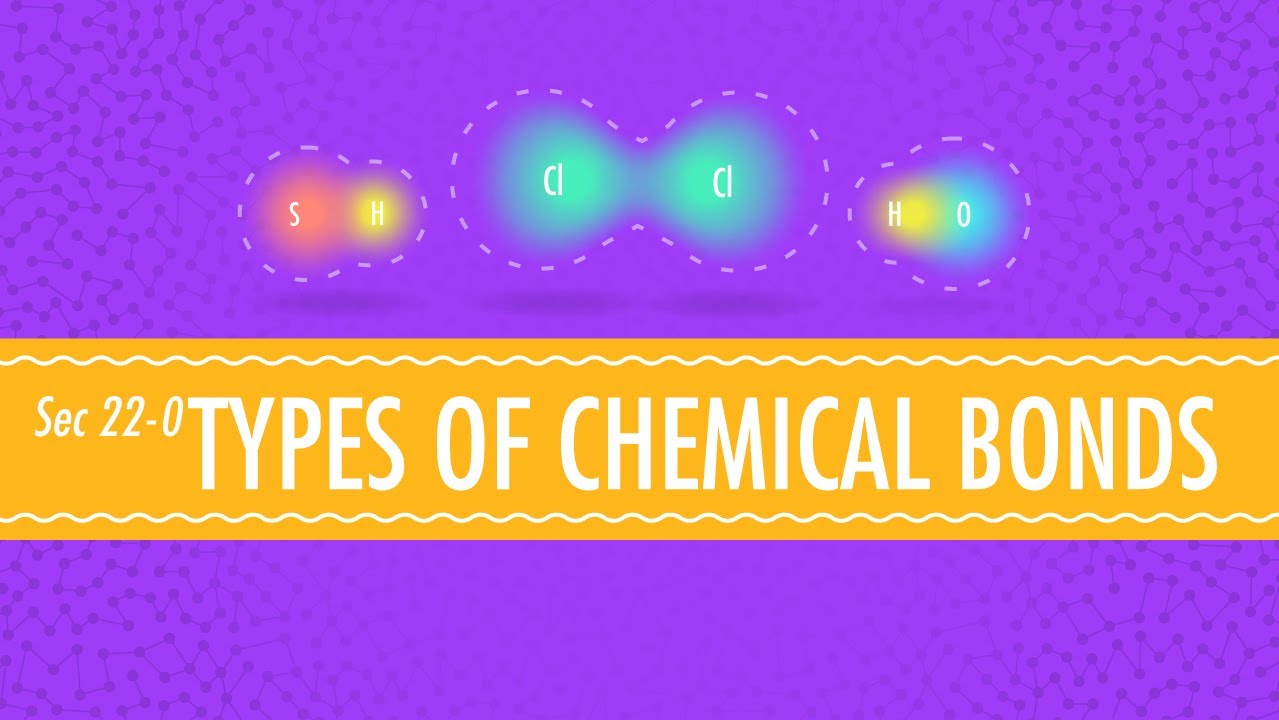
Throughout this general chemistry series, the phenomenon of chemical bonding has been a primary focus. We know that there are different types of chemical bonds, and the kind of bond that will form between atoms of two elements depends on the identity of those elements. If one element has a low ionization energy and the other has a high electron affinity, electron transfer will occur, resulting in ions, which will then interact via ionic bonds.
So for this reason, a metal and a nonmetal will tend to make ionic bonds, due to their positions on the periodic table. By contrast, two nonmetals, being from this region in the upper right section of the table, will tend to share electrons to make covalent bonds. The same can be said for a metalloid and a nonmetal, again covalent bonding will be most likely.
But what occurs between two metals? What kind of bonding is present in a substance that is made entirely of metal, whether just one metallic element or several? This is called metallic bonding.
We have previously discussed metallic bonding in passing, but now it’s time to get a closer look at this phenomenon so that we can be better prepared for more advanced topics like inorganic chemistry and materials science, as these will be important applications of our general chemistry knowledge. Metallic bonds are quite different from ionic or covalent bonds. One major difference stems from their extreme strength, which is what gives most metals such high melting and boiling points.
