pH is a useful way to measure the strengths of acids and bases. In the lab, you might use pH paper to quickly tell whether a solution is acidic or basic, based on a colour change. But what does that mean, to be an acid or a base?
Here are two of the most useful definitions. An Arrhenius acid (named after a Swedish scientist, Svante August Arrhenius) is a substance that generates H+ ions when dissolved in water. Bases, when dissolved in water, generate OH- ions.
We use the Arrhenius definition when dealing with aqueous solutions of substances. A more general definition was proposed simultaneously and independently by a Danish chemist named Johannes Nicolaus Brønsted and an English Chemist named Thomas Martin Lowry. The Brønsted-Lowry concept is based on the transfer of a proton - an H+ ion - from one substance to another.
A Brønsted-Lowry acid releases a proton. A Brønsted-Lowry base accepts a proton. The Brønsted-Lowry definition does NOT depend on the substances being dissolved in water.
We’ll discuss the chemical behavior of acids and bases more in another video, but for now, let’s focus on how we measure the relative strengths of acids and bases using the pH scale. A strong acid more readily donates a proton than a weak acid. A strong base more readily accepts a proton than a weak base.
But the amounts of protons we are talking about here are very, very small. The pH scale was suggested by Søren Sørensen, another Danish scientist. It is a logarithmic scale that makes it easier to write the very small amounts of H+ ions that are produced by acids and bases.
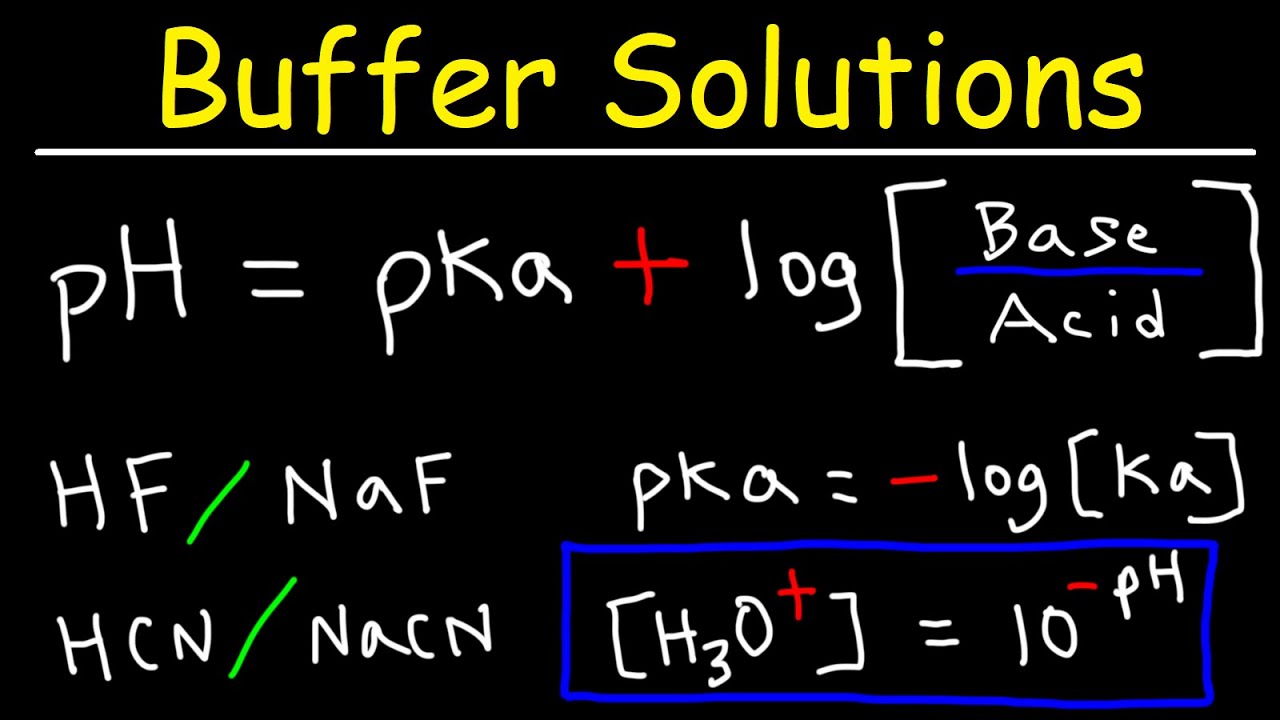
The equation is: pH = -log [H+] The concentration of H+ is in terms of moles per liter. Is water an acid or a base? A pure sample of water only slightly donates and accepts protons.
