[Music] hello today we start with the lecture one uh intermolecular forces so intermolecular forces the forces between the two molecules uh I have pasted two examples one is the nucleus which is having Neutron proton and electron and they are very well organized through the intermolecular forces another example is a biomicro molecules uh this is a DNA so you can see different molecules are organized through various hydrogen bonds and other intermolecular forces if you remember the DNA is organized as a helical structure so this is stabilized by the various intermolecular forces also if you just see the protein they have primary structure secondary structure tertiary structure and quary structure and all the structures are stabilized by different intermolecular forces so in this lecture we'll just see what is the origin of this intermolecular forces what is it historical perspective of this intermolecular forces and also we'll see few examples how intermolecular forces are calculated how what are the functions so let's start with the first uh in the nature uh various forces we used to experience and if you understand this code so intermolecular forces embrace all forms of matter so in any form of matter always the intermolecular forces are involved so we can classify four forces two are the strong and weak interactions second is the electromagnetic and gravitational interaction the first is strong and weak interactions so this acts between the neutron proton electron and other Elementary particles and these interactions have a very short range of action less than 10 the^ minus 5 nanometer and belong to the domain of nuclear and high energy physics other two forces are electromagnetic and gravitational interactions so they act between atoms molecules and also sometime between Elementary particles and these forces are effective over a much larger range of distance it may vary from a subatomic distance to a infinite distance so these are the four different type of forces which embrace all forms of matter and these intermolecular forces ultimately determine the property of a solid a liquid a gas behavior of particles in a solution different types of chemical reactions and as I told organization of biological structures like DNA RNA and proteins carbohydrates so ultimately if we see the intermolecular forces are very much responsible in all the aspects so these are few illustration of intermolecular forces the first one is you can see a nuclei where protons and neutrons are there in the nucleus so here this protons and neutron they combine the strong nuclear interactions also they are surrounded by the electrons but electrons can leave the orbit they can go to other orbit as they are interacted through a weaker interactions or weak interactions so this is a very classical example of the strong and weak inter action the nucleus itself the interaction between proton and neutron is the strong one and interaction with electron is the weaker interaction another example is the gas and liquid so in the in case of gas they are having sort of repulsive energy so they live in the gaseous phace but when they try to attract each other the cohesive forces strong start working there so the gas condens to the liquid so in the liquid they are attracted together but in the gas at the same time they are experience the repulsive forces another example this is the earth we can see various pictorial representation we start with the Galileo so what Galileo tried he threw two different balls when he was throwing two balls he understood that there is a gravitational force also arodes he saw if you put something in another food you you will experience the bcy forces classical example of Newton if something falls from the top so this was the example apple is falling from the tree apple is falling from the tree this is the example of again gravitational forces so gravitational and intermolecular forces it acts together to determine the maximum possible size of building mountains trees and animals are you can see a system so these two forces gravitational well the intermolecular forces they decides about the tights they decide what should be the height of a particular building this decide what maximum size of a particular animal how the satellites work so basically if you understand these forces so they acts in all form of matter whatever we see in the nature so this is the historical perspective so earlier uh before 1500 or 1600 of course there were all intermolecular forces they are also being experienced also sometimes for the calculation sometimes for the ayura medicines but only like now they were not generalized they were not well documented their function were not defined so if you just see the typical example of Newton it doesn't mean that before Newton no one experien the apple is falling from the tree but that phenomena that observation was not generalized so it 17th century is the first considered as the first scientific method where we started with the Galileo then Bo law the Newtons they derive few breakthrough Theory B law he derived the gas law pval to RT constant so here gas law it says interaction between gas molecules are repulsive in nature but when Newton discovered the law of gravity so he always say there there is always attractive interaction between two different molecules the next comes your 18th century because of this contradiction between repulsive and attractive forces there have been lot of confusion and contradiction in the 18th century but of course we got few mathematical