the discovery of the electron in 1897 by JJ Thompson marked the beginning of a series of changes in both the understanding of and in the modeling of the theoretical structure of the atom shortly after Thompson's cathode ray experiments he proposed his Plum Pudding model of the atom in 1904 which stated that an atom consists of tiny negatively charged particles called cor pusles at the time that float around a positively charged soup this model helped incorporate the electron into the atomic structure but still there was much work to be done to move towards a truer understanding
according to Thompson's model the entirety of the mass of an atom is carried by electrons meaning that for atoms to have the weight that they do there would have to be thousands of them occupying each one to add to this modeling the arrangements of these electrons proved to be a rather daunting task Thompson never claimed to be able to model such a complex in structure but did however come up with a practical experiment to try and model a simple arrangement with a few atoms in his Plum Pudding model in his experiment he used magnetic pins
pushed into corks and placed in a water-filled basin above the Basin was an electromagnet which attracted the pins towards it what he found was that as he added more and more pins they would form concentric rings around the center of the Basin which led Thompson to infer that electrons would form concentric shells around the center of the atom Thompson did further work on this model over the next year but by 1907 conflicting experiments by other scientists showed that there must be considerably fewer than thousands of electrons in atoms and even Thompson himself was convinced that
something was off with his model just one year later two physicists and a student teamed up to tackle the issue their names were Ernest Rutherford Hans Gyer and Ernest Marsden by this time Rutherford was already a well-known scientist and a Nobel laurate for his previous work with Alpha and beta rays and he was working as a professor at the University of Manchester in 1906 he was visited by Guyer and upon the visit Rutherford asked him to stay and help him with his radiation research Marsden was a student studying under Rutherford at the time Rutherford guer
and marsden's initial Quest was not to remodel the theoretical structure of the atom rather it came about when they encountered a problem in their research on alpha particles for their research they had built a device to count alpha particles and measured their overall charge so that they could then calculate the charge of a single alpha particle this counter however showed inconsistent results and rather concluded that this was because the alpha particles were being deflected by the air molecules causing them to ionize different amounts of gas molecules each time they ran the experiment this puzzled rord
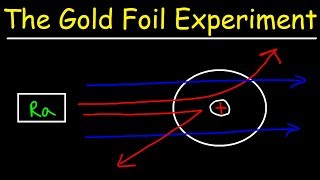
because according to the plum pudding model deflections of this severity shouldn't happen after noticing this inconsistency they turned their attention towards studying the deflection of ala particles Gyer and Marsden under Rutherford's supervision created Creed an experiment with the following setup a source of radon would emit alpha particles which would hit a sheet of metal and deflect onto a phosphorescent screen a plate of lead was placed in between the radon source and the screen so that alpha particles couldn't directly hit the screen and throw off the results a successful deflection would be indicated by a small
flash on the phosphorescent screen so the task was simply to count the number of flashes to measure the number of deflections they tested many different Metals for the deflecting plate such as lead tin aluminum copper and silver they preferred gold the most however due to its malleability and how thin they could cut it after using different metals they noticed that as the atomic mass of the material used for the deflecting screen increased the number of deflected particles also increased guer and Marsen later wanted to test for severe deflection and for that setup they used a
tiny source of radon placed on the lead plate and changed the orientation of the deflecting plate so that the screen flashes would indicate an almost full deflection what they found was that one in every 8,000 particles that hit the metal deflector bounced onto the screen as small a number as that is it was still significant because the plum pudding model predicted no deflections of this severity they published their findings on this in 1909 upon getting these results Rutherford decided to remodel the theoretical structure of the atom suggesting in 1911 that there was a concentrated positively
charged mass at the center of the atom called a nucleus and that this nucleus was surrounded by electrons and that most of the atom outside of the nucleus in the electrons consisted of merely empty space this proposed model unfortunately received essentially no praise at first with the general consensus just leading to more confusion amongst the scientific Community due to how widely varied the results of the 1909 experiment were however this work set in motion many important events to unfold over the coming years Guyer inspired by his frustrating days of counting flashes on a screen would
later go on to develop a counter that would relieve future scientists of such a grueling and tedious task known today as the guer counter also in the same year that Rutherford proposed his nuclear model of the atom he invited a post-doctoral student by the name of Neils bore to Manchester to conduct research alongside him and upon working with Rutherford bore was one of the first to drop the plum puding model and join Rutherford on his new nuclear model bore would become a key figure in giving the nuclear model mainstream popularity and would also help r
for refine the theoretical structure of the atom even further in the years to come if you enjoyed this video please consider liking and subscribing click here if you want to see more scientific progress made during this time period thank you for watching and I will see you in the next video