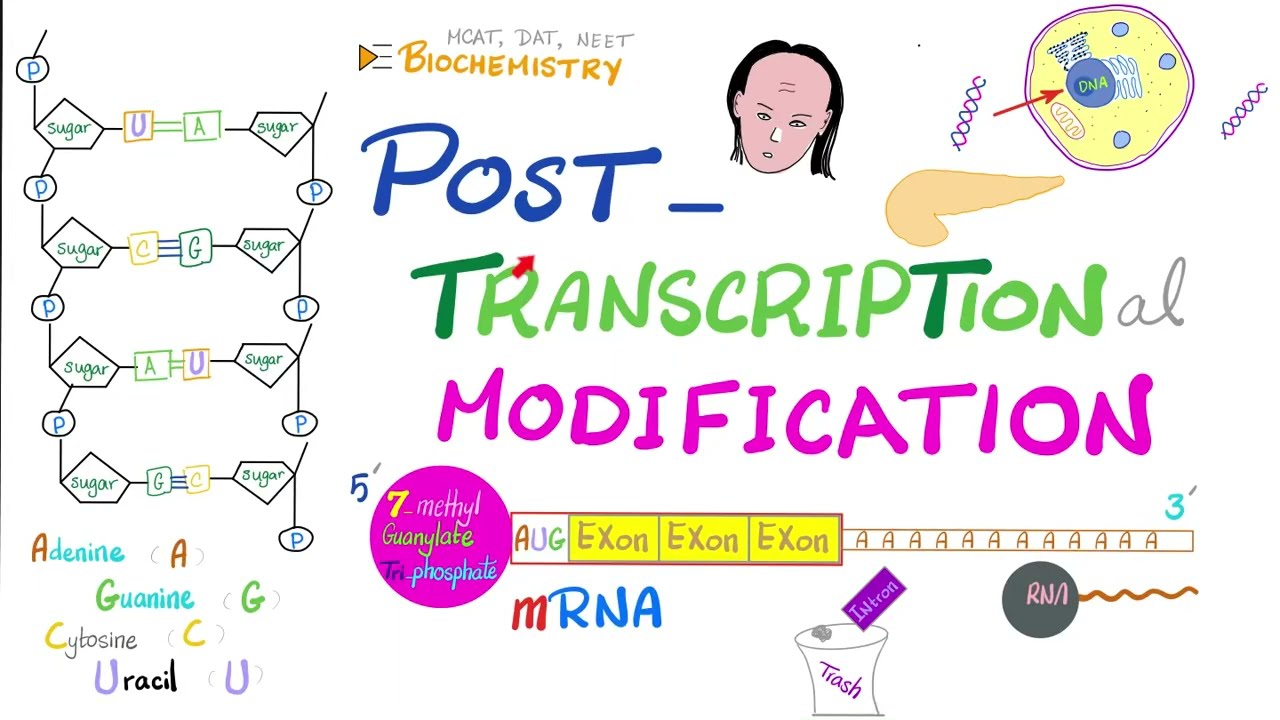
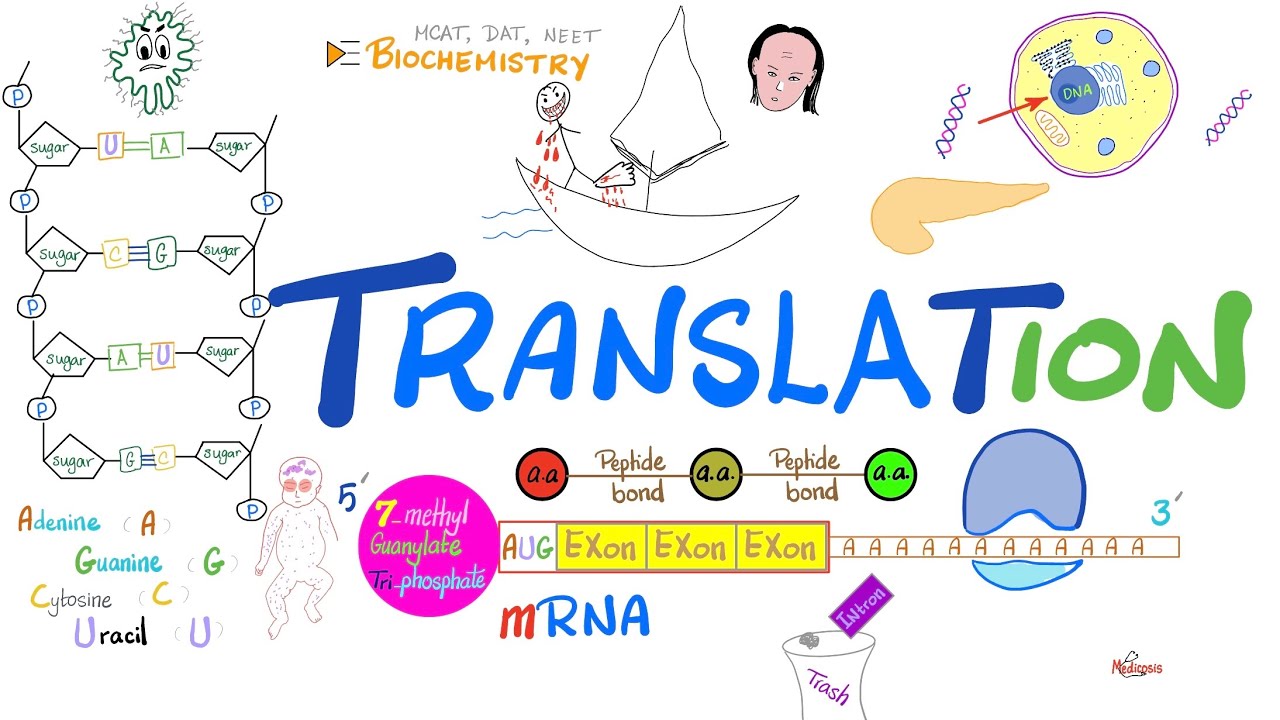
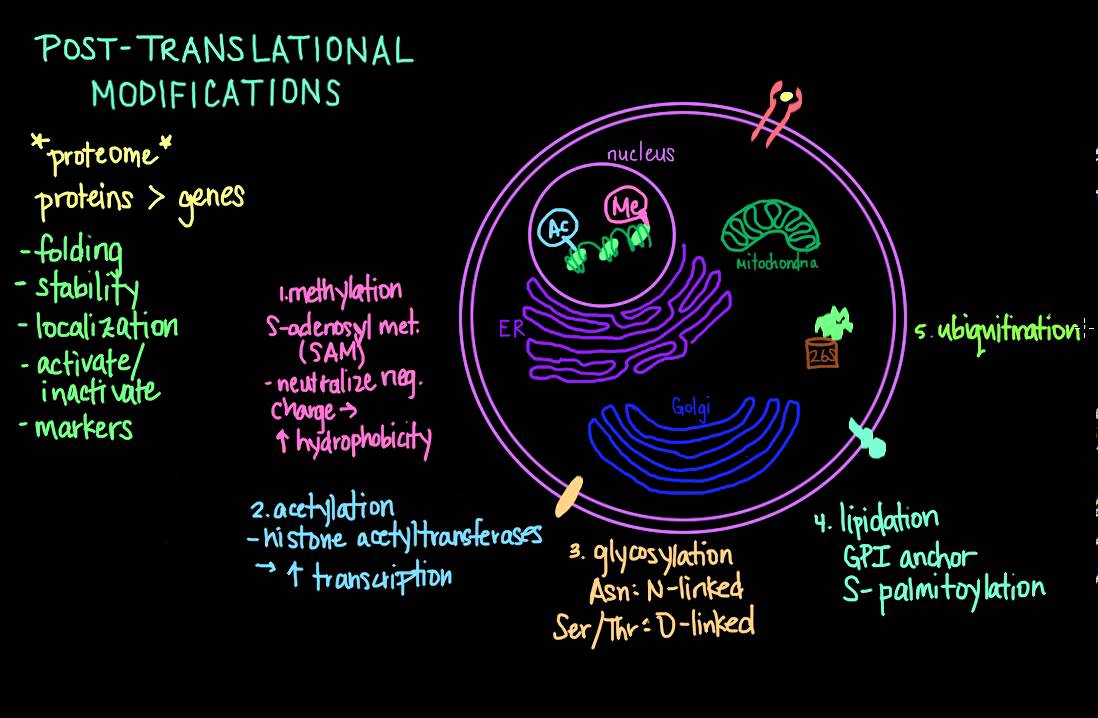
in the last few decades researchers have discovered that the human proteome is vastly more complex than the human genome post transational modification of proteins increases this functional diversity of the proteome which is the entire set of proteins expressed by a genome at a certain time the fact that there are way more proteins than there are genes demonstrates that single genes can encode for multiple different proteins and the increased complexity of the proteome as compared to the genome is facilitated by many different mechanisms including post transational modifications which help to regulate localization activity and interactions with
other cellular molecules these modifications can occur at any time during the life cycle of a protein many proteins are modified right after translation is completed and these modifications help to mediate folding into proper confirmation increase the stability of the nent protein protein or help to localize this protein to distinct cellular compartments like a shipping label other modifications occur after folding is completed and a protein has been localized to its proper cellular local these modifications serve to alter the biological activity of the protein either by activating or inactivating catalytic activity for example some other modifications serve
as markers that Target a protein for degradation and besides chemical modifications proteins can be modified via cleavage or proteolysis the key thing to remember is that the proteum is dynamic and responsive to all sorts of changes in stimuli and post transational modifications are a common mechanism for regulating these cellular activities now there are many many types of post transational modifications so I'll go over only the most common ones here and let's start with methylation methylation involves the transfer of one carbon methyl groups to Amino acid side chains by methyl transferases using S adenos methionine or