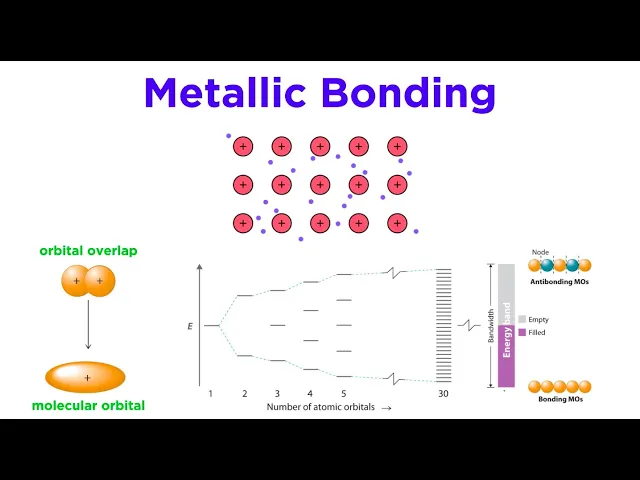
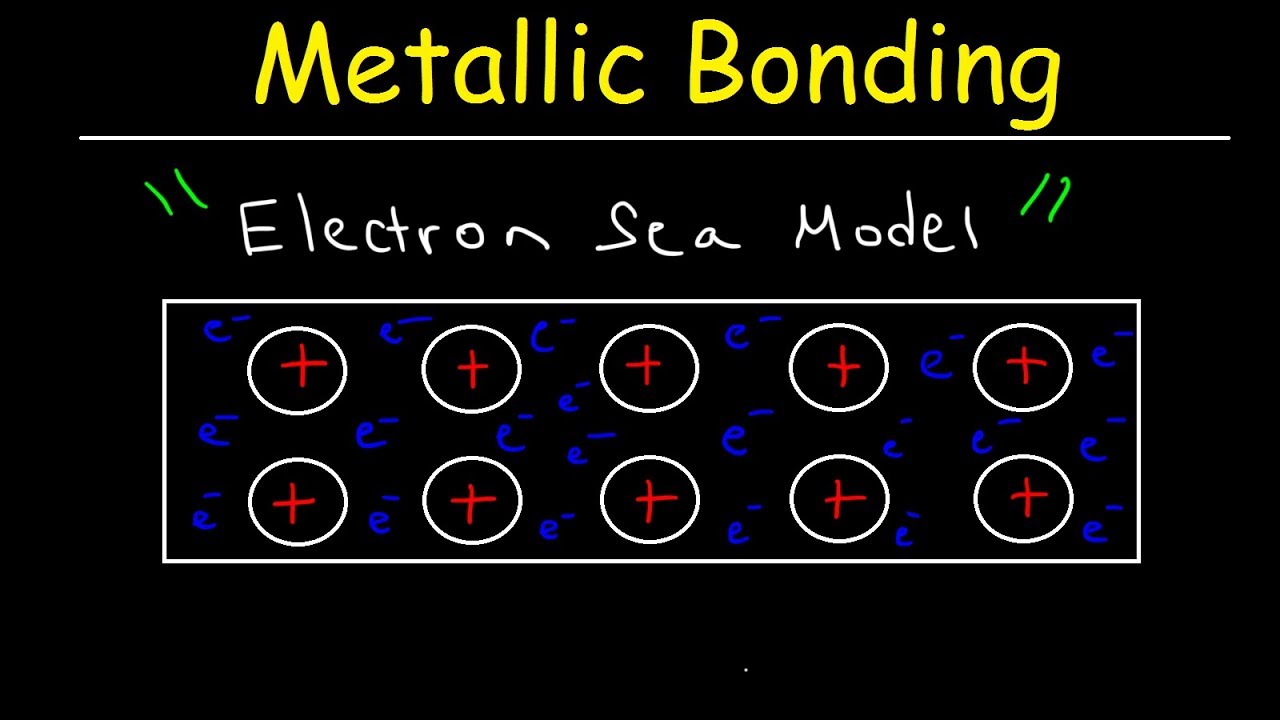
Each atom within a metal is undergoing atomic orbital overlap with many neighboring atoms, and this generates an enormous number of molecular orbitals that extend across the entire substance, holding the lattice together. Although we technically consider all of the atoms to be neutral, we can also view a metal as a grid of metal cations, with all of their valence electrons completely delocalized over the entire lattice. Electrons can move freely within the molecular orbitals throughout the lattice, forming a sea of electrons, and the metal is held together by the attraction between the nuclei and these electrons, thereby giving a metal both ionic and covalent characteristics.
Electrons dissociate from their respective atoms, like with ionic bonds, but the electrons are shared among the atoms like with covalent bonds. So this is the other major difference between metallic bonds and the other bonds we are used to. The extreme delocalization of electrons is the key feature that gives metals their signature properties, which we will discuss in a moment.
Metallic bonds can exhibit varying strengths, and this will depend on the degree of attraction between the nuclei and the electrons. This is where electron configuration becomes relevant. For example, sodium has one valence electron and therefore tends to form a 1+ cation, while magnesium has two valence electrons and therefore tends to form a 2+ cation.
Coulomb’s law dictates that a charge of a greater magnitude results in a stronger attraction, so magnesium will make stronger metallic bonds than sodium. In addition, more delocalized valence electrons will also correlate with stronger metallic bonds. We must also understand that the strength of these metallic bonds does not depend on the directionality of the bond, as their strength persists even in the liquid phase.
Gallium, for example, has such a low melting point that it will melt in your hands simply due to the warmth emanating from your body. And yet, gallium has a high boiling point typical of other metals, like copper. Metallic bonding remains strong in the liquid phase.
Continuing, we can look at common substances like aluminum and copper in order to illustrate the key properties that result from the delocalized electrons. These are malleability, ductility, and luster. Malleability describes the ability of a metal to be shaped by physical force, via hammering, pressing, rolling, and other such methods.
So we are essentially describing the metal’s ability to withstand pressure without breaking. Some metals are extremely malleable, which is why they have been so useful in building tools and weapons throughout human history. Aluminum and copper are examples of malleable metals, as are tin, gold, silver, and iron.
Ductility is somewhat related to malleability, but instead of describing the ability of a metal to be compressed, ductility describes the ability of a metal to be stretched, particularly into long wires. Again, metals like platinum, gold, and copper display high levels of ductility. Finally, luster describes the way light interacts with the surface of a substance.
This is a property that will be discussed in more detail in the upcoming geology series, due to its relevance with a wide variety of minerals. And of course as we have already learned when examining conductors, insulators, and semiconductors, it is the delocalization of the valence electrons that allows for electrical conductivity. This means that beyond raw materials for building, all of our technology is also based upon the properties of metals, and the metallic bonds that give them those properties.
Without a doubt, understanding metallic bonds is of supreme importance when it comes to materials science, engineering, computer science, and other fields that are leading us into the future.