theories from the llas ER and colums so this this century is basically known as some mathematical methods as well as this Newton's principia followed by this uh 18th century in the 19th century we had very good breakthrough starting from Youngs then classes Maxwell Wonderwall gifs Bullman so they derived basically the kinetic theory as well as the thermodynamics to understand how this intermolecular forces affect the behavior of a system this is one example of uh wonder wall equation so if you just see the gas law and the wonder wall equation SO gas Li is applicable only for the gases but in the wonder wall they have added extra term this is for the attractive term this is for the repulsive term in the same equation now this equation is called the equation of state it means it can apply to the both gas as well as liquids Then followed by in the 20th century we had lard Zone potentials and other law so so here uh the potential is defined as a function of mass between two as well as the distance between two and later on if you if you see in the next slide so we'll have both the terms in the same potential it will have both attractive as well as the repulsive terms so I'll just give one example of attractive forces attractive forces is simple gravitational forces one object is M other object having Mass M2 they position that is distance R the simple force between them will be M1 into M2 upon r² into g g is the gravitational constant so this is simple attractive Force now as I told B law it says the gas is repulsive if the gas is is repulsive then how there will be formation of solid and liquid it means from repulsive again it is going to the attractive force between gas molecule so they form liquids again attraction they go to the solid but after formation of solids it does not vanish it means again it's not attractive forces does not keep continuing it means that's why the universe or the system exists so it is like repulsive attractive and again repulsive it is a balance between these two forces one classical example of this attraction and attractive and repulsive forces is capillary if we just see capillary so there are two kind of interaction one is interaction between liquid liquid interaction between the liquid liquid molecule and second is interaction between the liquid solid interaction so this this is called coh this is adeson so if you just see put the capillary tube of any wall thickness you are going to have the same height capillary rise so the interaction between liquid liquid it prevent the capillary rise but at the same time interaction between liquid and solid promote the capillary rise so this is classical example where we are seeing both adhesive as well as the cohesive forces and in the 20th century various intermolecular interactions and bonds where considers their function or Expressions were defined is starting from this ionic interactions ionic bond metallic bond Wonder Walls forces hydrophobic interaction hydrogen bonds and the solution forces so this is a overview of historical perspective of the intermolecular forces so the 17th century is known as scientific method or first scientific method 18th century is known as Newton principia 19th century is known as mathematical methods 20th century is known as this kinetic theory and thermodynamics and 21st Century We are continuing the continum theory and collidal Theory so we'll just see one example let's say this is your gas law pbal to constant so if we write P inversely proportional to 1 upon V to the^ M we are giving a random number M here one upon V you can write this is a density and expression for the repulsive forces between two molecules is given by C is a constant C is a constant and R is distance between the r is the distance or you can say intermolecular distance so this is a typical expression for the pive forces now let's see an example we are having a cubical space this Dimension is L and we it is filled with few gas molecules whose radius is R the total volume of the cube or system will be L to the^ 3 and volume of each molecule will be proportional to the R so if you want to calculate how many number of molecules are present we can simply divide the volume of these two so these L CU upon R Cube number of molecules are present in the system now if you are talking about the pressure pressure is always applied on the area or one side or one face of the system so how many number of molecules are present at each side of so area of each side will be the L Square so number of molecules will be l s upon r² so repulsive forces between two molecules is defined as C into r^ n if you are having this this number of molecules the total force will be this multiplied by this it will be C upon r ^ n into l s upon r² now if I want to calculate the pressure I have to divide this Force divided by the area and this area is L squ I'm considering one side of this Cube so it is coming like C upon R to the power n + 2 now let's go to the experimental what gas La suggest G gas La suggest V = 1 upon V and V can be written in terms of R Cube so this will be 1 upon r^ Cube so if I compare this R the power n + 2 and R the^ 3 so it says n + 2 = 3 that means n = 1 so for this repulsive nature n is coming to the one further if you want one can generalize this n = 3 m + 2 so here for the gas law value of M is equal to 1 so if you put one here so n will be coming again one so this is this proves that there is a repulsive interaction between the gas molecule we are having this repulsive interaction between the gas molecule