going to have some x-rays i'm going to have some light shining on this and it's going to be incident and it's going to be reflected but see i'm going to have another wave here okay let's see if i can get through this drawing sort of almost kind of um now here's the deal okay these are waves right these are waves so if i had if if i want to draw this as a wave i might draw it like that and if these are if these waves are constructively interfering let's just complete that then this wave would look like that right if they're the same if they're in phase then that's what they would look like now this one's getting reflected off the surface right so i'm going to do that reflection here okay and if i want it to come back off of the surface and i want and this one got through the first layer this didn't get reflected it's going down and if i want it to come back up then this one must also look like that to be in phase right they must be in phases as they come out or else they're not going to interfere constructively you see that so those are my x-rays right they're waves they're just waves right oh but this is the this is the whole secret right because you know if if this is if this angle here is theta then what that means is that this angle is theta okay well yeah and if that's true then this distance is d sine theta where this is d that's just some some simple geometry okay so so what you know then is okay now now we're getting somewhere right now we're getting somewhere because you know if i had a wave this is one wavelength right and so so if i had a wave um come in like this and and one of them is going to get reflected off of this lower surface and the other one got reflected off this but i don't want them to interfere in any way but constructively that's what that's what bragg said bragging brag that's what they said then the only way for that to happen is if this distance plus that distance so d sine theta plus d sine theta is equal to some multiple of the wavelength it has to be right and so what you get is well that's what they have there n lambda some multiple of the wavelength equals 2d sine theta where theta is the incident angle of that x-ray okay so this is incident just to be clear incident x-ray and these are reflected reflected x-rays okay now to keep it simple in this class we're just going to say n equals 1. n equals 1 for 3091 just to keep things simple because we're really just i want to grasp the basic concepts here all right we're not using x-rays to get the the structure of dna but we're going to use x-rays to figure out cubic crystal structures we're going to show how that works now so this must be true for so this is in parentheses four construction interference constructive interference those dots that you saw interface interface no interference interference right for this is true for constructive interference of course you could write any equation you want but if you want them to be constructively interfering when they come out that has to be true and that's what the bragg said but see that we're not there yet right because now we got to do experiments okay so we got to do experiments so how so what does this mean well okay i'm going to i'm going to take x-rays of some lambda okay and i'm going to shine them on a sample right okay and i'm going to measure so what i'm going to do oh i think i have a picture i'm going to measure there it is this is what an x-ray diffraction experiment looks like okay so i've got some sample and i shine x-rays i've got a source of x-rays we now know how to make that source right and we'll just filter out one of these lines right okay so i've got that source and i can change the angle and then i've got a detector and and i can measure did i get interference or not it's just like the dots right so so now we know that if i if i do that and and i and i say i i scan the um you know i scan the end okay so i've got intensity and now i'm plotting it with angle so so now i'm moving this intensity i'm moving the angle around and i'm changing it bam i get interference and i see a spot in my detector so literally it would look like this right you would get some angle where there's interference and the detector will say ding ding ding ding ding i see a lot of x-rays coming off when i didn't and now you change the angle by a few degrees and i don't see anything right because it's all destructive from these crystal planes okay okay so it seems like then if i just vary theta um am i there yet the problem is that i might not know d i might not know d ah ha but we do know d we do know d because we learned about d for cubic crystals we know d because because for cubic systems we know that d of h k l right of any plane the distance between those planes is equal to a over the square root of h squared plus k squared plus l squared and that's something that we learned this is distance between miller planes miller planes because then tight in a cubic crystal oh i'm saving time i wrote excel i saved a lot of time which i just wasted by being so proud of it okay so i know d well okay so what does that mean well let's take a look because now i'm going to go back i'm going to go back to my equation and i'm going to say okay lambda equals 2d and i'm saying n equals one okay and i'm going to be very specific this is now a d that comes from the spacing between planes that are specified by the miller indices right times the sine of theta right and i'm putting h k l on the theta as well and you'll see why because here's the thing now i've got constants now i've got constants because look this is fixed by the source by the source so this is a constant you know if if i have copper then it's 1.
