In the previous tutorial, we talked about minerals and the different types of bonding they can exhibit. Now we are going to take that knowledge and expand upon it so that you can start identifying minerals in the wild. Geologists start with one simple property to identify minerals by eye: luster.
Luster describes the way light reflects off a mineral’s surface. A mineral’s luster should first be grouped into one of two categories: metallic or non-metallic. Luster is related to the speed that light travels through a mineral, or its refractive index, and minerals that transmit light slowly, with a refractive index greater than 3, generally have a metallic luster.
Metallic minerals are opaque with a mirror-like, shiny luster, while non-metallic minerals, which can range from opaque to transparent, tend to be less shiny than metallic minerals. There are seven types of non-metallic lusters: adamantine, vitreous, pearly, silky, resinous, greasy, and dull. Minerals with adamantine luster are transparent to translucent and are the shiniest of the non-metallics.
Diamonds, with their high refractive index, are the most well-known example, but other minerals, like Herkimer diamonds, a particularly shiny type of quartz from upstate New York, as well as the popular diamond knock-off, cubic zirconium, also get a chance to shine. Vitreous luster looks like a freshly cleaned pane of glass. These minerals are transparent to translucent and have a low refractive index.
Some of the more common minerals such as quartz and calcite have a vitreous luster. Pearly luster resembles the subtle iridescence, or play of colors, found in natural pearls. Iridescence occurs when light reflects and refracts off different crystallographic planes as it passes through a crystal.
The merging of two or more light waves that have been reflected off atomic planes creates a new light wave with a new and distinct waveform. Because the distance that a light wave travels between two layers is a function of the angle of incidence, changing it will result in different wave combinations, creating a shifting rainbow effect, where the color reflected changes based on the angle of observation. It makes sense then, that minerals with a pearly luster consist of transparent, coplanar sheets.
A common mineral with a pearly luster is muscovite, which is sometimes used as glitter. Minerals with a silky luster are composed of thin, parallel fibers, making them reminiscent of silk. If these “fibers” are particularly coarse, more like twine then silk, it is said to have a fibrous luster.
Ulexite, a rare hydrated borate, is an excellent example of silky luster. Another example is a variety of gypsum, a mineral composed of hydrated calcium sulfate, called satin spar. Resinous luster resembles hardened maple syrup or frozen pine resin.
Sphalerite, which is named after the Greek word for deceptive, due to its many different crystal appearances, can sometimes be found in nature with a resinous luster, along with metallic, or even adamantine lusters. Minerals with a greasy luster appear as though they are covered in a thin layer of oil, and many minerals with a greasy luster also feel greasy. An excellent example of this is graphite.
Comprised of sp2 hybridized carbon atoms forming an extended pi system, it both has a greasy luster and feels greasy. Minerals with a dull, or earthy luster are the least shiny of them all, as light is primarily scattered at the surface instead of reflected. The clay mineral kaolinite is an example of a mineral with a dull luster.
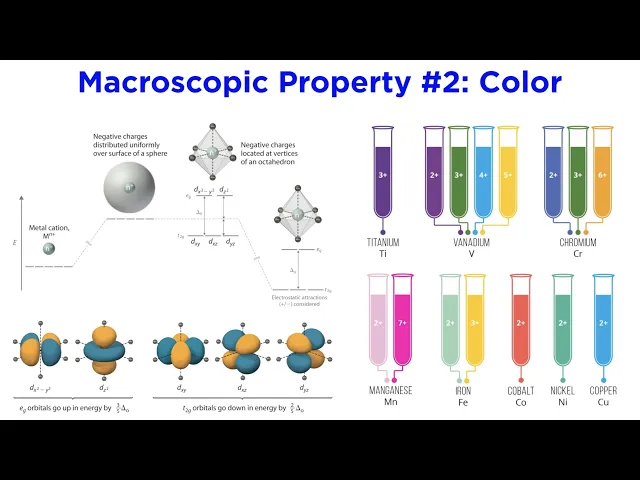
Color, one of the most obvious properties of a mineral, is also the easiest to misinterpret. Minerals typically get their coloration from the presence of transition metals. Due to their partially filled d-orbitals, transition metals are very efficient at absorbing specific wavelengths of visible light, which affects the color that is reflected off a mineral’s surface into your eye.
Allochromatic minerals only contain trace amounts of the transition metals. Corundum, Al2O3, is colorless until about one percent of the aluminum ions are replaced with a transition metal, such as iron, titanium, or chromium. Corundum with trace amounts of iron or titanium is blue in color and is called sapphire, whereas corundum with trace amounts of chromium is red, and is called ruby.
Transition metals do not color all minerals identically. For example, chromium makes rubies red, but it also makes emeralds green. Idiochromatic minerals are “self-colored” by the presence of a transition metal in their standard chemical formula.
The carbonate mineral malachite appears bright green due to the absorption of red light by copper ions, which makes up over half of its chemical composition. Other sources of color include structural defects in the crystal lattice. The mineral fluorite gets its purple color from Frenkel defects, which occur when a small ion slips out of its position, leaving an electron behind in its hole, which can absorb and reemit light.
One of the ways that color can be useful for identifying a mineral is by observing its color in a powdered form, which is done by a streak test. The streak is the powdered residue left behind after rubbing a mineral on a piece of unglazed porcelain. The streak of a mineral is much more consistent than its macroscopic color.
This is because when a non-opaque material is finely powdered, most of the light is reflected off its surface, rather than being transmitted and subsequently absorbed by its electrons. Most non-metallic minerals have a white streak, while metallic minerals tend to have dark-colored streaks. For the ferric oxide mineral hematite, the streak test is one of the quickest ways to identify it.
Although it can appear black, brown, or even silver in color, its streak will always be a reddish brown. Similarly, calcite, which can appear in a range of colors from pink to blue, always has a white streak. If you want a way to tell the difference between pyrite, also known as fool’s gold, and real gold, pyrite always has a dark gray streak, while gold has a pale-yellow streak.
That covers two very important macroscopic characteristics of minerals, luster and color. Let’s move forward and learn about a couple more.