so next we'll start uh followed by this we started with model interaction potential in the 19th century which says the potentially simple function of mass between two and inversely proportional to the intermolecular distance between two this is very much known as the gravitational force G gravitational potential G M1 M2 upon R where G is the gravitational constant it's value is written here so from this the model interaction potential was defined as you have taken G as a constant c g was replaced by C then m M1 M2 and we put the r to the power n n is another constant so this is the potential and typically if you want to calculate the force you have force is the derivative of the potential so f r is simply a derivative of potential so if you derite this you'll get n c M1 M2 r the^ n + 1 now depending on the value of n in this equation if I put n = 1 if I put here Nal to 1 then f r Will beus C M1 M2 on r² if you go to the last slide this is same gravitational force so what it suggested depending on the value of n we can decide which type of intermolecular interaction is this like for n isal to 1 it is a gravitational force so typically gravitational force is always attractive gravitational force is always attractive in nature so typically uh if you go for and also attractive plus it is also a long range Force so gravitational force attractive as well as long range Force so if you go for n equal to 1 so it goes for the long range forces typically the value of n lies in the range of 4 to 5 for the short range intermolecular forces so that we'll discuss in the next slide now one can calculate the total interaction energy let's say in the system one molecules is going to interact with the all other molecules in the system so total interaction we have to integrate this is the size of one molecule size of molecule diameter of the molecule and L is the the size of system so total number of particles will be let's say this is we are talking interaction at R this is r and this is the Drso at this distance we are consider considering the interactions it will be 4 Pi r² into Drhow many molecules if you multiply with the number density so these number of molecules are interacting this is the potential between two molecule WR R and we have to integrate from this size Sigma to the L now w r potential value if you just remember the last slide you can write w r as - c upon r^ n so here if you write C upon R the power n so it will turn out to be 4 Pi z r to the power 2 - Y into Drso if you integrate this very simply so you are going to get - 4 Pi z n - 3 Sigma to the power n 3 1 - Sigma upon L nus 3 so again if we just see the total interaction energy is again depends on the value of n so if value as value is n of n is greater than three so we can uh ignore this term so it will be simply like 4 Pi 0 N - 3 Sigma to the^ n minus 3 and this is very much understood that always size of particle will be much much lesser than the the size of system or you can see here Sigma upon L is less than one so here again depending on the value of n one will be able to understand whether it is a short range interaction or the long range typically if it is one it is a long range if it is three it is at the boundary between short and long range and if it is greater than 3 4 and five it goes for the short range intermolecular forces n isal to 1 n = 3 Nal to greater than 3 let's say 4 five and so on so this is called long range this is at the boundary and this is the short R so typically depending on the N value of y we we will be able to define whether it is a long red or it is the at the transition between s and long range or it is the short range intermolecular forces so this is very good example if you just see uh gas laes it it is having the repulsive interaction between the gas molecules now if you can modify the gas law through the intermolecular interactions so it can be applicable for the both gases and as well as the liquids because in the gas it says it is a rep repulsive interaction but at the same time in the liquid it says it is coming through the attractive interactions so considering this uh Wonder Walls considered the attractive effect between the molecules and he just modified so he added two terms P he added a term a attractive term at the same time in the B volum he substracted the terms B so this is a modified gas law proposed by the ball also known as the ball interaction a ball equation of state so equation of State means it can be applied to the both gas and liquids so as I told this A+ v² accounts for the attractive intermolecular forces now if you remember we calculated the potential in the last slide so it says total energy is the 4 Pi C / by nus 3 Sigma to the power nus 3 4 < c row / N - 3 Sigma to the power N - 3 so this is the potential energy between the two molecules if you want to consider for one molecules you have to divide by two so if you divide by two it will be same as the value of a a which is coming here as the attractive Force so if you just see the value of a is very much coming from the total interaction energy between the molecules so a is equal to so this will be now it will be the two 2 pi and of course nus 3 Sigma to the^ nus 3 so it can be further broken so you can write C / Sigma to the power n into 2 < Sigma to the^ 3 will go up side/ nus 3 so it says again it is coming to the same expression of the potential derived here we says potential is a function of C upon r the^ n so this is the potential or you can say this is w0 and this is the molecular volume so the