54 angstroms right this is a constant this is fixed by the crystal that's also a constant fixed by the crystal because that's also a constant because it's the lattice constant right we're not changing that so for a given set of planes that these that these waves are bouncing off of and and maybe maybe constructively interfering with right that depending on the theta then then these are constants right okay so so if i regroup them so i'm going to regroup them right and so i'm going to say that let's see d so i'm going to substitute in that expression up there down here okay and i'm going to use a copper source right so i'm going to say well let me let me go through this one step at a time so d equals 1. 54 over let's see 2 sine theta h k l now what have i done here this is for a copper source so i'm i'm now adding in i'm fixing in the constant okay fixing in the constant okay so that's the brad condition but i also know that d is equal to it's also equal to ah i don't want to write it again it's also equal to that okay good okay so let's put that together we'll do a little division and what we get is 1. 54 angstroms over 2 a squared the whole thing's squared i'm squaring it i don't want the square root equals sine squared theta h k l over h squared plus k squared plus l squared four let's be very specific for constructive interference okay constructive interference and a copper source so now i'm getting specific because this is how experiments with x-ray diffraction are done right so but now the last time i checked right if you got something equal to a constant then that something also is a constant right and and this is a constant yeah and and so so what i need to do now is figure out right i'm going to measure these thetas where i get a signal remember i'm going to i'm going to now change theta so that i see where do i get signals right now those thetas divided by the plane that that that by the hkl that they are bouncing off of must be a constant they must not change that is at the heart of x-ray diffraction that is at the heart of it right all right so we're going to do that with a specific example today and friday but before we do that there's another thing that we can observe there's another thing that we can observe oh this is what it would look like so here it is right so so now i i've changed now why do we do two theta it's kind of historical you plot x-ray diffraction uh spectra so this is an xrd right x-ray diffraction spectrum this is the intensity of the peaks right and these are the peaks this is like this is a beautiful thing i'm seeing a crystal here i'm seeing a crystal and by friday you will be seeing a crystal that those aren't just peaks those are planes in a crystal those are planes in a crystal which tells me not only that that i have these planes but it tells me what crystal i have but that's not how we start the way we start is we do these measurements and we just read off angles so we got to get from there to there to crystal structure so what i want to determine is the crystal structure and the lattice constant that's my goal what i have is a spectrum that looks like this where all i've done here is put these specific angles here and you have to be given okay so it's it this is the aluminum xrd spectrum so it's it's if you shine x-rays on aluminum this is what you get if you know also that those x-rays are from copper which means that lambda is fixed right so this would be like the in you know the the information you'd get to to start you'd be given this spectrum you you're given these peaks and you'll be given this information here it's a copper target and from that we can determine the crystal structure in the lattice now um now there's there's something oh why do we write why do we do two theta well it's historical it could have been theta to make all those dividing by twos go away but instead you can see that as i as i rotate this right this changes by theta this the detector changes by two theta so that's why it's x-ray spectra are given in two theta there's no other real good reason for it um even though in the bragg condition it's not two theta it's theta right this comes from geometry okay of the planes and this just comes from historical setups and then how you move the the detector okay so what's measured and plotted is the two theta okay now but before we go before we do this transformation where we take an x-ray spectrum like this and and we and we get the information we want there's one more thing and that is not all reflections are allowed not all reflections are allowed and and so and so let's talk about that and then we'll come back to this spectrum right now you can you can kind of understand this by looking at just a simple uh kind of comparison here okay simple comparison all right if i take so these are the hkls remember that's the hkl for a miller plane uh this is h squared plus k squared plus l squared why do we put that because we know we're going to need it right there it is right there so we know we're going to need it okay but if you look at the simple cubic that's simple cubic right any combination of hkl is okay there is no combination that would that would give you interference along that along those planes stacking let me say well i don't okay yeah what are you talking about why are you even bringing this up well when you see the other two crystal structures you'll see what i mean so now we have the case of bcc right when you see this you see bcc and fcc and what i'm showing you here isn't the 100 plane it's the 20 plane right so this is the family remember the family of two old planes there they are there they are right oh but now now you see that what happens and i have a picture here to show you but i'll tell you first what happens is the light comes in so there's those squiggly x-rays right it comes in and and there's the okay d which depends on the lattice constant right is related to the last but look now there's another plane in between there's another plane in between and in fact with the 200 planes that plane in between exactly cancels out the constructive interference here it is right so there's there's what i would have had right there's the the if you want to think about this as the 100 100 right there's the 2os okay but notice when i go from 100 to 20s i add this plane in here and and because that plane has atoms in it because that plane has atoms in it it acts like a mirror and it can also reflect right it can also reflect and so and so what happens is i would have had this nice there it is there's a picture i drew there's that that first x-ray bouncing off there's the second one bouncing off and those are nicely in phase so i would see that if that's the angle that gave that to me i'd see that in the detector but now for bcc or fcc i've got something in the middle and that something in the middle is exactly canceling out you see that so now it cancels that out in fact that's that's called forbidden you won't see a signal it's right and so and so this is called these are called selection rules right these are called selection rules and for simple cubic you can see there's nothing inside right so so there's nothing in this unit cell that could do this so