We say there is a very slight auto-ionization, or self-ionization, of water. This means that there is enough energy for a small proportion of the water molecules moving around to bump into each other and transfer a hydrogen ion from one molecule to another. That is to say, a small proportion of water molecules break up into H+ and OH- ions.
The rest of the water stays as intact water molecules. The tiny bit of H+ and OH- generated is so small, we write the concentration of H+ in terms of pH, using a base 10 logarithmic scale. The autoionization of water is so minimal that water is considered neutral (neither acidic nor basic) and it has a concentration of H+ ions of 1.
0 x 10^( -7) moles per liter. If you plug that into the equation for pH, you find that water has a pH of 7. pH = -log (1.
0 x 10^ -7) = - (-7. 00) = 7. 00 It’s a lot easier to write pH 7 than “the concentration of H+ ions is 1.
0 x 10^ -7,” but they mean the same thing. All substances that have a pH of 7 are neutral. This means there is an equal concentration of H+ ions and OH- ions.
If the pH is less than 7, it’s an acid. That’s when there are more H+ ions than OH- ions. If the pH is greater than 7, it’s a base.
That means more OH- ions than H+ ions. Another way to think about this is that the concentration of H+ and OH-ions depend on each other. The more H+ ions there are in a solution, the fewer OH- ions.
The more OH- ions there are in a solution, the fewer H+ ions. We can express this mathematically. For aqueous solutions, the product of H+ and OH- ions is a constant: “the concentration of H+ ions times the concentration of OH- ions = 1 x 10^ -14.
Take another look at the formula for pH. Notice that because of the negative sign in front of the log, the pH decreases as the concentration of H+ increases. And because the scale is log-based-10, each difference in pH represents a 10-fold difference in the concentration of H+ ions.
A substance with pH 6 has 10 times more hydrogen ions than water has with a pH of 7. A substance with pH 5 has 100 times more H+ ions than pH 7, pH 4 has 1000 times more H+ ions, and so on. Let’s see a real-world example.
Lemon juice is acidic. It has a pH of 2. What is the concentration of H+ ions in lemon juice?
We can figure this out using the equation for pH: pH = -log [H+] substitute in 2 for pH. Divide both sides by -1 so the log is on its own. -2 = log [H+] we can get rid of the log on the right by taking the antilog, or inverse log, of both sides.
This means we have 10 to the -2 on the left, and 10 to the log[H+]on the right. 10-2 = 10(log [H+]) that gets rid of the log on the right, leaving just the [H+] . 01 = [H+] We can write this using scientific notation.
[H+] = 1 x 10^-2 moles per liter. Let’s try another example. What’s something you use in your daily life that is a base?
The detergent you use to wash your clothes feels quite slippery when it dissolves in water. That’s a sign that it’s basic. The typical pH of washing detergent is 8.
5. What is the concentration of H+ ions in this solution? pH = -log [H+] -8.
5 = log [H+] Take the antilog, or inverse log, of both sides. 10^-8. 5 = 10^(log [H+]) [H+] = 3.
16^ -9 moles per liter Notice that this is much less H+ than in lemon juice. Here’s one last example. Your favourite soda has a concentration of H+ ions of 3.
16x 10^-3 moles per liter. What’s the soda’s pH? pH = -log [H+] pH = -log[3.
16 x 10-3 ] pH = 2. 5 wow, that’s pretty acidic. You drink that stuff?
Here are some average pH values of other common substances. Which ones do you find surprising?









![Intro to Chemistry & What is Chemistry? - [1-1-1]](https://img.youtube.com/vi/pdyDmXtye2w/maxresdefault.jpg)