a is coming simply from the intermolecular interactions which you can see is proportional to the inter molecular potential into molecular volume divided by nus 3 and simply B is the simple volume function so for half of the you can say this is V by 2 so V will be 4 upon 3 p Sigma Q / 2 so again this will be the 2x 3 Pi Sigma Q so this is how these two terms is is coming in the Wonder equation and again this is very interesting example gas is only considers the repulsive intermolecular forces but the equation of State considered both attractive as well as the repulsive intermolecular forces so term a considers comes for the attractive correction for the attractive forces and B is the correction for the repulsive interactions so as we see in most of the cases uh we are going to have both attractive as well as the repulsive intermolecular forces so my in 1903 propose very interesting pair potential which accounts for both attractive as well as the repulsive term so one term is attractive other term is repulsive in this and this is also known as the my potential and here the constant A and B can be related to the A and B of the wonder wall forces or wonder wall equations later on this Leonard Jone modified the value of N and M he proposed the value of 6 n = 6 and M = 12 so this is for the attractive term and this accounts for the repulsive ter so this is very uh very good to see that especially this depending on the value of yan or you can say the Y this pair potential can be applied for the different systems so uh we'll see in subsequent slides okay so we'll just discuss one example here uh so this is a typical enerion potential it says this is the attractive term this is the repulsive term and typical value of a and b let's consider 10 the power -77 and 10 the powerus 134 so we'll just see uh how this Force are potential vary with the distance so with the above value of A and B can we calculate the what is the minimum value of potential and what will be the maximum adhes Force the two atoms and whether whatever the force is coming between the two atom can it be measured experiment ly so this is uh force and energy as a function of intermolecular separation or the inter molecular distance so if the distance is high so before that I'll just say the positive is the repulsive negative is the attractive these are the attractive and these are the Rive range okay so if the distance is very high a very high means it's a larger distance so for the larger distance between two molecules are interatomic so we are going to have the attractive forces so attractive forces is keep on increasing up to the here or you can say attractive potential is increas increasing up to the here and suddenly if you come very closer it suddenly goes to the repulsive side so here is the transition it goes to the minimum so minimum means be the equilibrium equilibrium between the two particles where we are going to have the minimum energy or the minimum potential this is the equilibrium point if you come even closer to the equilibrium again it will go to the very much to the repulsive side so once this function is known for the potential the similar function we can calculate for the force simply F minus DW upon Drso the first part was what should be the W minimum so for w minimum you know uh you want to calculate the Minima of a function then w r minus a upon r^ 6 + P upon r^ 12 you want to calculate minum you have to say DW upon Dr= to Z so DW upon DrR will be uh 6 a upon R the^ 7 minus 12 B upon r^ 13 and if you put the this value as zero so we can simply get the value of R so R will be if you compare this 12 B upon 6 a 12 B upon 6 a to the power 1 upon 6 so you can say 2 B Upon A to the power whole 6 and so its value is coming out to point 355 nanometer this is also called the re e equilibrium as I told where the minimum force come that is known at the equilibrium point so it is coming 355 nanometer now once R is known we can calculate W re so here W in the same function in place of R we have to write the r and we can calculate the value of w minimum and is coming out to be - 2. 51 into 10 ^ -21 J but it is asked to calculate in terms of KT so KT is the thermal energy KT is we say KT as see K T thermal energy and value of K is 1381 into 10^ minus 23 J per Kelvin and temperature in the problem was given as uh 298 K T was given as 290 8 K so this energy can be calculated in terms of thermal energy so it is coming out to be 61 KT this is the first part now if you remember the problem the second part says what should be the maximum addition force between the two atoms so again you have to do follow the same exercise as we did for the Minima so let's start with the value of f so f is defined as minus DW upon Drso f r will be 6 a upon r ^ 7 - + 12 B upon r^ 13 so again you have to calculate here d f r upon Dror you can simply say you have to calculate D2 W upon Drr² that is equal to Z and if we solve this so This r value will says the value here will have the maximum Force so if I integrate it further so and put it zero so you'll get Ral to 26 B upon 7 a to the^ 1x 6 6 B upon 7 a ^ 1X 6 and this R is known as the RS RS means separation the force the distance where the separation between two molecules will take place and it is coming out to be 3935 nanom 3935 nanom if you take the ratio of re and rs re means the value where there was the equilibrium means potential was Zero RS is the distance where the maximum force is there ratio is coming to the 1. 24 this is a typical value we always consider R upon RS as 1.