Sam as the primary methyl group donor this can neutralize a negative amino acid charge when bound to carboxilic acids and leads to increased hydrophobicity of the protein a well-known use for methylation is epigenetic regulation of transcription histone methylation and demethylation can alter the availability of DNA for transcription another type of protein modification is acetalation specifically to nitrogen atoms on a protein so n acetalation which occurs as the nent protein is being translated the end terminal methionine on the growing polypeptide chain is cleaved by methionine aminopeptidase and then replaced by an acetyl group donated by acetyl
COA via an enzyme called n acetal transferase up to 90% of eukariotic proteins are acetalated this way though the biochemical sign significance of this modification remains to be known acetalation also occurs on the liing residues of histones via the action of histon acetal transferases and this is also used to alter transcription in a similar manner to that of methylation the acetalation of histones helps to promote transcription by reducing the chromosomal condensation around these proteins the next post transational modification that can occur to proteins is called glycosilation and it is one of the most significant types
of post transational modifications because it has implications on protein folding and Confirmation distribution stability and also activity glycosilation involves the addition of a diverse array of sugar moities and ranges from simple monosaccharide modifications of transcription factors to highly highly complex branched polysaccharide modifications of cell surface receptors these carbohydrates can be added to the nitrogen atom in the side chain of asparagine residu those are UNL or to the oxygen atom in the side chains of serene or threonine residues these are oink these types of glycosilation changes these types of glycosilation changes form major structural components of