everything's allowed whether there's a plane in there or not it doesn't matter the selection rule is whether it's ever allowed whether it's ever allowed and for simple cubic everything's fine because nothing would cancel out but in here you see in this 2-0 case you can see very clearly from that picture how it cancels out but there's many other kinds of kinds of angles or or planes that might also do that right and so i'm going to just give you what the selection rules are we won't go through and derive them all but let's see they are actually quite simple and so i'm going to write them down right so for if we look at allowed allowed reflections and then we look at forbidden reflections so this is what the selection rules tell us okay so if it's if it's simple cubic then it's any h k and l and there's no forbidden reflections but if we go through bcc and fcc then what we find is that for bcc the selection rule is that h plus k plus l equals even if that is even and we won't derive these but it comes from the same very simple picture i just showed you if something is in there that can cancel out the constructive interference it's going to be forbidden otherwise it can constructively interfere and that's what this tells us h for bcc it turns out to be h plus k plus l and so here what's forbidden for bcc is h plus k plus l is odd and for fcc it's h k l all odd or all even or all even and the forbidden fcc is h k l mixed odd even these are the selection rules okay all right so if i were to give you uh you know if i were to just to give you the the planes that you see then right away from an x-ray spectrum you could just use these these selection rules right away to know something right to know something about it um and if you work this out and you look at okay so we have simple cubic i mean there's this so okay so h squared plus k squared plus they have one 100 bcc fcc you're not going to see it that doesn't mean that there's no 100 plane in those crystals it just means that if you shine x-rays on it you will not see it right you will not see it okay so uh the 1-1-0 though now here okay so we so we see our oh by the way mixed even odd right and it adds to odd that's why neither one of these works here we go adds to even bcc okay but it's still mixed even on won't be fcc right three right it's not mixed so it can it can be a allowable allowable reflection for fcc but not for bcc because if you um add them up it's odd and so forth and so forth and look at seven doesn't exist because you can't do no matter how hard you try you can't get seven that's okay and there's eight and nine and so forth right so nine also yeah so so right so here it's allowed but it's not allowed in either of these because you can't get um you can't get the e you know you can't get either of these to be satisfied okay this is what selection rules give us and um and it comes again from simple uh uh oh okay well that that's another thing i said simple i have to tell you something because the brag condition it relies on an assumption that's mostly true but you know the the bragg condition requires that the reflection is independent some of you may be thinking did i did i draw that onto the atom or did i draw it in between the atoms where did that thing ruffle doesn't have to reflect off man we're not going there with the brag condition it's independent independent okay of the atom positions in a plane and the second thing which is what i've been sort of alluding to is that the atomic planes are mirror-like are mirror-like i mean this is sort of an obvious assumption since i've been assuming they've been mirrors but you know it's it's not so if you start thinking about atom position then you might go back to that you know that selection rule picture and say oh wait a second why you know does it always have to hit the atom there what if this one was over or something like that no no we assume it's just one continuous plane if there are atoms in it if there are atoms in it right if there's no atom in it then it's not a reflective plane okay but it's continuous in the assumption of of the bragg condition okay now um now we go back to our picture okay so what we want to do is again our goal our goal should we choose should we choose our goal is to go from this spectrum knowing this information right it's a copper target and and we and being able to read off the peaks our goal is to determine the crystal structure and the lattice constant that's our goal and and let me let me just write this again because it's extremely important so i'm gonna i'm gonna maybe i'll keep that one i'm gonna ignore i'm gonna erase this and put it right in the middle because this is this is what drives xrd this is what drives xrd okay which is that our goal is figure out what makes this constant always constant now you say what is this constant always okay this is the expression which i'm going to write again just for so so the what i have if it's a copper source is 1.
54 angstroms divided by 2a squared that's a constant goes in tight that equals sine squared of the theta for some plane divided by h squared plus k squared plus l squared so i'm just kind of repeating what i've said and what i've written elsewhere but that is really it that is what we do in x-ray diffraction what makes this well by this what i mean is this term on the right how do i make sure that this never changes its value because the thing on the left never changes its value right and just making sure that you've got that concept that is this oh yeah okay and and it turns out i got i got a set i got a recipe for you to follow to do this and it's on the previous slide and so so we'll start thinking about it now and we've got five more minutes and then on friday we will we will finish this and then talk about what to do with those continuous x-rays so what do i do well the the way you do this is systematically because in tight the way you do this is systematically and the first thing you do is you read off the two theta values okay that generate a set of sine squared values so the x-ray spectrum measures 2 theta but we know from here that i need sine squared okay so i'm going to write down the sine squared values okay so that's a first step so okay good right so for the first one i've got uh okay so i've got the first peak so i would start to log my data right the first peak and the 2 theta is 38. 43 and the sine squared theta okay is and i'm just going to do that math 0. 1083 okay okay good and the next one the next peak is 44.
67 and the sine squared of that is 0. 14 four four and so on so you read off all the peaks and you make that those columns okay so i've gone through one now the second one normalize the sine squared theta values by the smallest value right okay you say why am i doing that trust me this will achieve our goal this will get us there this is a nice simple recipe to achieve our goal so the next column would simply be sine squared theta divided by sine squared theta min okay well i ran out of room there but that's going to be obviously one and this would be one point three three three and so on so that's the next one so i'm just i'm just setting the the top row to one okay i'm setting the top row of 1.