24 so of f Max you can calculate the value of f what you have to do here in place of R you have to write the value of RS so its value is coming out to be uh 1. 89 into 10^ - 11 Newton or you can see you can say 18. 9 P Newton now this value is coming 3935 3935 just see very interesting at nanometer level at 35 it was having the minimum value of energy but at the same time we are observing separation at a potenti maximum Force at 3 935 just see this is if you just see 3539 it's not much difference also we'll just see the value the distance at which we are going to have the zero potential so if you want to just say the zero potential means the value of WR R should be zero if you simply put the value of w r should be zero so the r not will be here B Upon A to the^ 1x 6 it's very simple R not will be B Upon A to the power 1 by 6 and if you calculate this R kns it's coming to be 1 Point R not this R not one need to calculate and the ratio of R upon R not is coming to the 1.
12 so we got two ratio r e upon R KN 1. 12 and rs upon r e = to 1 24 if you just see this is just a factor of one even not a factor of two so here very interesting observation so if you just see the LaRon potential at this distance potential is zero at the same time at the factor of 1. 12 its value is the Minima at the same time at the factor of 1.





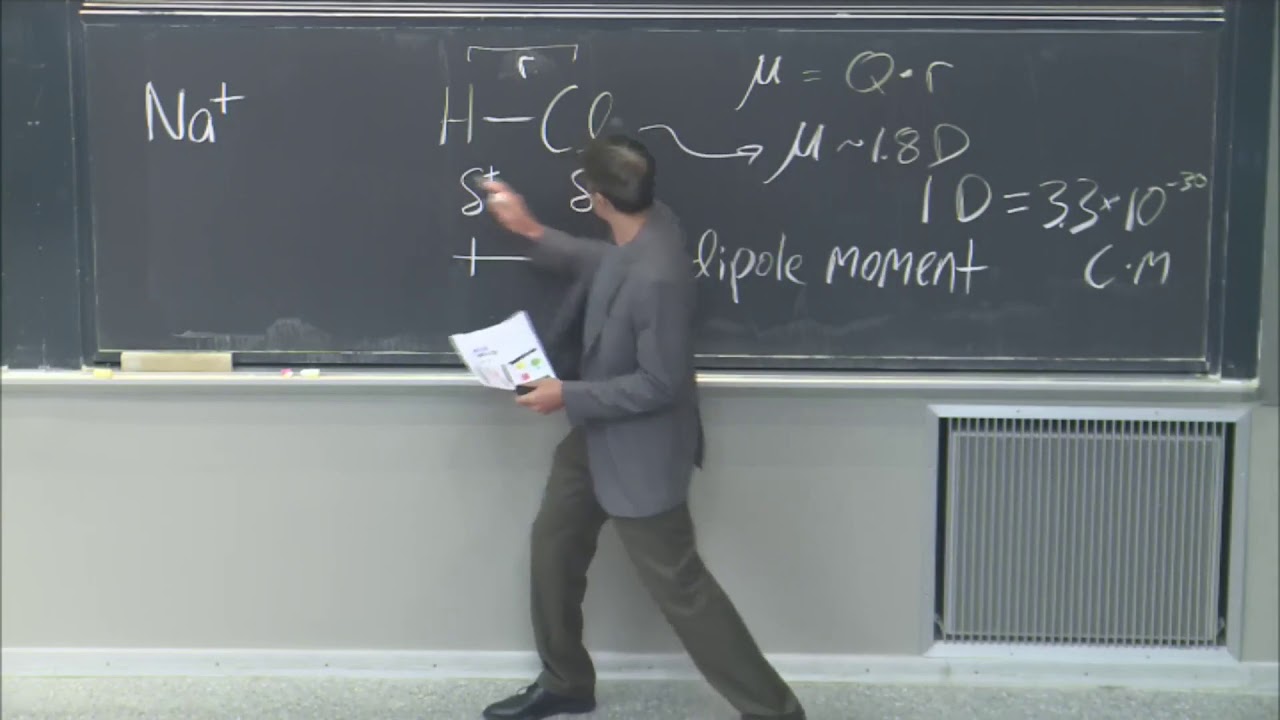






![Ludovico Einaudi [relax, study, sleep, soft, calm, inspired piano medley]](https://img.youtube.com/vi/DIvewN35QT4/maxresdefault.jpg)


![5 Pieces by Hans Zimmer \\ Iconic Soundtracks \\ Relaxing Piano [20min]](https://img.youtube.com/vi/Os47nMrjw_Y/maxresdefault.jpg)