cell surface and secreted proteins the next modification lipidation is a posttranslational modification pled to Target proteins to particular membrane bound organel such as the endo plasmic reticulum ggia apparatus or mitochondria it is also used to Target proteins to endosomes lysosomes and the plasma membrane two types of lipidation modifications are GPI anchors and S palmation C terminal glycosyl inositol or GPI anchors help to tether proteins bound to the plasma membrane of the cell surface these hydrophobic Moes are prepared in the endoplasmic reticulum where they are added to nin proteins and used to localize cell surface proteins
to cholesterol or singal lipid rich areas in the plasma membrane s pilation involves the addition of 16 carbon long palmoil groups to dilate side chains of cysteine residues this modification adds a long hydrophobic chain that can be used in a similar manner as a GPI anchor it helps to Anchor proteins in the hydrophobic cell membrane next is ubiquitination which is a protein modification used to Target proteins for degradation ubiquitin is a polypeptide consisting of 76 amino acids that is attached to lysine residues of Target proteins via the C terminal glycine of ubiqutin poly ubiquinated proteins
are recognized by the 26s proteosome which is an enzyme that catalyzes the degradation of the protein and the recycling of the uquid in next up is one of the most common post transational modifications that you'll come across and that's phosphorilation which is a reversible modification that occurs principally on Serene threonine or tyrosine residues and is used to regulate proteins that play a role in a vast array of cellular processes including signal transduction Pathways the cell cycle cell growth and apoptosis protein kinases are the enzymes that help facilitate the phosphate group transfer and phosphor laes help
to remove them and lastly enzymes called proteases May remove amino acids from the amino end of the protein or cut the peptide chain in the middle a process known as proteolysis one example of this is the peptide hormone insulin which is cut twice after disulfide bonds are formed and a propeptide is removed from the middle of the chain the resulting protein consists of two polypeptide chains connected by disulfide bonds proteases also play roles in cell signaling antigen processing and and apoptosis